Abstract
We report here a case of pentastomiasis infection in a 3-year-old girl who had high fever, abdominal pain, abdominal tension and anemia. Ultrasound scanning of the abdomen revealed disseminated hyperechoic nodules in the liver and a small amount of ascites. Abdominal MRI showed marked hepatomegaly with disseminated miliary nodules of high signal intensity throughout the hepatic parenchyma on T2-weighted images; retroperitoneal lymphadenopathy and disseminated miliary nodules on the peritoneum were also noted. Chest CT showed scattered small hyperdense nodules on both sides of the lungs. The laparoscopy demonstrated diffuse white nodules on the liver surface and the peritoneum. After the small intestinal wall and peritoneal biopsy, histological examination revealed parenchymal tubercles containing several larvae of pentastomids and a large amount of inflammatory cell infiltration around them. The pathological diagnosis was parasitic granuloma from pentastomiasis infection.
Human pentastomiasis is a series of food-borne or water-borne parasitic zoonoses that are caused by pentastomid parasites. Human pentastomiasis is rare. To date, there have been no reports on the image characteristics of human pentastomiasis. The few published reports of this disease have all been concerned with the patients' etiology, the pathology, the pathogenesis and the clinical manifestations, as well as the diagnosis. Here we present a case of pentastomiasis infection and we summarize its imaging features and the clinical manifestations.
A 3-year-old girl was admitted to our hospital with unexplained fever and paroxysmal abdominal pain that had persisted for three days. Her body temperature at the time of admission was 39.5℃. Physical examination showed abdominal tension, though there was no obvious tenderness and rebound tenderness. The liver was palpable, and the edges were blunt and smooth. Investigations revealed the presence of severe anemia (hemoglobin 9.1 g/dL). The blood platelets count (240×103/µL) was normal, and the C-reactive protein (> 1.6 mg/dL), the white blood cell count (13.9×103/µL) and eosinophil count (4%, 0.58×103/µL) were elevated, while hepatic glutamic oxalacetic transaminase (24 U/L) and glutamate pyruvate transaminase (21 U/L) were normal. However, the level of cholinesterase (2,395 U/L) was below normal. In addition, the blood and cerebrospinal fluid cultures were sterile.
Ultrasound scanning of the abdomen and pelvis was performed on the day following admission. This revealed disseminated hyperechoic nodules in the liver (Fig. 1A), a small amount of ascites and no other remarkable findings. The abdominal non-contrast CT obtained on the fifth hospital day revealed diffusely hypodense nodular lesions in the hepatic parenchyma with marked hepatomegaly, retroperitoneal lymphadenopathy and ascites in the pelvic cavity. Eight days after admission, an abdominal MRI revealed marked hepatomegaly with diffuse high intensity of the homogeneous nodular lesions (2-4 mm diameter) throughout the hepatic parenchyma and retroperitoneal lymphadenopathy. In addition, disseminated miliary nodules of high signal intensity were located on the peritoneum on the T2-weighted images, as well as a group of diffuse, low intensity nodular lesions were seen throughout the hepatic parenchyma on the axial T1-weighted images (Fig. 1B-E). On the same day as the MRI examination, a chest CT showed multiple small hyperdense nodules (2-4 mm in diameter) in both lungs (Fig. 1F).
Both laparoscopy and a biopsy were performed fifteen days after admission. The laparoscopy demonstrated diffuse white nodules on the liver's surface and the peritoneum. A small amount of bloody ascites in the abdominal cavity was also disclosed. After the small intestinal wall and peritoneal biopsy, histological examination revealed parenchymal tubercles containing several larvae of pentastomids and a large amount of inflammatory cell infiltration surrounding them (Fig. 1G). The pathological diagnosis was parasitic granuloma from pentastomiasis infection. At this point, it was revealed by the patient's parents that the patient had consumed snake meat several months prior to her admission.
Following the diagnosis, she was administered Praziquantel (0.5 g, tid, per day) and Albendazole (0.75 g, bid, per day) in order to expel the intestinal worms. The patient's temperature began to slowly decrease two days after the start of the therapy, and after 10 days of the therapy the fever had disappeared and the abdominal pain was diminished. On examination, pentastomid worms were observed in a stool (Fig. 1H). The patient made an uneventful recovery after a half a month of therapy and she was discharged from our hospital.
Human pentastomiasis is divided into two types: the more common visceral pentastomiasis (which is the main portion of the cases of pentastomiasis) and nasopharyngeal pentastomiasis (1). Visceral pentastomiasis is an unusual parasitic zoonosis that is caused by the larval stages (nymphs) of several species of pentastomes ('tongue worms'), and these are parasites that form a unique phylum with characteristics of both arthropods and annelids (2). At present, there are ten known pathogenic species of pentastomid parasites and six known kinds of human pentastomiasis. The latter include armilliferosis, linguatulosis, porocephalosis, raillietiellosis, leiperiosis and sebekiosis (3). The species that affect humans belong to the families Linguatulidae, Armilliferidae and Porocephalidae, and all of which have different geographic distributions. More than 90% of the human cases are caused by the nymphs of only two species, L. serrata and A. armillatus (4).
The infection develops in humans when parasite ova are ingested from the respiratory secretions or feces from the final hosts (dogs and other carnivores for Linguatula, several species of large snakes for Armillifer and Porocephalus). In the digestive tract of the human host, the minute four-legged primary larvae hatch and invade the viscera of the digestive tract in the human host (5, 6). Migration in the body produced visceral larva migrans. The nymphs involve many organs, including the liver, esophagus, stomach, duodenum, jejunum, ileum, colon, appendix, rectum, mesentery, gallbladder, lung, pleura, broad ligament, pancreas, omentum, bladder, adrenal gland, heart (pericardium), lymph nodes, skin, epididymis, eye, brain, inguinal hernia sac, etc; however, the liver is the most common site (7). Visceral pentastomiasis causes the majority of asymptomatic infections in humans, but a small number of cases are fatal, and the severity of symptoms is related to the organs that are infected and the degree of infection by the nymph (8). The patients with a heavy infestation showed chronic fever, ascites, abdominal pain, diarrhea, mild anemia, hepatosplenomegaly, eosinophilia in the bone marrow and blood, and multiple polyps in the whole colon. The pathologic features of the liver biopsy are degeneration and necrosis of hepatocytes, along with obvious infiltration of eosinophils. As was demonstrated in this study, Praziquantel or mebendazole is a medical treatment for this disease (9).
Only a few cases of pentastomiasis infections have been reported in humans around the world, and there is limited literature about the imaging features of pentastomiasis infection. We report here a case where the patient had a history of eating snake meat several months prior to her hospital admission, and her main symptoms were a high fever and abdominal pain. An abdominal MRI demonstrated diffuse nodules throughout the liver, an enlarged liver, a little ascites, diffuse retroperitoneal lymphadenopathy and disseminated miliary nodules of high signal intensity on the peritoneum. The chest CT showed scattered hyperdense nodules on both lungs. The laparoscopy revealed that the liver was diffusely enlarged, as well as there were diffusely scattered micro-cyst nodules of varying sizes on the surface of the liver and the peritoneum. Microscopically, the small intestinal wall and peritoneal biopsy showed several larvae of pentastomids in the cyst nodules. The parasite worms were surrounded by a group of macrophages, inflammatory cells and fibrosis. The pathological diagnosis was parasitic granuloma from a pentastomiasis infection.
When liver enlargement and diffuse hepatic hypodense nodular lesion is seen on a T1-weighted image, it is necessary to differentiate pentastomiasis hepatic involvement from other diseases that present with diffuse liver enlargement, high fever and abdominal pain (10, 11) such as diffuse liver metastasis, diffuse hepatocellular carcinoma, lymphoma and diffuse granulomatosis (12). Biopsy or autopsy is necessary to determine the cause of the symptoms. In this report we studied the patient with magnetic resonance imaging. Due to the spatial resolution of the current T2-weighted MR imagers, the detection of these microcysts and larvae within the microcysts was well accomplished in tandem with the clinical features and epidemiology, which aided in making the differential diagnosis (13).
In summary, we have presented a rare case of pentastomiasis infection that involved many organs, including the liver, small intestine, peritoneum, lung and retroperitoneal lymph nodes. The patient's symptoms consisted of high fever, abdominal pain, abdominal tension and anemia. The imaging features of the abdominal MRI revealed marked hepatomegaly with diffuse homogeneous nodular lesions throughout the hepatic parenchyma, diffuse retroperitoneal lymphadenopathy and disseminated miliary nodules on the peritoneum. The chest CT showed scattered small nodules in both lungs. When liver enlargement and hepatic disseminated miliary nodules are identified, it is necessary to differentiate pentastomiasis hepatic involvement from other diseases. Pentastomiasis infections should be considered in the diagnoses of patients with a history of such abdominal symptoms and who have consumed snake meat, though the definite diagnosis depends on the pathology report.
Figures and Tables
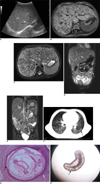
Fig. 1
3-year-old girl with pentastomiasis infection, and she had high fever, abdominal pain, abdominal tension and anemia.
A. Abdominal ultrasonography reveals disseminated miliary hyperechoic nodules in liver (arrow).
B. T1-weighted transverse MRI (TR, 1,540 ms; TE, 2.3 ms) reveals marked hepatomegaly with miliary nodular lesions (2-4 mm diameter) of low signal intensity throughout hepatic parenchyma (arrow).
C. T2-weighted transverse MRI (TR, 4,577 ms; TE, 101 ms) reveals marked hepatomegaly with disseminated nodular lesions of high signal intensity throughout hepatic parenchyma (arrow).
D, E. T2-weighted coronal MRI (TR, 3.7 ms; TE, 1.9 ms) shows disseminated miliary mesenteric lymph nodes (short arrows, D), peritoneal nodules (long arrow, D) and diffuse retroperitoneal lymphadenopathy of high signal intensity (arrow, E).
F. Chest CT shows multiple small hyperdense nodules in both lungs on lung window (arrow).
G. Cross section of pentastomid worm in peritoneum shows primordial genital tract (GT), red acidophilic gland (AG), intestine (IN) and worm wall (black arrow), which are surrounded by group of macrophages and inflammatory cells and fibrosis (white arrow) (Hematoxylin & Eosin stain, × 40).
H. Vital well-preserved pentastomid worm from patient feces. Worm is cylindrical, translucent and has prominent spiral rings (arrow). We can see its chest, abdomen and blunt round tail, which is gradually shrinking.
References
1. Zhang Q, Wang B, Huang M. Armillifer agkistrodontis disease: report of case. Zhonghua Nei Ke Za Zhi. 1996. 35:747–749. [Chinese].
2. Lavrov DV, Brown WM, Boore JL. Phylogenetic position of the Pentastomida and (pan)crustacean relationships. Proc Biol Sci. 2004. 271:537–544.
3. Qiu MH. Pentastomiasis. Tropical medicine. 2004. 2nd ed. Beijing: People's Health Press;1169–1175.
4. Liu JD, Cheng NY, Chu SY. Parasitic disease in the gastroenterological field. Jap J Gastrointest Endosc. 1993. 5:1553–1559.
5. Qiu MH, Jiang YY. Advances in studies of human pentastomiasis. Int J Med Parasit Dis. 2006. 33:281–287.
6. Tappe D, Büttner DW. Diagnosis of human visceral pentastomiasis. PLoS Negl Trop Dis. 2009. 3:E320.
7. Baird JK, Kassebaum LJ, Ludwig GK. Hepatic granuloma in a man from North America caused by a nymph of Linguatula serrata. Pathology. 1988. 20:198–199.
8. Smith JA, Oladiran B, Lagundoye SB. Pentastomiasis and malignancy. Ann Trop Med Parasitol. 1975. 69:503–512.
9. Qiu MH, Cheng ML. Armilliferiosis agkistrodontis is a visceral larva migrans echinococcosis. Chin J Parasitol Parasite Dis. 1997. 15:117–118.
10. Berenji F, Fata A, Hosseininejad Z. A case of Moniliformis moniliformis (Acanthocephala) infection in Iran. Korean J Parasitol. 2007. 45:145–148.
11. Pan CM, Tang HF, Qiu MH, Xiong QX. A case with severe infestation with Armilliferiasis moniliformis. Zhonghua Er Ke Za Zhi. 2005. 43:73–74.
12. Ihara N, Yashiro N, Kinoshita T, Yoshigi J, Ouchi T, Narita M, et al. Diffuse intrasinusoidal liver metastasis of small cell lung cancer causing fulminant hepatic failure: CT findings-a case report. Radiat Med. 2001. 19:275–277.
13. Yao MH, Wu F, Tang LF. Human pentastomiasis in China: case report and literature review. J Parasitol. 2008. 94:1295–1298.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download