Abstract
Objective
To measure the intra-fraction displacements of the mediastinal metastatic lymph nodes by using four-dimensional CT (4D-CT) in non-small cell lung cancer (NSCLC).
Materials and Methods
Twenty-four patients with NSCLC, who were to be treated by using three dimensional conformal radiation therapy (3D-CRT), underwent a 4D-CT simulation during free breathing. The mediastinal metastatic lymph nodes were delineated on the CT images of 10 phases of the breath cycle. The lymph nodes were grouped as the upper, middle and lower mediastinal groups depending on the mediastinal regions. The displacements of the center of the lymph node in the left-right (LR), anterior-posterior (AP), and superior-inferior (SI) directions were measured.
Results
The mean displacements of the center of the mediastinal lymph node in the LR, AP, and SI directions were 2.24 mm, 1.87 mm, and 3.28 mm, respectively. There were statistically significant differences between the displacements in the SI and LR, and the SI and AP directions (p < 0.05). For the middle and lower mediastinal lymph nodes, the displacement difference between the AP and SI was statistically significant (p = 0.005; p = 0.015), while there was no significant difference between the LR and AP directions (p < 0.05).
Conclusion
The metastatic mediastinal lymph node movements are different in the LR, AP, and SI directions in patients with NSCLC, particularly for the middle and lower mediastinal lymph nodes. The spatial non-uniform margins should be considered for the metastatic mediastinal lymph nodes in involved-field radiotherapy.
Radiation therapy is a dominated therapeutic modality for non-small cell lung cancer (NSCLC), and improves the long-term survival and local control rates in patients with locally advanced stages (1, 2). Elective nodal irradiation (ENI) was once regarded as an essential component in definitive radiation therapy for locally advanced NSCLC. Despite the controversies over the range of the irradiated regional lymph nodes, some studies have suggested that ENI has no superiority over the involved field radiotherapy (IFRT) technique, in which only the metastatic lymph nodes are irradiated, with respect to local control and survival rate (3-5). Therefore, ENI has been extensively replaced by IFRT (6-8). Although the irradiated volume is reduced in IFRT, the overall survival and local control rates by IFRT are equivalent and even superior to those by ENI. Meanwhile, IFRT will deliver a higher dose to the lesion while decreasing the normal tissue toxicities.
Three-dimensional conformal radiotherapy (3D-CRT) may allow a dose escalation without excessive toxicity, and improve overall survival rates (9). With the implementation of 3D-CRT, radiation oncologists are now able to deliver radiation with a more precise dose distribution, providing a higher overall dose to the tumor while limiting radiation to normal tissue. Even with the ability to improve radiation dose conformity, the margins are still necessary to account for both the setup error and organ motion. The application of 3D-CRT underlines the importance of accurate quantification of the tumor movement. The modern radiation planning systems are therefore evolving to visualize the tumor motion and prevent geographical miss.
Generally, to generate the planning target volume (PTV) in radiotherapy for NSCLC, an expanding margin of 10-20 mm is added on the clinical target volume (CTV) to account for the tumor movement as well as the set-up errors (10, 11). The positions of the lymph lodes within the mediastinum are easily affected by the respiratory movement and heart beat. The lymph node movements will in turn affect the accurate delivery of radiation to the targets. Conventionally, a symmetric margin of 5 mm is considered adequate to account for the lymph node mobility. However, the respiratory mediastinal lymph nodal motion is not necessarily symmetric: it may differ among the lymph nodal regions; and it may be highly variable among individuals (12, 13). Thus, the population-based margins may compromise the dose received by the tumor or increase the severity of radiation side effects on the surrounding normal tissues.
Accurate knowledge of the mediastinal lymph node displacement is important to derive a treatment plan to deliver a homogeneous irradiation to the highest feasible dose, and to maintain the normal tissue doses within the desired constraints. This will improve the overall treatment efficacy. In previous studies, the investigators have studied the lymph node movements using the X-ray fluoroscopy, co-registered computed tomography (CT) scans, and the inhale and exhale CT scans (14-16). The conventional imaging methods are often unable to provide accurate delineation of the tumor size, location and movements.
Four-dimensional computed tomography (4D-CT) is a new imaging technology used for the assessment of the anatomic translation and the deformation during respiration. 4D-CT images can effectively eliminate the motion artifacts caused by the respiratory motion, thus reflecting the motion of the tumors and organs throughout the respiratory cycle (17-21).
The purpose of the present study was to investigate the displacements of the mediastinal metastatic lymph nodes at different respiratory phases using 4D-CT. We further analyzed the differences in the directional displacements of the mediastinal metastatic lymph nodes. In addition, we attempted to investigate whether the ipsilateral diaphragm movement could predict the movement of the mediastinal lymph nodes in the superior-interior direction. We anticipate that our study would provide evidence for the definition of internal target volume (ITV) for metastatic mediastinal lymph nodes.
Institutional Review Board approval and informed consent were obtained for the present study. The patients with pathologically proven NSCLC who underwent 4D-CT at the Department of Radiology, Shandong Tumor Hospital between September 2009 and May 2010 were included in the present study. All the patients had positive metastatic lymph nodes defined as a short-axis diameter ≥ 1 cm on the contrast-enhanced CT images.
During simulation, all the patients were immobilized using the vacuum bags in the supine position with their arms raised above their head. For each person, a thoracic 4D-CT scan was performed under uncoached free breathing using a 16-slice Brilliance big bore CT scanner® (Philips Medical Systems, Inc., Cleveland, OH, USA). The total scanning time was > 60 seconds. In order to clearly identify the mediastinal metastatic lymph nodes, 100 mL of the contrast agent labeled as Iobitridol® was injected (350 mg iodine/mL; Guerbet, Roissy, France) intravenously to each patient within 50 seconds prior to 4D-CT. During the 4D-CT scanning, the respiratory signal was recorded with the Real-time Positioning Management gating system® (Varian Medical Systems, Palo Alto, CA, USA) by tracking the trajectory of the infrared markers placed on the patient's abdomen. The respiratory signal was sent to the scanner to label a time tag on each CT image. The reconstructed 4D-CT images were sorted into ten respiratory phases on the basis of these time tags using Advantage 4D software® (GE Healthcare, Waukesha, WI, USA). The 0% phase corresponded to the end-inhalation and 50% phase corresponding to the end-exhalation. The 4D-CT images were reconstructed using a thickness of 3 mm and then transferred to the Eclipse treatment planning system® (Eclipse 8.6, Varian Medical Systems).
The mediastinal lymph nodes were classified according to the definition of Mountain and Dresler (22), and were delineated following the atlas from the University of Michigan (23). The mediastinal lymph nodes were divided into three groups: the upper mediastinal compartment included lymph nodes from stations 1, 2, and 3; the middle mediastinal compartment included stations 4, 5, and 6; and the lower mediastinal compartment included stations 7 and 9.
The image of the 0% phase was regarded as the baseline, whereas the other phases were overlaid on the baseline image. The mediastinal metastatic lymph nodes and the dome of the ipsilateral diaphragm were delineated on all the images of 10 respiratory phases. The range of CT image display window was chosen as 40-390 HU (24). In order to avoid the inter-observor differences in target volume contouring, only one radiologist was used (Suzhen Wang). When several small nodes were present in a specific station, or several lymph nodes were fused together, the conglomerate volume was contoured. The lymph nodes located at the borderline between two stations were counted to the station in which the majority of the lymph nodes were located. Figure 1 shows the lymph node contours of 10 phases that were mapped on the CT image of the 0% (end-inhalation) phase.
The position of each metastatic lymph node was represented by its geometric center of the volume, which could be calculated by the planning system. The positions of the lymph node and the dome of the diaphragm were compared in all 10 phases of 4D-CT. The lymph node displacements were measured independently in the three directions of the patient coordinates: left-right (LR); anterior-posterior (AP); and superior-inferior (SI). The displacements of the dome of the diaphragm were measured in the SI direction. The largest lymph node displacements in the three directions obtained were denoted with LR, AP, and SI, respectively. The 3D range of the motion of one lymph node was arbitrarily represented with the following quantity √LR2 + AP2 + SI2 (16). The displacements of the metastatic lymph nodes in the different groups were compared and the correlations between the displacement of the mediastinal lymph nodes and the motion of the dome of ipsilateral diaphragm were analyzed.
All statistical analyses were performed using the commercially available software SPSS 17.0 package (SPSS Inc, Chicago, IL, USA). The Wilcoxon rank-sum test was used to assess the lymph node displacements between the various directions in the same group, or in the same direction between different groups. A Pearson correlation analysis was utilized to study the relationship between the mediastinal lymph node displacements and the dome of the ipsilateral diaphragm movement in the SI direction. A p value of < 0.05 was considered to be statistically significant.
Twenty-four patients with NSCLC were included in the present study, consisting of 16 males and eight females. The average age was 60 years (age range, 39-80 years). Of these 24 patients, adenocarcinoma was diagnosed in 18 patients, squamous cell carcinoma in four, and adenosquamous carcinoma in two patients.
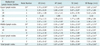
The displacement was measured for 53 mediastinal metastatic lymph nodes. The mean and standard deviation of displacement in LR, AP, and SI directions for the mediastinal lymph nodes of the different stations are listed in Table 1.
The displacement of the mediastinal lymph nodes within the same region were compared among the three directions and listed in Table 2. For the upper mediastinal lymph nodes, there was no statistically significant difference among the LR, AP, and SI directions (p > 0.05). For the middle mediastinal lymph nodes, the displacement in the SI direction was greater than that in the AP direction, and the difference was statistically significant (z = -2.835, p = 0.005). There was no statistically significant difference between the displacements in AP and SI or LR and SI directions (p > 0.05). For the lower mediastinal lymph nodes, there was a statistically significant difference between SI and LR, as well as SI and AP (SI vs. LR, p = 0.015; SI vs. AP, p = 0.015). There was no statistically significant difference between the LR and AP directions (z = -0.892, p = 0.373). For total mediastinal lymph nodes, there was statistically significant differences between SI and LR as well as SI and AP (SI vs. LR, p = 0.000; SI vs. AP, p = 0.008), but no statistically significant difference between the LR and AP directions (z = -1.329, p = 0.184).
The displacements of lymph nodes in the same direction among different regions are compared in Table 3. There was no statistically significant difference regarding the displacement of lymph nodes in the same direction among the three regions (p > 0.05). The 3D ranges among different regions are compared and no statistically significant difference was observed (p > 0.05).
In the SI direction, the displacement of the dome of the ipsilateral diaphragm was 9.33 ± 3.52 mm, 9.67 ± 4.65 mm, and 10.17 ± 0.98 mm in the upper, middle, and lower mediastinal lymph nodes, respectively.
The correlation between the mediastinal lymph node displacement and the dome of the ipsilateral diaphragm movement in the SI direction is illustrated in Figure 2. In the SI direction, there was no correlation between the mediastinal lymph node displacement and the dome of the ipsilateral diaphragm movement (p > 0.05).
Radiation therapy for the mediastinal lymph nodes is an important therapeutic component in the patients with locally advanced NSCLC. Accurately determining the target margin is a prerequisite for the successful treatment of NSCLC. The mediastinal lymph nodes consist of several lymph node groups along the esophagus and between the lung and the diaphragm. The mediastinal lymph nodes are close to the trachea, heart and major blood vessels, and the movement of the adjacent organs may lead to displacement of these nodes.
The temporal images acquired during conventional axial three-dimensional CT (3D-CT) scanning might not completely encompass the respiration-induced mediastinal lymph node displacement. Therefore, the CTV contoured on the 3D-CT image requires expansion with an additional margin. These margins derived from the previous published guidelines or clinical experiences are regarded as population-based. However, such population-based margins may not account for the variation of nodal motion in a specific patient. Although X-ray fluoroscopy allows for the individualized measurement of lung tumor displacement, it is not accurate for measuring lymph node displacement due to poor tissue contrast (16). A spiral CT scan at the end phases of inhalation and exhalation during active breathing control may reflect the maximum lymph node displacement. However, this cannot monitor the lymph node displacement during free breathing and the recorded lymph node displacement may be overestimated (15). 4D-CT allows for clear delineation of the anatomical structures and the lymph node displacements during free breathing, and can minimize motion artifacts. Therefore, this technique is widely used in evaluating the displacement of tumor and organ for thoracic and abdominal cancers (13, 25, 26).
The results of our study suggest that the mediastinal lymph node displacements vary widely in different directions during free breathing. We found that the mean displacements of 53 metastatic mediastinal lymph nodes in the SI, AP and LR directions were 3.28 mm, 1.87 mm, and 2.24 mm, respectively. These numbers suggest that lymph node displacement primarily occurs in the SI direction. Jenkins et al. (14) investigated 21 patients with calcified lymph nodes using orthogonal fluoroscopic imaging and found that the mean displacements of the lymph nodes in the SI, AP and LR directions were 6.6 mm, 2.6 mm, and 1.4 mm, respectively. Pantarotto et al. (24) investigated the displacements of 100 mediastinal lymph nodes using 4D-CT, and reported that the mean displacements of the lymph nodes in the SI, AP and LR directions were 5.2 mm, 3.1 mm, and 2.4 mm, respectively. Both groups reported that the lymph node SI displacement was significantly greater than the AP and LR directions (p < 0.001). These findings suggest that it might not be appropriate to use a spatial uniform margin for ITV of the mediastinal lymph nodes since the tumor motion is likely to be anisotropic. We propose that the internal margin should be greater in the SI direction than the LR and AP directions. Our present study suggests that the SI displacements are shorter than previously reported. This might be explained by the difference in the lymph node distributions. In Jenkins' study (14), the hilar lymph nodes which had larger shifts, accounted for about 50% of the lymph nodes. In Jason's study, the lower mediastinal lymph nodes which were also prone to larger movement, accounted for 25% of the lymph nodes. In our study, no hilar lymph nodes were included and the lower mediastinal lymph nodes only accounted for 17% of the lymph nodes. In addition, using the image acquisition technique, the immobilization method, and the difference in respiratory exercise may also contribute to the difference in lymph node shifts (27, 28).
The results of our study show that the displacements of lymph nodes in the LR, AP, and SI directions are similar (p > 0.05). Our results are different from Donnelly's findings (29), who investigated 44 paratracheal, subcarinal and hilar lymph nodes using 4D-CT. They found that the displacement of the hilar lymph nodes was similar to that of the mediastinal lymph nodes in the SI direction (p = 0.23). The subcarinal lymph nodes displacement was significantly greater than the paratracheal lymph nodes (p = 0.01). The displacement of the mediastinal and subcarinal lymph nodes was similar in the AP and LR directions (p > 0.05). This might be due to different classification methods. In order to avoid a geometrical target miss and unnecessary doses to the normal tissues during IFRT, the dependence of target expansion on the mediastinal lymph node station requires further study.
The lymph node movements are irregular in the LR, AP and SI directions. In our study, the 3D ranges of the upper, middle and inferior mediastinal lymph node displacements were 3.87 ± 2.45 mm, 4.97 ± 2.75 mm, and 5.23 ± 2.67 mm, respectively. A geometrical target miss induced by the mediastinal lymph node motion is considered as one of the main reasons of local failure. In the absence of patient specific information on the nodal motion during IFRT, we propose that we should expand the internal margin according to the upper values of the 95% confidence interval for the 3D range. In our data, these values are 6.47 mm, 6.64 mm, and 7.28 mm for the superior, middle and inferior mediastinal lymph nodes, respectively. The results of our study also suggest that for the mediastinal lymph nodes with 3D motion range > 5 mm, the superior, middle and inferior mediastinal lymph nodes accounted for 20.00%, 41.38% and 33.33%, respectively. This is consistent with Sher's study, which showed that a 5-mm uniform expansion would not cover an ITV of 23%, 73% and 55% of the paratracheal, subcarinal, and hilar lymph nodes, respectively. These suggest that a standard expansion of 5 mm around a gross tumor volume contoured on a helical scan for the internal margin would have missed the gross tumor, especially for the inferior mediastinal lymph nodes.
The inferior nodal displacement during inhalation is consistent with a downward pull from the contracting diaphragm, which is the primary force of inspiration. Therefore, the dome-shaped sheet of the diaphragm affects the movement of the adjacent organs (30). The mediastinal lymph nodes displacement is greatly affected by the ipsilateral diaphragm contractions, and is more obvious in the SI direction. In the previous reports, the lymph node displacement has been associated with the size of the primary tumor, lung function and gender (14, 24). However, few studies have investigated the diaphragm versus lymph node displacements. We initially speculated that the displacement of the lymph node might be related to the amplitude of the ipsilateral diaphragm movement in the SI direction. However, no predictive value was found between the range of displacement of the lymph node and the ipsilateral diaphragm. The results of our study suggest that the mediastinal lymph node displacement is not associated with the ipsilateral diaphragm displacement (p > 0.05). This might be due to complex breathing mechanisms. The mediastinal lymph node movements are not only easily affected by the contraction of the diaphragm, intercostal muscles, and heart beat, but also affected by its volume and the degree of adhesion with the adjacent tissue (14, 24, 31).
In summary, the results of our study suggest that the mediastinal lymph node displacements are different in the LR, AP and SI directions in patients with NSCLC. It might not be appropriate to use an isotropic margin in applying IFRT to NSCLC. The expansion in the SI direction should be larger than for the AP or LR directions. The displacement of the ipsilateral diaphragm is not predictive of the displacement observed in the mediastinal lymph nodes.
Figures and Tables
 | Fig. 1Four-dimensional computed tomography image of 0% phase showing contours of 2R lymph node.
These contours are drawn in all of 10 phases and mapped to current image. A. Transverse view. B. Sagittal view. C. Coronal view.
|
 | Fig. 2Correlation between mediastinal lymph node displacement and ipsilateral diaphragm dome movement in superior-inferior direction. |
References
1. Fidler MJ, Kim AW, Zusag T, Bonomi P. Treatment of locally advanced non-small cell lung cancer. Clin Adv Hematol Oncol. 2009. 7:455–464. 479–480.
2. Wagner TD, Yang GY. The role of chemotherapy and radiation in the treatment of locally advanced non-small cell lung cancer (NSCLC). Curr Drug Targets. 2010. 11:67–73.
3. Emami B, Mirkovic N, Scott C, Byhardt R, Graham MV, James Andras E, et al. The impact of regional nodal radiotherapy (dose/volume) on regional progression and survival in unresectable non-small cell lung cancer: an analysis of RTOG data. Lung Cancer. 2003. 41:207–214.
4. Liengswangwong V, Bonner JA. Point: the potential importance of elective nodal irradiation in the treatment of non-small cell lung cancer. Semin Radiat Oncol. 2000. 10:308–314.
5. Fernandes AT, Shen J, Finlay J, Mitra N, Evans T, Stevenson J, et al. Elective nodal irradiation (ENI) vs. involved field radiotherapy (IFRT) for locally advanced non-small cell lung cancer (NSCLC): a comparative analysis of toxicities and clinical outcomes. Radiother Oncol. 2010. 95:178–184.
6. Rengan R, Rosenzweig KE, Venkatraman E, Koutcher LA, Fox JL, Nayak R, et al. Improved local control with higher doses of radiation in large-volume stage III non-small-cell lung cancer. Int J Radiat Oncol Biol Phys. 2004. 60:741–747.
7. Yuan S, Sun X, Li M, Yu J, Ren R, Yu Y, et al. A randomized study of involved-field irradiation versus elective nodal irradiation in combination with concurrent chemotherapy for inoperable stage III nonsmall cell lung cancer. Am J Clin Oncol. 2007. 30:239–244.
8. Rosenzweig KE, Sura S, Jackson A, Yorke E. Involved-field radiation therapy for inoperable non small-cell lung cancer. J Clin Oncol. 2007. 25:5557–5561.
9. Nakayama H, Satoh H, Kurishima K, Ishikawa H, Tokuuye K. High-dose conformal radiotherapy for patients with stage III non-small-cell lung carcinoma. Int J Radiat Oncol Biol Phys. 2010. 78:645–650.
10. Ekberg L, Holmberg O, Wittgren L, Bjelkengren G, Landberg T. What margins should be added to the clinical target volume in radiotherapy treatment planning for lung cancer? Radiother Oncol. 1998. 48:71–77.
11. Saunders MI, Dische S, Barrett A, Parmar MK, Harvey A, Gibson D. Randomised multicentre trials of CHART vs conventional radiotherapy in head and neck and non-small-cell lung cancer: an interim report. CHART Steering Committee. Br J Cancer. 1996. 73:1455–1462.
12. Shih HA, Jiang SB, Aljarrah KM, Doppke KP, Choi NC. Internal target volume determined with expansion margins beyond composite gross tumor volume in three-dimensional conformal radiotherapy for lung cancer. Int J Radiat Oncol Biol Phys. 2004. 60:613–622.
13. Low D. 4D imaging and 4D radiation therapy: a New Era of therapy design and delivery. Front Radiat Ther Oncol. 2011. 43:99–117.
14. Jenkins P, Salmon C, Mannion C. Analysis of the movement of calcified lymph nodes during breathing. Int J Radiat Oncol Biol Phys. 2005. 61:329–334.
15. van Sörnsen de Koste JR, Lagerwaard FJ, Nijssen-Visser MR, Schuchhard-Schipper R, Joosten H, Senan S. What margins are necessary for incorporating mediastinal nodal mobility into involved-field radiotherapy for lung cancer? Int J Radiat Oncol Biol Phys. 2002. 53:1211–1215.
16. Sher DJ, Wolfgang JA, Niemierko A, Choi NC. Quantification of mediastinal and hilar lymph node movement using four-dimensional computed tomography scan: implications for radiation treatment planning. Int J Radiat Oncol Biol Phys. 2007. 69:1402–1408.
17. Keall P. 4-dimensional computed tomography imaging and treatment planning. Semin Radiat Oncol. 2004. 14:81–90.
18. Low DA, Nystrom M, Kalinin E, Parikh P, Dempsey JF, Bradley JD, et al. A method for the reconstruction of four-dimensional synchronized CT scans acquired during free breathing. Med Phys. 2003. 30:1254–1263.
19. Lu W, Parikh PJ, El Naqa IM, Nystrom MM, Hubenschmidt JP, Wahab SH, et al. Quantitation of the reconstruction quality of a four-dimensional computed tomography process for lung cancer patients. Med Phys. 2005. 32:890–901.
20. Rietzel E, Pan T, Chen GT. Four-dimensional computed tomography: image formation and clinical protocol. Med Phys. 2005. 32:874–889.
21. Keall PJ, Starkschall G, Shukla H, Forster KM, Ortiz V, Stevens CW, et al. Acquiring 4D thoracic CT scans using a multislice helical method. Phys Med Biol. 2004. 49:2053–2067.
22. Mountain CF, Dresler CM. Regional lymph node classification for lung cancer staging. Chest. 1997. 111:1718–1723.
23. Chapet O, Kong FM, Quint LE, Chang AC, Ten Haken RK, Eisbruch A, et al. CT-based definition of thoracic lymph node stations: an atlas from the University of Michigan. Int J Radiat Oncol Biol Phys. 2005. 63:170–178.
24. Pantarotto JR, Piet AH, Vincent A, van Sörnsen de Koste JR, Senan S. Motion analysis of 100 mediastinal lymph nodes: potential pitfalls in treatment planning and adaptive strategies. Int J Radiat Oncol Biol Phys. 2009. 74:1092–1099.
25. Liu HH, Balter P, Tutt T, Choi B, Zhang J, Wang C, et al. Assessing respiration-induced tumor motion and internal target volume using four-dimensional computed tomography for radiotherapy of lung cancer. Int J Radiat Oncol Biol Phys. 2007. 68:531–540.
26. Li FX, Li JB, Zhang YJ, Liu TH, Tian SY, Xu M, et al. Comparison of the planning target volume based on three-dimensional CT and four-dimensional CT images of non-small-cell lung cancer. Radiother Oncol. 2011. 99:176–180.
27. Persson GF, Nygaard DE, Af Rosenschöld PM, Richter Vogelius I, Josipovic M, Specht L, et al. Artifacts in conventional computed tomography (CT) and free breathing four-dimensional CT induce uncertainty in gross tumor volume determination. Int J Radiat Oncol Biol Phys. 2011. 80:1573–1580.
28. Sarker J, Chu A, Mui K, Wolfgang JA, Hirsch AE, Chen GT, et al. Variations in tumor size and position due to irregular breathing in 4D-CT: a simulation study. Med Phys. 2010. 37:1254–1260.
29. Donnelly ED, Parikh PJ, Lu W, Zhao T, Lechleiter K, Nystrom M, et al. Assessment of intrafraction mediastinal and hilar lymph node movement and comparison to lung tumor motion using four-dimensional CT. Int J Radiat Oncol Biol Phys. 2007. 69:580–588.
30. Weiss E, Wijesooriya K, Dill SV, Keall PJ. Tumor and normal tissue motion in the thorax during respiration: analysis of volumetric and positional variations using 4D CT. Int J Radiat Oncol Biol Phys. 2007. 67:296–307.
31. West JB. Respiratory physiology: The essentials. 2000. 6th ed. Baltimore: Lippincott Williams & Wilkins;113–127.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download