Abstract
Autophagy has been studied as a therapeutic strategy for cardiovascular diseases. However, insufficient studies have been reported concerning the influence of vascular smooth muscle cells (VSMCs) through autophagy regulation. The aim of the present study was to determine the effects of VSMCs on the regulation of autophagy under in vitro conditions similar to vascular status of the equipped microtubule target agent-eluting stent and increased release of platelet-derived growth factor-BB (PDGF-BB). Cell viability and proliferation were measured using MTT and cell counting assays. Immunofluorescence using an anti-α-tubulin antibody was performed to determine microtubule dynamic formation. Cell apoptosis was measured by cleavage of caspase-3 using western blot analysis, and by nuclear fragmentation using a fluorescence assay. Autophagy activity was assessed by microtubule-associated protein light chain 3-II (LC-II) using western blot analysis. Levels of intracellular reactive oxygen species (ROS) were measured using H2DCFDA. The proliferation and viability of VSMCs were inhibited by microtubule regulation. Additionally, microtubule-regulated and PDGF-BB-stimulated VSMCs increased the cleavage of caspase-3 more than only the microtubule-regulated condition, similar to that of LC3-II, implying autophagy. Inhibitory autophagy of microtubule-regulated and PDGF-BB-stimulated VSMCs resulted in low viability. However, enhancement of autophagy maintained survival through the reduction of ROS. These results suggest that the apoptosis of conditioned VSMCs is decreased by the blocking generation of ROS via the promotion of autophagy, and proliferation is also inhibited. Thus, promoting autophagy as a therapeutic target for vascular restenosis and atherosclerosis may be a good strategy.
Vascular smooth muscle cells (VSMCs) are one of the cell types constituting the arterial wall, and their proliferation plays an important role in the progression of early atherosclerosis and vascular restenosis [1]. Additionally, the viability of VSMCs is known to be an important factor in the rupture of arteriosclerosis [2]. The atherosclerotic plaque rupture occurs myocardial infarction and stroke through thrombus formation [3]. In this process, various cytokines [4] and growth factors [56] induce vascular inflammation and remodeling. Platelet-derived growth factor-BB (PDGF-BB) is a growth factor that is increased in the blood vessels of cardiovascular disease [7]. It is also known to be a powerful stimulation factor of proliferation or migration of VSMCs [8]. Most treatments for cardiovascular diseases, such as vascular restenosis and arteriosclerosis, are performed through surgical operations. The stent is coated with a microtubule-targeted drug such as paclitaxel for the prevention of vascular restenosis [9]. After the surgery, thrombosis and approximately 12% angiographic restenosis occur [10]. Michigan Cancer Foundation-7 (MCF-7), one of the breast cancer cell lines, is known to be resistant to apoptotic cell death by paclitaxel through increased stimulation of the autophagic pathway [11]. Therefore, it is expected that angiographic restenosis after angioplastic surgery with a microtubule target agent-eluting stent may be associated with autophagy. However, insufficient studies have reported the effects of microtubules and autophagy in VSMCs.
Autophagy contributes to the maintenance of intracellular homeostasis through recycling or degradation of intracellular elements [1213]. The targeted proteins of autophagy are surrounded by double-membraned vesicles called autophagosomes [14]. The autophagosome fuses with lysosomes, and then forms autolysosome in which intracellularly damaged organs and proteins are decomposed [15]. In vascular cells, autophagy acts as a regulator of cell viability and function [1617]. It also prevents the abnormal function of blood vessels, such as inflammation, by protecting against potential damage by various stresses [18]. Autophagy in VSMCs is stimulated or inhibited by various stimuli and stress factors (metabolic stresses, reactive species, cytokines, and drugs) [19]. It has been reported that autophagy in VSMCs promotes senescence, neointima formation, and atherogenesis [20]. However, it has also been reported that the promotion of autophagy inhibits neointima formation induced by the balloon injury model [21]. Together, these findings indicate that differences in the autophagy levels of VSMCs may be completely different phenomena (regulation of VSMC homeostasis). In the treatment strategy of cardiovascular disease by VSMC regulation through inhibition or promotion of autophagy, there remains a lack of evidence to determine which direction is the right strategy. The results of the present study suggest that the strategy of increasing autophagy is valid for the treatment of atherosclerosis and restenosis.
Cell culture materials were purchased from Invitrogen (Carlsbad, CA, USA). PDGF-BB was obtained from PeproTech (Rocky Hill, NJ, USA). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), bafilomycin A1, rapamycin, and 3-methyladenine (3-MA) were purchased from Millipore Corporation (Billerica, MA, USA). N-Acetyl-L-cysteine (NAC), 2′,7′-dichlorofluorescin diacetate (H2DCFDA), paclitaxel, and vinorelbine were purchased from Sigma-Aldrich (Saint Louis, MO, USA). Anti-β-actin, anti-microtubule-associated protein light chain 3 (LC3) A/B, anti-α-tubulin, and anti-caspase-3 antibodies were purchased from Cell Signaling Technology, Inc. (Beverly, MA, USA).
Rat aortic VSMCs were obtained from Cell Applications Inc. (San Diego, CA, USA). Rat aortic VSMCs were cultured in DMEM media containing 10% (v/v) fetal bovine serum (FBS), 100 IU/ml penicillin, 100 µg/ml streptomycin, 4.5 g/L D-glucose, 110 mg/L sodium pyruvate, and 2 mM L-glutamine at 37℃ in a humidified atmosphere of 95% air and 5% CO2. VSMCs were used at passages 4-9. Before all assays, VSMCs were maintained under serum-starved conditions.
To evaluate the effects of paclitaxel and vinorelbine on cell proliferation, the proliferation and viability levels of PDGF-BB-stimulated VSMCs were determined using the MTT assay [22] and cell counting assay [23], respectively, as described previously. Briefly, for MTT assay, VSMCs were seeded in 96-well plates at 3.5×104 cells/ml and were cultured in DMEM containing 10% FBS at 37℃ and 5% CO2 for 24 h. After incubation in serum-free DMEM for 24 h, the cells were pretreated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA, 0.2 µM rapamycin, or 5 mM NAC for 24 h, followed by treatment with or without 25 ng/ml PDGF-BB for another 24–72 h. At the end of the reaction, the medium was removed, and 200 µl of MTT solution was added to each well for 4 h. After the MTT solution was replaced with 200 µl of dimethyl sulfoxide (DMSO), the absorbance was measured at 565 nm using a microplate reader (Infinite M200 PRO; Tecan Group Ltd., Zürich, Switzerland). For cell counting assay, VSMCs were seeded into 12-well plates at a density of 1.6×105 cells per well and were incubated for 24 h. After incubation in serum-free DMEM for 24 h, the VSMCs were pretreated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA, 0.2 µM rapamycin, or 5 mM NAC for 24 h. Next, the VSMCs were further cultured in medium with or without 25 ng/ml PDGF-BB for 48 h, and the VSMCs were counted using a hemocytometer.
ROS production was determined as described previously [2224]. H2DCFDA was used to evaluate the intracellular ROS level. VSMCs (1×105 cells/ml) were pretreated with or without 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA, and 0.2 µM rapamycin for 24 h, followed by the addition of 25 ng/ml PDGF-BB for 48 h. The cells were stained for 30 min at 37℃ by 20 µM H2D-CFDA. The intracellular ROS level was determined by measuring the fluorescence intensity using a microplate reader (Infinite F200 PRO, Tecan Group Ltd., Männedorf, Switzerland).
Western blotting was performed as described previously [22]. Total cell lysates were prepared using RIPA lysis buffer (150 mM sodium chloride, 1% Triton X-100, 1% sodium deoxycholate, 0.1% sodium dodecyl sulfate [SDS], 50 mM Tris-HCl, pH 7.5, and 2 mM EDTA) containing proteinase inhibitors. After 10 min incubation on ice, cell lysates were centrifuged at 13,000 rpm for 10 min, and the supernatants were collected. The protein concentrations were determined using the bicinchoninic acid protein assay kit (Pierce, Rockford, IL, USA). The proteins were subjected to SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and were transferred to a polyvinylidene fluoride membrane (ATTO Corporation, Tokyo, Japan). After blocking, the membranes were incubated with primary antibodies to determine the levels of LC3A/B (1:1000) and caspase-3 (1:1000). The membrane was washed and incubated with a corresponding horseradish peroxidaseconjugated secondary antibody, and the signals were detected using an ECL western blotting detection system. The activated proteins were normalized against β-actin levels. Band intensities were quantified using Quantity One software (Bio-Rad, Hercules, CA, USA).
Immunofluorescence staining was performed as described previously [22]. VSMCs were seeded on cover slips and were incubated for 24 h, followed by starvation in serum-free medium for 24 h. The serum-starved cells were pretreated with 1 µM paclitaxel or 0.2 µM vinorelbine for another 24 h. Following stimulation, the cells were treated with 25 ng/ml PDGF-BB for 48 h, and then were washed twice in cold phosphate-buffered saline (PBS). The cells were fixed with cold 4% formaldehyde for 10 min and were permeabilized with chilled 0.25% Triton X-100 for 2 min. Next, a blocking step was performed using 3% bovine serum albumin (BSA) in PBS for 1 h at room temperature. The cells were incubated with primary anti-α-tubulin antibody for 1 h, followed by incubation with FITC-labeled secondary antibody for 1 h. Cell nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI), and immunofluorescence images were obtained using a confocal laser scanning microscope (LSM5 LIVE; Zeiss, Oberkochen, Germany).
All data are expressed as means±standard error of the mean (SEM) of three independent experiments. Analysis of variance (ANOVA) was used to compare parameters among multiple groups (GraphPad, San Diego, CA, USA). If a significant difference was observed between the treated groups, Dunnett's test was applied. Differences with p<0.05 were deemed to indicate statistical significance.
We carried out the MTT assay to test the effects of paclitaxel and vinorelbine on VSMC proliferation and viability via microtubule regulation (Figs. 1A and B). We also observed microtubules of VSMCs using immunofluorescence analysis (Fig. 1C). After binding to microtubules, paclitaxel enhances tubulin polymerization, resulting in stabilizing the microtubules and subsequently arresting metaphase of cell cycle [25]. Vinorelbine binds to free-tubulin dimers, and causes aggregation, resulting in the prevention of tubulin polymerization [26]. The concentration of these drugs used in this study were determined by performing preliminary tests for selecting doses at maximal effects without cytotoxicity (see Supplementary Fig. 1 of Supporting Information). Serum-starved VSMCs were pretreated with 1 µM paclitaxel, 0.2 µM vinorelbine, or 0.1% DMSO as a control for 24 h, and then the VSMCs were treated with or without 25 ng/ml PDGF-BB for 24–72 h. As shown in Fig. 1A, the proliferation of PDGF-BBstimulated VSMCs was inhibited significantly by paclitaxel and vinorelbine in a time-dependent manner.
Vinorelbine inhibited the proliferation of PDGF-BB-stimulated VSMCs by almost 100% at 2 days, and paclitaxel blocked the proliferation by almost 100% at 3 days. We also evaluated the cytotoxic effects of paclitaxel and vinorelbine. The viability of VSMCs was significantly decreased by vinorelbine at 2 days, and by paclitaxel and vinorelbine at 3 days. Paclitaxel led to inhibition of cell proliferation and induction of apoptosis via the stabilization of microtubules [27], and vinorelbine has demonstrated anti-proliferation and induction of apoptosis as a microtubuledestabilizing agent [28]. We confirmed the microtubule regulation of VSMCs by paclitaxel and vinorelbine through immunofluorescence staining of α-tubulin (component of microtubule). Fig. 1C shows that the microtubules were more likely to have a line shape in paclitaxel-treated VSMCs than in untreated VSMCs. However, vinorelbine-treated VSMCs were not observed to have a line shape; instead, microtubules were observed as dots. These results indicate that the proliferation of PDGF-BB-stimulated VSMC is inhibited by paclitaxel and vinorelbine via microtubule regulation. Additionally, VSMC viability was decreased by paclitaxel and vinorelbine.
To examine the mechanism responsible for the apoptotic effects of paclitaxel and vinorelbine via microtubule regulation, we measured the cleavage of caspase-3 (from 35 to 17–19 kDa) as an apoptosis marker [22] and nuclear fragmentation [29], using western blotting and fluorescent staining of nuclei, respectively (Fig. 2). Serum-starved VSMCs were incubated with 1 µM paclitaxel, 0.2 µM vinorelbine, or 0.1% DMSO as a control for 24 h, and the VSMCs were incubated with or without 25 ng/ml PDGF-BB for 0–72 h. Fig. 2A shows that the PDGF-BB-stimulated VSMCs increased caspase-3 cleavage by microtubule regulation in a timedependent manner. The cleavage of caspase-3 through microtubule regulation in PDGF-BB-stimulated VSMCs was larger than that in unstimulated VSMCs. Next, we confirmed nuclear fragmentation, which is a feature of apoptosis [30]. Fig. 2B shows that the PDGF-BB-stimulated VSMCs demonstrated nuclear fragmentation by microtubule regulation. These results indicate that the microtubule-regulated VSMCs increasingly induce apoptosis by PDGF-BB stimulation through the blockade of microtubule dynamic formation.
To investigate the effects of microtubule regulation on VSMC autophagy, we measured the autophagy activity of PDGF-BBstimulated VSMCs via microtubule regulation. 3-MA inhibits autophagy by the inhibition of type III phosphatidylinositol 3-kinases [31]. Activation of mechanistic target of rapamycin (mTOR) leads to suppression of autophagy via phosphorylation of multiple autophagy-related proteins such as ULK1 (UNC-5 like autophagy activating kinase 1), ATG13 (autophagy related gene 13), AMBRA1 (autophagy/beclin 1 regulator 1), and ATG14L (baclin 1-associated autophagy-related key regulator) [32]. Rapamycin promotes autophagy through inhibition of mTOR [33]. The concentrations of 3-MA and rapamycin in VSMC studies are widely used at 5 mM [34] and 0.2 µM [35], respectively. Thus, after confirming the effect of 5 mM 3-MA and 0.2 µM rapamycin on LC3-II levels (see Supplementary Fig. 2 in Supporting Information), these concentrations of 3-MA and rapamycin were used in this study.
The autophagy activity was evaluated by the conversion levels of LC3-I to -II [36] using western blotting (Fig. 3). Quiescent VSMCs were incubated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA, 0.2 µM rapamycin, or 0.1% DMSO as a control for 24 h, and VSMCs were incubated with or without 25 ng/ml PDGF-BB for 0–48 h, and 0.1 µM bafilomycin A1 was treated 4 h before the end of the reaction. The PDGF-BB-stimulated or microtubule-regulated VSMCs increased autophagy activity in a time-dependent manner (Fig. 3A). Bafilomycin A1 is known to induce autophagosome accumulation through the inhibition of autophagosome and lysosome fusion [37]. Thus, bafilomycin A1 can be used to accurately measure LC3-II levels on the membrane of autophagosomes [36]. Figs. 3B and C show that VSMC autophagy activity increased microtubule regulation with PDGF-BB compared with that in unregulated microtubules or unstimulated VSMCs. The results were similar to those with the cleavage of caspase-3 shown in Fig. 2. We also confirmed the effects of viability via autophagy regulation in PDGF-BB-stimulated VSMCs. Fig. 3D shows that VSMCs inhibited autophagy by 3-MA (autophagy inhibitor), which increased caspase-3 cleavage. However, VSMCs promoted autophagy by rapamycin (autophagy stimulator), which decreased caspase-3 cleavage. These results suggest that autophagy plays an important role as a defense mechanism in inducing apoptosis by microtubule regulation of VSMCs.
To further understand the role of autophagy in VSMC proliferation and viability, we investigated the regulatory effects of autophagy using 5 mM 3-MA and 0.2 µM rapamycin in microtubule-regulated VSMCs. The proliferation and viability of VSMCs were determined using the cell counting and MTT assays. Serum-starved VSMCs were cultured using 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA, 0.2 µM rapamycin, or 0.1% DMSO as a control for 24 h, and the VSMCs were incubated with or without 25 ng/ml PDGF-BB for 48 h. Fig. 4A shows that the cell number of VSMCs was significantly more decreased by the regulation of microtubules and inhibition of autophagy than by only microtubule-regulated VSMCs. Moreover, activated VSMCs exhibited a decreased cell number via the regulation of microtubules and inhibition of autophagy compared with that of conditioned VSMCs not treated with PDGF-BB. As shown in Fig. 4B, the results of the inhibition of autophagy (Fig. 4A) and via the promotion of autophagy demonstrated the opposite pattern. Autophagy-promoted VSMCs significantly increased the viability and inhibited proliferation by rapamycin.
We further confirmed the role of autophagy using the MTT assay. The MTT assay is a method of evaluating cell proliferation and survival through mitochondrial activity [38]. Figs. 4C and D show that the VSMCs exhibited decreased proliferation and viability through the regulation of microtubules and inhibition of autophagy. However, the promotion of autophagy increased viability and inhibited proliferation in PDGF-BB-stimulated VSMCs. Taken together, microtubule regulation of PDGF-BB-stimulated VSMCs decreased cell viability via mitochondrial damage. Thus, activation of autophagy is a defense mechanism against the decrease in cell viability by microtubule regulation. Additionally, the proliferation was inhibited by almost 100% in conditioned VSMCs through autophagy promoting and microtubule regulation.
Generation of ROS induces apoptosis by the damage of VSMCs through oxidative stress [39]. To investigate whether intracellular ROS generation is involved in the regulation of microtubules and autophagy in PDGF-BB-stimulated VSMCs, intracellular ROS levels were measured using the H2DCFDA assay (Fig. 5). Quiescent VSMCs were pretreated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA, 0.2 µM rapamycin, or 0.1% DMSO as a control, followed by incubation for 24 h, and then the VSMCs were incubated with or without 25 ng/ml PDGF-BB for 48 h. Fig. 5A shows that the VSMCs exhibited increased intracellular ROS generation by microtubule regulation. The ROS levels revealed significantly increased regulation of microtubule-regulated and autophagy-inhibited VSMCs compared with that in only microtubule-regulated VSMCs, whereas the ROS levels of autophagystimulated VSMCs were significantly decreased compared with that in only microtubule-regulated VSMCs. Intracellular ROS were increasingly generated in PDGF-BB-stimulated and microtubule-regulated VSMCs (Fig. 5B). The PDGF-BB-stimulated VSMCs also demonstrated changes in the ROS level with the same pattern as shown in Fig. 5A through regulation of autophagy (Fig. 5B). These results indicate that the microtubule-regulated and PDGF-BB-stimulated VSMCs exhibited increased ROS generation, and the promotion of autophagy decreased intracellular ROS generation.
To determine the effects of intracellular ROS regulation on the regulation of microtubules and autophagy in PDGF-BB-stimulated VSMCs, the ROS level, proliferation, and viability were measured using H2DCFDA, cell counting, and MTT assays, respectively. Serum-starved VSMCs were cultured with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA, 0.2 µM rapamycin, 5 mM NAC, or 0.1% DMSO as a control for 24 h, and VSMCs were incubated with or without 25 ng/ml PDGF-BB for 48 h. As shown in Fig. 6A, 5 mM NAC had no toxic effects on VSMCs after 48–72 h. The microtubule- and autophagy-regulated and PDGF-BB-stimulated VSMCs inhibited intracellular ROS generation by 5 mM NAC (Fig. 6B). Fig. 6C shows that the microtubule-regulated VSMCs inhibited autophagy, causing a decreased cell number. However, the decrease in cell number was significantly alleviated in NAC-treated VSMCs. The reduction in the cell number of microtubule-regulated and PDGF-BB-stimulated VSMCs was also significantly alleviated in the NAC-treated groups (Fig. 6D). The cell numbers of microtubule-regulated and autophagy-stimulated VSMCs were similar to those of the NAC-treated groups (Figs. 6E and F). Together, these results suggest that the reduction of ROS by activating autophagy can enhance the VSMC viability.
The present study yielded three major findings: (1) the potent cytotoxic effect is induced in PDGF-BB-stimulated VSMCs through excessive generation of intracellular ROS by blocking microtubule dynamic formation; (2) apoptosis is increased more in autophagy-inhibited and conditioned VSMCs than in only microtubule-regulated VSMCs; and (3) the promotion of autophagy decreases apoptosis via the reduction of ROS levels. These findings are illustrated in Fig. 7.
The anti-cancer drugs paclitaxel and vinorelbine stabilize and destabilize microtubules, respectively, inducing the inhibition of cell proliferation and migration and induction of apoptosis [4041]. Paclitaxel and vinorelbine decreased the proliferation and viability levels of VSMCs in a time-dependent manner (Figs. 1A and B). It was also confirmed that the drugs inhibit the dynamic formation of microtubules (Fig. 1C). Therefore, we confirmed that paclitaxel and vinorelbine inhibit the proliferation of VSMCs by microtubule regulation, supporting the results of previous studies [40]. Caspase-3 is a major marker of apoptosis and promotes the apoptosis pathway [42]. We observed that microtubule dynamic formation in VSMCs was suppressed using paclitaxel and vinorelbine, and stimulation with PDGF-BB induced apoptosis via the cleavage of caspase-3 (Fig. 2). PDGF-BB-stimulated VSMCs activate microtubule dynamic formation, as well as promote proliferation and migration [43]. Suppression of microtubule dynamic formation may induce the apoptosis pathway of activated VSMCs.
Because the autophagy activity of PDGF-BB-stimulated VSMCs is increased and plays an important role in resistance to oxidative stress [36], we further evaluated the effects of microtubule regulation on PDGF-BB-stimulated VSMC autophagy, as well as the apoptotic effects of autophagy regulation on VSMCs. The autophagy levels of microtubule-regulated VSMCs were increased in a time-dependent manner. The autophagy levels of microtubule-regulated and PDGF-BB-stimulated VSMCs were further increased (Figs. 3A, B and C). Additionally, inhibition of autophagy using 3-MA increased apoptosis, and activation of autophagy using rapamycin maintained the viability (Fig. 3D). These results indicate that the control of apoptosis by autophagy regulation in VSMCs is possible.
Apoptosis was induced by the regulation of microtubules and stimulation of PDGF-BB (Fig. 2), and autophagy activity was also increased in the same conditioned VSMCs (Fig. 3A). Previous studies have reported that the increased activity of autophagy inhibits the apoptosis pathway [4445]. Beclin 1 plays an important role in autophagy. Beclin 1 forms core complexes (beclin 1-vps34-vps15), that induce the autophagy pathway, and it also plays an anti-apoptotic role under some conditions, such as chemotherapy, irradiation, immunotherapy, nutrient deprivation, and angiogenesis [46]. Therefore, it may be considered that the autophagy pathway is activated as a defense mechanism of conditioned VSMCs in apoptosis.
We further confirmed the effects of autophagy on the proliferation and viability of conditioned VSMCs. 3-MA significantly reduced the number of VSMCs through autophagy inhibition. The proliferation of VSMCs was inhibited by autophagy stimulation using rapamycin, but the viability was maintained (Figs. 4A and B). Since MTT assays have revealed cell viability and proliferation in response to mitochondrial activity [38], the low values of the MTT assay indicate low levels of mitochondria activity and mitochondrial dysfunction. The results shown in Fig. 4C and D revealed the same patterns as those shown in Figs. 4A and B. These results suggest that apoptosis is induced by mitochondrial damage in microtubule-regulated and PDGF-BB-stimulated VSMCs, and that mitochondrial damage is decreased by promoting autophagy.
Oxidative stress due to excessive ROS levels damages cellular organelles and induces apoptosis [4748]. PDGF-BB-stimulated VSMCs exhibited increased ROS, which acts as a signaling pathway molecule [49]. Antioxidants reduce PDGF β-receptor phosphorylation through the inhibitory oxidation of protein tyrosine phosphatases [50]. The ROS levels of microtubule-regulated VSMCs were substantially increased (Fig. 5A). The ROS levels of microtubule-regulated and PDGF-BB-stimulated VSMCs were higher than those of only microtubule-regulated VSMCs (Fig. 5B). The increase in autophagy activity affects ROS reduction [51]. Promoting the autophagy of VSMCs decreased the ROS levels (Fig. 5). Thus, these results suggest that the promotion of autophagy in the conditioned VSMCs decreases the ROS levels and prevents cell damage. We also confirmed the relationship between ROS and autophagy. ROS levels were reduced using NAC to suppress oxidative stress. Conditioned VSMCs were incubated with NAC, improving the viability of the VSMCs (Figs. 6C and D). Additionally, the anti-apoptotic effects of the autophagy stimulator and effects of NAC on VSMC viability were similar (Figs. 6E and F). These results suggest that the inhibition of oxidative stress through ROS reduction plays an important role in the viability of microtubule-regulated and PDGF-BB-stimulated VSMCs, and the promotion of autophagy is thought to maintain the viability of cells through ROS reduction.
In conclusion, our results indicate that the increased levels of autophagy in the microtubule-regulated and PDGF-BB-stimulated VSMCs reduce the levels of intracellular ROS and suppress mitochondrial damage. Therefore, we suggest that regulation of the autophagy level in VSMCs as a tool for the treatment of diseases, such as arteriosclerosis and vascular restenosis, is a good strategy to promote autophagy.
Figures and Tables
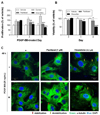 | Fig. 1Effects of paclitaxel and vinorelbine on VSMC proliferation, viability and microtubule regulation.Serum-starved VSMCs were incubated with 1 µM paclitaxel or 0.2 µM vinorelbine for 24 h followed by 25 ng/ml PDGF-BB-treatment for 24–2 h. VSMC proliferation and viability were evaluated using the MTT assay (A, B). Mean values of the vehicle group (0.1% DMSO) were set to 100%. Data are expressed as means±SEM (n = 3). *p<0.05, **p<0.01, ***p<0.001 vs. control (PDGF-BB alone) or vehicle. (C) Microtubules were observed by confocal microscopy. After cells were fixed with 4% formaldehyde and membrane-permeabilized using 0.25% Triton X-100, immunofluorescence staining was performed using anti-α-tubulin and anti-FITC antibodies. Nuclei were stained with DAPI. Red arrow indicates stabilization of microtubule and yellow arrow indicates destabilization of microtubule. Immunofluorescence images are representative of those obtained from three independent experiments (scale bar: 20 µm; nuclei: blue; α-tubulin: green).
|
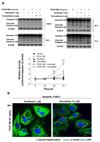 | Fig. 2Effects of microtubule regulation on caspase-3 in PDGF-BB-stimulated VSMCs.Serum-starved VSMCs were incubated with 1 µM paclitaxel or 0.2 µM vinorelbine for 24 h followed by 25 ng/ml PDGF-BB treatment for 24–72 h. (A) VSMC apoptosis was evaluated according to the levels of caspase-3 cleavage (an apoptosis marker). The levels of full-length caspase 3 (35 kDa) and the cleaved fragment (17–19 kDa) were assessed by western blotting. The band densities were normalized to those of β-actin. The gel images shown are representative of those obtained from three independent experiments. The relative density was plotted by line graph. Mean values of the vehicle group (0.1% DMSO) were set to 1 fold. Data are expressed as means±SEM. *p<0.05, **p<0.01 vs. vehicle. (B) Immunofluorescence staining was performed using anti-α-tubulin and anti-FITC antibodies, and the VSMC nuclei were stained with DAPI (arrow: nuclear fragmentation; scale bar: 20 µm; nuclei: blue; α-tubulin: green).
|
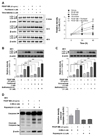 | Fig. 3Effects of autophagy via microtubule regulation and effects of caspase-3 cleavage via autophagy regulation in PDGF-BB-stimulated VSMCs.Serum-starved VSMCs were incubated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA (an autophagy inhibitor), or 0.2 µM rapamycin (an autophagy stimulator) for 24 h followed by 25 ng/ml PDGF-BB treatment for 0–48 h. Additionally, the cells were treated with 0.1 µM bafilomycin A1 for 4 h before the end of the reaction. Cell lysates applied to SDS-PAGE and subsequently immunoblotted. The band densities were normalized to those of β-actin. The gel images shown are representative of those obtained from three or five independent experiments. Data shown in graphs for relative density are expressed as means±SEM. (A) The conversion of LC3-I to LC3-II was measured as an autophagy key marker in microtubule-regulated VSMCs before and after (24–48 h) PDGF-BB treatment. Mean values of the vehicle group (0.1% DMSO) were set to 1 fold. **p<0.01 vs. vehicle. (B, C) Measurement of autophagic flux. Bafilomycin A1 (0.1 µM) was added to the vehicle (0.1% DMSO)-treated, microtubule-regulated (paclitaxel or vinorelbine), or PDGF-BB-stimulated VSMCs 4 h prior to harvest, and accumulation of LC3-II was measured by western blotting. The mean value of vehicle was set to 1 fold. *p<0.05, **p<0.01 vs. the indicated group. (D) The changes of apoptosis by autophagy regulation were tested by the measurement of caspase-3 cleavage levels. Mean values of the only rapamycin-treated group were set to 1 fold.
|
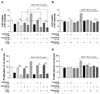 | Fig. 4Effects of the regulation of microtubules and autophagy on the proliferation and viability in PDGF-BB-stimulated VSMCs.Serum-starved VSMCs were incubated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA (autophagy inhibitor), or 0.2 µM rapamycin (autophagy stimulator) for 24 h followed by 25 ng/ml PDGF-BB treatment for 48 h. Microtubule-regulated and PDGF-BB-stimulated VSMCs viability and proliferation by autophagy regulation were determined using the cell counting assay (A, B) and MTT assay (C, D). Data are expressed as means±SEM (n=3). *p<0.05, **p<0.01 vs. the indicated group.
|
 | Fig. 5Intracellular ROS levels through microtubule regulation and autophagy in PDGF-BB-stimulated VSMCs.Serum-deprived VSMCs were incubated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA (autophagy inhibitor), or 0.2 µM rapamycin (autophagy stimulator) for 24 h followed by 25 ng/ml PDGF-BB treatment for 48 h. After stimulation, the cells were stained with 20 µM H2DCFDA for 30 min at 37℃, and the fluorescence intensities were measured. (A) ROS levels of microtubule- and autophagy-regulated VSMCs. (B) ROS levels of microtubule- and autophagy-regulated, and PDGF-BB-stimulated VSMCs. Mean values of the vehicle group (0.1% DMSO) were set to 100%. Data are expressed as means±SEM (n=3). *p<0.05, **p<0.01 vs. the indicated group.
|
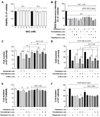 | Fig. 6Effects of the regulation of intracellular ROS, microtubule and autophagy on proliferation and viability in PDGF-BB-stimulated VSMCs.Serum-deprived VSMCs were incubated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA (autophagy inhibitor), 0.2 µM rapamycin (autophagy stimulator), or 5 mM NAC (ROS scavenger) for 24 h followed by 25 ng/ml PDGF-BB treatment for 48 h. (A) Effects of NAC on cell viability. VSMCs cultured in serum-free medium were incubated with 1–5 mM NAC for the indicated periods, followed by subjecting the cells to the MTT assay. (B) Effects of NAC (5 mM) on ROS reduction under the regulation of microtubule and autophagy. The conditioned VSMCs were cultured with 5 mM NAC, and the ROS levels were measured using the H2DCFDA assay. Mean values of the vehicle group (0.1% DMSO) were set to 100%. ***p<0.001 vs. vehicle. The proliferation and viability of microtubule-regulated, autophagy-inhibited (C, D), and autophagy-stimulated (E, F), with or without PDGF-BB-treated VSMCs under the regulation of ROS were determined using the cell counting assay. Data are expressed as means±SEM (n=3). *p<0.05, **p<0.01 vs. the indicated group.
|
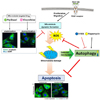 | Fig. 7Proposed scheme for the antiapoptotic effect of autophagy via ROS regulation in microtubule-regulated and PDGF-BB-stimulated VSMCs.PDGF receptor activation induces microtubule dynamic formation in VSMCs for cell proliferation and migration, which can be inhibited by microtubuletargeted drugs such as paclitaxel and vinorelbine, resulting in excessive production of ROS. This causes mitochondria damage, followed by apoptosis. Rapamycin as an autophagy activator can prevent excessive ROS production and apoptosis, resulting in cell viable. Thus, autophagy can act as a defense mechanism in microtubule-regulated VSMCs, and a regulator of apoptosis of conditioned VSMCs.
|
ACKNOWLEDGEMENTS
This research was supported financially by the research fund of Chungnam National University (2016).
Notes
References
1. Chistiakov DA, Orekhov AN, Bobryshev YV. Vascular smooth muscle cell in atherosclerosis. Acta Physiol (Oxf). 2015; 214:33–50.

2. Shioi A, Ikari Y. Plaque calcification during atherosclerosis progression and regression. J Atheroscler Thromb. 2018; 25:294–303.

3. van der Wal AC, Becker AE. Atherosclerotic plaque rupture— pathologic basis of plaque stability and instability. Cardiovasc Res. 1999; 41:334–344.
4. Ramji DP, Davies TS. Cytokines in atherosclerosis: key players in all stages of disease and promising therapeutic targets. Cytokine Growth Factor Rev. 2015; 26:673–685.

5. Ross R. Growth factors in the pathogenesis of atherosclerosis. Acta Med Scand Suppl. 1987; 715:33–38.

6. Raines EW, Ross R. Multiple growth factors are associated with lesions of atherosclerosis: specificity or redundancy? Bioessays. 1996; 18:271–282.

7. Cagnin S, Biscuola M, Patuzzo C, Trabetti E, Pasquali A, Laveder P, Faggian G, Iafrancesco M, Mazzucco A, Pignatti PF, Lanfranchi G. Reconstruction and functional analysis of altered molecular pathways in human atherosclerotic arteries. BMC Genomics. 2009; 10:13.

8. Andrae J, Gallini R, Betsholtz C. Role of platelet-derived growth factors in physiology and medicine. Genes Dev. 2008; 22:1276–1312.

9. Gershlick A, De Scheerder I, Chevalier B, Stephens-Lloyd A, Camenzind E, Vrints C, Reifart N, Missault L, Goy JJ, Brinker JA, Raizner AE, Urban P, Heldman AW. Inhibition of restenosis with a paclitaxel-eluting, polymer-free coronary stent: the European evaLUation of pacliTaxel Eluting Stent (ELUTES) trial. Circulation. 2004; 109:487–493.
10. Alfonso F, Byrne RA, Rivero F, Kastrati A. Current treatment of instent restenosis. J Am Coll Cardiol. 2014; 63:2659–2673.

11. Sifringer M, Bendix I, Börner C, Endesfelder S, von Haefen C, Kalb A, Holifanjaniaina S, Prager S, Schlager GW, Keller M, Jacotot E, Felderhoff-Mueser U. Prevention of neonatal oxygen-induced brain damage by reduction of intrinsic apoptosis. Cell Death Dis. 2012; 3:e250.

12. Kobayashi S. Choose delicately and reuse adequately: the newly revealed process of autophagy. Biol Pharm Bull. 2015; 38:1098–1103.

13. Levine B, Mizushima N, Virgin HW. Autophagy in immunity and inflammation. Nature. 2011; 469:323–335.

14. Mizushima N, Yoshimori T, Ohsumi Y. The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol. 2011; 27:107–132.

15. Burman C, Ktistakis NT. Autophagosome formation in mammalian cells. Semin Immunopathol. 2010; 32:397–413.

16. Nussenzweig SC, Verma S, Finkel T. The role of autophagy in vascular biology. Circ Res. 2015; 116:480–488.

17. De Meyer GR, Grootaert MO, Michiels CF, Kurdi A, Schrijvers DM, Martinet W. Autophagy in vascular disease. Circ Res. 2015; 116:468–479.

19. Salabei JK, Hill BG. Autophagic regulation of smooth muscle cell biology. Redox Biol. 2015; 4:97–103.

20. Grootaert MO, da Costa Martins PA, Bitsch N, Pintelon I, De Meyer GR, Martinet W, Schrijvers DM. Defective autophagy in vascular smooth muscle cells accelerates senescence and promotes neointima formation and atherogenesis. Autophagy. 2015; 11:2014–2032.

21. Cho YS, Yen CN, Shim JS, Kang DH, Kang SW, Liu JO, Kwon HJ. Antidepressant indatraline induces autophagy and inhibits restenosis via suppression of mTOR/S6 kinase signaling pathway. Sci Rep. 2016; 6:34655.

22. Park HS, Quan KT, Han JH, Jung SH, Lee DH, Jo E, Lim TW, Heo KS, Na M, Myung CS. Rubiarbonone C inhibits platelet-derived growth factor-induced proliferation and migration of vascular smooth muscle cells through the focal adhesion kinase, MAPK and STAT3 Tyr705 signalling pathways. Br J Pharmacol. 2017; 174:4140–4154.
23. Lee JJ, Zhang WY, Yi H, Kim Y, Kim IS, Shen GN, Song GY, Myung CS. Anti-proliferative actions of 2-decylamino-5,8-dimethoxy-1,4-naphthoquinone in vascular smooth muscle cells. Biochem Biophys Res Commun. 2011; 411:213–218.

24. Choi KW, Park HJ, Jung DH, Kim TW, Park YM, Kim BO, Sohn EH, Moon EY, Um SH, Rhee DK, Pyo S. Inhibition of TNF-α-induced adhesion molecule expression by diosgenin in mouse vascular smooth muscle cells via downregulation of the MAPK, Akt and NF-κB signaling pathways. Vascul Pharmacol. 2010; 53:273–280.

25. Horwitz SB. Taxol (paclitaxel): mechanisms of action. Ann Oncol. 1994; 5:Suppl 6. S3–S6.
26. Fukuoka K, Arioka H, Iwamoto Y, Fukumoto H, Kurokawa H, Ishida T, Tomonari A, Suzuki T, Usuda J, Kanzawa F, Saijo N, Nishio K. Mechanism of the radiosensitization induced by vinorelbine in human non-small cell lung cancer cells. Lung Cancer. 2001; 34:451–460.

27. Perez EA. Microtubule inhibitors: differentiating tubulin-inhibiting agents based on mechanisms of action, clinical activity, and resistance. Mol Cancer Ther. 2009; 8:2086–2095.

28. Wang RC, Chen X, Parissenti AM, Joy AA, Tuszynski J, Brindley DN, Wang Z. Sensitivity of docetaxel-resistant MCF-7 breast cancer cells to microtubule-destabilizing agents including vinca alkaloids and colchicine-site binding agents. PLoS One. 2017; 12:e0182400.

29. Perez-Vizcaino F, Bishop-Bailley D, Lodi F, Duarte J, Cogolludo A, Moreno L, Bosca L, Mitchell JA, Warner TD. The flavonoid quercetin induces apoptosis and inhibits JNK activation in intimal vascular smooth muscle cells. Biochem Biophys Res Commun. 2006; 346:919–925.

30. Tixeira R, Caruso S, Paone S, Baxter AA, Atkin-Smith GK, Hulett MD, Poon IK. Defining the morphologic features and products of cell disassembly during apoptosis. Apoptosis. 2017; 22:475–477.

31. Petiot A, Ogier-Denis E, Blommaart EF, Meijer AJ, Codogno P. Distinct classes of phosphatidylinositol 3’-kinases are involved in signaling pathways that control macroautophagy in HT-29 cells. J Biol Chem. 2000; 275:992–998.

32. Kim YC, Guan KL. mTOR: a pharmacologic target for autophagy regulation. J Clin Invest. 2015; 125:25–32.

33. García-Arencibia M, Hochfeld WE, Toh PP, Rubinsztein DC. Autophagy, a guardian against neurodegeneration. Semin Cell Dev Biol. 2010; 21:691–698.

34. Li BH, Yin YW, Liu Y, Pi Y, Guo L, Cao XJ, Gao CY, Zhang LL, Li JC. TRPV1 activation impedes foam cell formation by inducing autophagy in oxLDL-treated vascular smooth muscle cells. Cell Death Dis. 2014; 5:e1182.

35. Hill BG, Haberzettl P, Ahmed Y, Srivastava S, Bhatnagar A. Unsaturated lipid peroxidation-derived aldehydes activate autophagy in vascular smooth-muscle cells. Biochem J. 2008; 410:525–534.

36. Salabei JK, Cummins TD, Singh M, Jones SP, Bhatnagar A, Hill BG. PDGF-mediated autophagy regulates vascular smooth muscle cell phenotype and resistance to oxidative stress. Biochem J. 2013; 451:375–388.

37. Cj P, Hv E, Vijayakurup V, R Menon G, Nair S, Gopala S. High LC3/Beclin expression correlates with poor survival in glioma: a definitive role for autophagy as evidenced by in vitro autophagic flux. Pathol Oncol Res. 2017; DOI: 10.1007/s12253-017-0310-7. [Epub ahead of print].

38. van Meerloo J, Kaspers GJ, Cloos J. Cell sensitivity assays: the MTT assay. Methods Mol Biol. 2011; 731:237–245.

39. Zhang Y, Xia G, Zhang Y, Liu J, Liu X, Li W, Lv Y, Wei S, Liu J, Quan J. Palmitate induces VSMC apoptosis via toll like receptor (TLR)4/ ROS/p53 pathway. Atherosclerosis. 2017; 263:74–81.
40. Cao YN, Zheng LL, Wang D, Liang XX, Gao F, Zhou XL. Recent advances in microtubule-stabilizing agents. Eur J Med Chem. 2018; 143:806–828.

41. Hugle M, Belz K, Fulda S. Identification of synthetic lethality of PLK1 inhibition and microtubule-destabilizing drugs. Cell Death Differ. 2015; 22:1946–1956.

42. Porter AG, Jänicke RU. Emerging roles of caspase-3 in apoptosis. Cell Death Differ. 1999; 6:99–104.

43. Sollott SJ, Cheng L, Pauly RR, Jenkins GM, Monticone RE, Kuzuya M, Froehlich JP, Crow MT, Lakatta EG, Rowinsky EK, et al. Taxol inhibits neointimal smooth muscle cell accumulation after angioplasty in the rat. J Clin Invest. 1995; 95:1869–1876.

44. Mariño G, Niso-Santano M, Baehrecke EH, Kroemer G. Self-consumption: the interplay of autophagy and apoptosis. Nat Rev Mol Cell Biol. 2014; 15:81–94.

45. Nikoletopoulou V, Markaki M, Palikaras K, Tavernarakis N. Crosstalk between apoptosis, necrosis and autophagy. Biochim Biophys Acta. 2013; 1833:3448–3459.

46. Kang R, Zeh HJ, Lotze MT, Tang D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ. 2011; 18:571–580.

47. Circu ML, Aw TY. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med. 2010; 48:749–762.

48. Simon HU, Haj-Yehia A, Levi-Schaffer F. Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis. 2000; 5:415–418.
49. Sundaresan M, Yu ZX, Ferrans VJ, Irani K, Finkel T. Requirement for generation of H2O2 for platelet-derived growth factor signal transduction. Science. 1995; 270:296–299.
SUPPLEMENTARY MATERIALS
Supplementary data including two figures can be found with this article online at http://pdf.medrang.co.kr/paper/pdf/Kjpp/Kjpp022-03-13-s001.pdf.
Supplementary Fig. 1
Effects of paclitaxel and vinorelbine on VSMC proliferation and viability. Serum-starved VSMCs were incubated with 0 to 2000 nM paclitaxel or 0 to 1000 nM vinorelbine for 24 h, followed by 25 ng/ml PDGF-BB treatment for 48 h. VSMC proliferation and viability were evaluated using the MTT assay (A-D). Mean values of the vehicle group (0.1% DMSO) were set to 100%. Data are expressed as means±standard error of the mean (SEM) (n=3). *p<0.05, **p<0.01, ***p<0.001 vs. control (PDGF-BB alone) or vehicle.
Supplementary Fig. 2
Effects of the autophagy regulation with 3-MA or rapamycin on conditioned VSMCs. Serum-starved VSMCs were incubated with 1 µM paclitaxel, 0.2 µM vinorelbine, 5 mM 3-MA (autophagy inhibitor), or 0.2 µM rapamycin (autophagy stimulator) for 24 h followed by 25 ng/ml PDGF-BB treatment for 48 h. Additionally, the cells were treated with 0.1 µM bafilomycin A1 for 4 h before the end of the reaction. (A and B) The levels of LC3 (autophagy key marker) in microtubule-regulated VSMCs with 3-MA or rapamycin for 48 h. Bafilomycin A1 was added to the conditioned VSMCs, and PDGF-stimulated VSMCs 4 h prior to harvest, and autophagic flux was examined by measuring the accumulation of LC3-II using immunoblotting. The band densities were normalized to those of β-actin.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download