Abstract
Previous reports have suggested that physical and psychological stresses may trigger fibromyalgia (FM). Stress is an important risk factor in the development of depression and memory impairments. Antidepressants have been used to prevent stress-induced abnormal pain sensation. Among various antidepressants, tianeptine has been reported to be able to prevent neurodegeneration due to chronic stress and reverse decreases in hippocampal volume. To assess the possible effect of tianeptine on FM symptoms, we constructed a FM animal model induced by restraint stress with intermittent cold stress. All mice underwent nociceptive assays using electronic von Frey anesthesiometer and Hargreaves equipment. To assess the relationship between tianeptine and expression levels of brain-derived neurotrophic factor (BDNF), cAMP response element-binding protein (CREB), and phosphorylated cAMP response element-binding protein (p-CREB), western blotting and immunohistochemistry analyses were performed. In behavioral analysis, nociception tests showed that pain threshold was significantly decreased in the FM group compared to that in the control group. Western blot and immunohistochemical analyses of medial prefrontal cortex (mPFC) and hippocampus showed downregulation of BDNF and p-CREB proteins in the FM group compared to the control group. However, tianeptine recovered these changes in behavioral tests and protein level. Therefore, this FM animal model might be useful for investigating mechanisms linking BDNF-CREB pathway and pain. Our results suggest that tianeptine might potentially have therapeutic efficacy for FM.
Fibromyalgia (FM) is characterized by widespread pain and other clinical manifestations such as sleep disturbance, fatigue, gastrointestinal symptoms, headache, and depression. It affects 2~4% of the population [12]. Stress has been discussed as an etiological factor in the manifestation of chronic pain [3]. Stress can produce hyperalgesia (increased sensitivity to painful stimuli) or allodynia (pain triggered by innocuous stimuli) [4]. It has been reported that chronic pain or FM-like rodent model induced by intermittent cold stress (ICS) is useful for inducing abnormal pain, including mechanical allodynia and hyperalgesia [5]. Chronic restraint stress (CRS) can particularly cause neuronal damage, including atrophy of the hippocampus CA3 region. In addition, CRS can decrease neurogenesis and synaptic plasticity, thus resulting in impaired cognitive function [67]. Previous studies have demonstrated that emotional dysfunction induced by continuous pain is related to functional alterations in the medial prefrontal cortex, hippocampus, and striatum in rodents [8910]. Moreover, mPFC and anterior cingulate cortex are involved in processing negative emotional components associated with pain [11].
FM-related pain is often treated by common painkillers such as non-steroidal anti-inflammatory drugs (NSAIDs) and opioids [12]. Other studies have demonstrated that antidepressants and antiepileptics such as gabapentin and pregabalin are effective for FM pain [1314]. The efficacy of some classes of antidepressants has been proposed to involve increased phosphorylation of cAMP response element binding protein (CREB) which leads to increased expression of brain-derived neurotrophic factor (BDNF) [15]. BDNF is highly expressed in the hippocampus and the cerebral cortex where it contributes to neuronal development, growth, plasticity, survival, neuroprotection, and repair [161718]. Antidepressant treatment can recover BDNF serum levels in depressed patients. In addition, BDNF levels are closely linked to chronic stress and psychiatric illnesses including depression [171819]. Both serum and plasma BDNF concentrations are decreased in patients with major depressive disorder [20]. However, antidepressants can normalize these abnormalities [21]. It has been reported that rats exhibiting thermal hyperalgesia after loose ligation of the sciatic nerve have significant increase in BDNF levels in the lumbar spinal dorsal horn [22]. BDNF function is also strongly associated with the transmission of nociceptive information related to BDNF receptor tropomyosin receptor kinase (TrkB), extracellular signal regulated kinase (ERK), and CREB of ERK downstream target [23]. CREB is a transcription factor that plays a key role in adaptive neuronal responses. It also has complex functions in the regulation of learning and memory [23]. A recent study has suggested that CREB is phosphorylated at serine 133 (p-CREB) and activated upon stimulation of painproducing signals [24]. CREB seems to mediated by ERK [24]. Activated CREB can further induce the activation of c-Fos, a marker of neuronal activation within spinal nociceptive and brainstem pathways [24]. Furthermore, this pain pathway partly affects the efficiency of glutamatergic and GABAergic/glycinergic synapses [25]. It has been reported that repeated injection of specific antibody to BDNF can markedly suppress thermal hyperalgesia associated with increased levels of tropomyosin receptor kinase B (TrkB) receptor following sciatic nerve ligation in mice [26]. These findings have demonstrated that the release of endogenous BDNF may cause excitability of some neurons through activating TrkB receptor, resulting in neuropathic painlike state. The role of BDNF in pain state has received more attention as a neuromediator of hyperalgesia and spinal central sensitization [2728]. It has been reported that infusion of BDNF into the midbrain can result in antidepressant-like effect in animal models [29]. Several additional tests have been conducted to determine whether BDNF infusions into hippocampus might result in behavioral effects [2829]. Interestingly, hippocampal BDNF administration has antidepressant-like effects [30]. Furthermore, antidepressants can significantly increase the expression of BDNF in major subfields of the hippocampus, including the granule cell layer and CA1 and CA3 pyramidal cell layers [3132]. It has been suggested that tianeptine may promote neuroplasticity by increasing BDNF expression which is decreased in animal models of stress involving CREB expression [33]. As one atypical antidepressant, tianeptine exhibits structural similarities to tricyclic antidepressants. Some antidepressants can affect presynaptic reuptake of serotonin and norepinephrine and increase their levels in the synaptic cleft [34]. Antinociceptive activity of tianeptine has been reported in animal models of acute nociception [35], morphine tolerance [36], and inflammatory pain [37]. Stress-induced morphological changes in the hippocampus are increased by some antidepressants such as tianeptine, lithium, and clomipramine [283839]. Effects of tianeptine on nociception in mice have been investigated previously through thermal analgesic tests [35]. It has been reported that tianeptine has significant dose-dependent analgesic activity in mice [35]. Chronic tianeptine therapy can significantly decrease both apoptosis aggravated by stress and apoptosis in normal samples in the temporal cortex and hippocampus [40]. Furthermore, some clinical studies have indicated that tianeptine can be used to effectively treat somatic anxiety symptoms accompanying depression with possible anxiolytic effects [414243].
Therefore, the objective of this study was to determine whether administration of tianeptine might have therapeutic efficacy in a FM mouse model. The model was constructed to evaluate the analgesic efficacy of tianeptine using behavioral pain tests involving either noxious or non-noxious mechanical (electronic von Frey filament) or thermal (tail flick test, plantar test) stimuli. Tail suspension test was used to evaluate the activity of depressant. We also examined the ability of tianeptine to modulate phosphorylated CREB (p-CREB) and BDNF expression in the mPFC and hippocampus of mouse.
Male adult C57BL/6J mice were obtained from Daehan Biolink, Ink. (Eumsung, Chungbuk, Korea). They were housed in clear cages with free access to food and water. They were maintained on a 12:12 h light-dark cycle (lights on at 6:30 a.m.). All animal procedures were performed in accordance with the guidelines for the care and use of laboratory animals of the US National Institutes of Health. Animal protocols were approved by the Institutional Animal Care and Use Committee of Soonchunhyang University (Asan, Korea, SCH16-0061).
Tianeptine (Sigma-Aldrich, Seoul, Korea) was dissolved in 0.9% NaCl and injected intraperitoneally (10 mg/kg/daily once) for 24 days. Each drug treatment was performed every day at 10 a.m. during stress procedure.
Adult mice were subjected to CRS as described previously [4445]. Briefly, mice were restrained for 6 h (from 12 A.M. to 6 P.M.) daily in well-ventilated 50-ml conical tube without food or water. Control mice remained undisturbed in their home cages. Restraint stressor was proceeded for 21 days. Cold stress procedure was performed on stainless mesh plate in a cold room at 4℃ overnight (from 4:30 pm to 10:00 am). Environmental temperatures were alternated between 24℃ and 4℃ every 30 min from 10:00 am to 4:30 pm. These procedures were repeated for 2 days. On day 3, mice were adapted to 24℃ for 1 h before behavior test. We designated day 3 following the onset of cold stress exposure after day 21 of restraint stress. Control group mice were kept at 24℃ for all 3 days (from 4:30 pm on day 1 to 10:00 am on day 3) [46].
Behavior assessments were started one day after the last day of clod stress (N=10~12 animals/group). Mice were allowed to acclimate to a testing room for at least 1 h before performing assessments. Modified behavioral tests including TST, TFT, PWL, and PWT were performed as described previously. In experiments of thermal and mechanical tests, thresholds were determined from three repeated challenges with 15 min intervals. Average values were used for statistical analysis.
TST has become one of the most widely used models for assessing depression-like behaviors in mice. Mice were subjected to short-term inescapable stress. Their tails were suspended to develop an immobile posture. The total duration of immobility induced by tail suspension was measured according to published procedures [47]. Mice were suspended 50 cm above the floor using adhesive tape placed approximately 1 cm from the tip of the tail. The total duration of immobility during a 6-min period was measured.
TFT was used to assess the antinociceptive effect of drug by measuring the latency time [4849]. Latency time is the time from the onset of radiant heat exposure to withdrawal of the tail. Thermal pain threshold of the tail was measured with a tail flick apparatus (IITC Inc., Woodland Hills, CA, USA). Each mouse was gently restrained under a 50-ml conical tube with light manual pressure to minimize stress. Radiant heat was applied to tail (2 cm distal part) of the animal and latency (in second) for the tail to flick away from the radiant heat was scored. A cutoff value of 20 sec of radiant heat application was used to avoid tissue damage to the tail. Each mouse was tested three times at each time point with an interval of 15 min between replicates. Three replicates were averaged to obtain tail withdrawal latency.
In PWL, the nociception threshold was assessed by latency time of paw withdrawal upon a thermal stimulus as described previously [5051]. Mice were placed in individual clear plastic chamber on the top of a glass sheet and acclimated for 1 h. Radiant heat (IITC Inc., Woodland Hills, CA, USA) was positioned under a glass sheet. The projection bulb was focused on the plantar surface of the hind paw of the animal. Paw withdrawal latency defined as the first occurrence of licking the hind paw was scored. Each mouse was tested three times at each time point with an interval of 15 min between replicates. Three replicates were averaged to obtain paw withdrawal latency.
The pressure needed to induce a flexor response was defined as the mechanical threshold. PWT was carried out using an electronic von Frey anesthesiometer (Anesthesiometer, IITC Inc., Woodland Hills, USA) as previously reported [5253]. Mice were placed in a plastic chamber on a wire mesh grid floor and allowed to acclimate for 1 h. The maximum response threshold was set at rigid tip to prevent tissue damage.
After behavior tests, experimental subjects were placed in 50-ml conical tubes for 60 min [54]. Mice were decapitated rapidly at the end of the stress test and trunk blood (about 1 ml) was collected into centrifugal tubes. Blood samples were centrifuged (12,000 rpm, 10 min) to separate serum for corticosterone assay. Serum samples were stored at −80℃ until assay. Serum corticosterone levels were determined using Corticosterone ELISA Kit (Cayman chemical, Cat No. 501320; Ann Arbor, MI, USA). Assays were conducted according to the manufacturer's protocol.
Whole tissue of medial prefrontal cortex (mPFC) and hippocampus were lysed in RIPA buffer containing protease inhibitors, leupeptin, and sodium fluoride followed by centrifugation at 14,000 rpm for 10 min at 4℃ (N=6~7 animals per group). Protein samples (100 µg) were electrophoretically separated on 15% sodium dodecyl sulfate-polyacrylamide gel and transferred to polyvinylidene difluoride membrane (Millipore, Milford, MA, USA). After blocking with 5% skim milk, membranes were probed with anti-phospho-CREB (p-CREB; 1:1,000; Cell Signaling Technology, Danvers, MA, USA), anti-CREB (CREB; 1:1,000; Cell Signaling Technology, Danvers, MA, USA), anti-BDNF (BDNF; 1:3,000; Abcam, Cambridge, UK), or anti-β-tubulin (Tubb; 1:3,000; Thermo Fisher Scientific, Inc., Rockford, IL) antibody overnight at 4℃. After washing with PBS three times, membranes were then incubated with peroxidase-conjugated secondary rabbit antibody (1:5,000; Abfrontier. Geumcheon-gu, Seoul, Korea) at room temperature for 1 h. Immunoreactive bands were detected using enhanced chemiluminescence kit (ELPIS-Biotech Inc., Daejeon, Korea). Quantitative measurements of p-CREB, CREB, BDNF, and β-tubulin proteins were obtained using ImageJ software (http://imagej.nih.gov/ij).
Mice were perfused through the left cardiac ventricle with 4% paraformaldehyde. Fixed brains were removed, frozen, and cut into 30-µm sections (N=4~5 animals/group). Frozen sections from the hippocampus were treated with 0.3% hydrogen peroxide (H2O2) for 5 min followed by blocking with normal horse serum. They were then incubated with anti-phospho-CREB (p-CREB1:800; Cell Signaling Technology, Danvers, MA, USA), anti-CREB (CREB; 1:800; Cell Signaling Technology, Danvers, MA, USA), and Cy3-conjugated anti-rabbit or anti-mouse secondary antibodies (1:500; Jackson ImmunoResearch Laboratories, Inc., West Grove, PA, USA). After washing with PBS, sections were stained with 4'6-diamidino-2-phenylindole (DAPI) to identify nuclei. Fluorescent images were captured using a confocal laser-scanning microscope (FV10-ASW; Olympus; Tokyo, Japan). Images were quantified with Image J software (http://imagej.nih.gov/ij) using a protocol described previously [55] with slight modifications.
All data are expressed as means±standard deviation and/or standard error of the mean. Results between groups were compared using Student's t-test. All statistical analyses were performed using IBM SPSS Statistics 19 software (SPSS Inc, Chicago, IL, USA). A p-value of less than 0.05 was considered as statistically significant.
In TST, the duration of immobility was measured to determine stress-related despairing status in mice. The immobility time of mice in the FM group was significantly (p<0.05) longer than that in the control group (Fig. 2). After tianeptine administration, the immobility time was significantly (p<0.01) shorter compared to that in the FM group (Fig. 2), suggesting that tianeptine could reverse the despairing status in the FM group.
The tail-flick latency was significantly different among the control group, FM group, and tianeptine-administered FM group in the 2 cm distal part of the tail (Fig. 3). The FM group showed significant (p<0.01) decrease in tail-flick latency compared to the control group (Fig. 3). Such decrease in latency was recovered (p<0.01) in the tianeptine-administered FM group (Fig. 3).
Significant differences in PWL were found among the control, FM, and tianeptine-administered FM groups for both right and left paws (Fig. 4). The FM group exhibited significant (p<0.01) decrease in PWL compared to the control group (Fig. 4). Such decrease in PWL in the plantar test was recovered (p<0.01) in the tianeptine-administered FM group (Fig. 4).
Tianeptine-administration showed beneficial effects on the PWT for both right and left paws (Fig. 5). The FM group exhibited significant (p<0.01) decrease in PWT compared to the control group (Fig. 5). Such decrease in response time in the FM group was recovered (p<0.01) in the tianeptine-administered FM group (Fig. 5).
The BDNF-CREB pathway is known to be related to depression and pain [56]. To determine whether BDNF-CREB pathway was involved in behavioral abnormalities of the FM group, the mPFC and hippocampus areas from the brains of the control, FM, and tianeptine-administered FM groups were subjected to Western blot analyses and immunohistochemical analyses. Western blot analyses results (Fig. 6) revealed that protein levels of BDNF and p-CREB in the mPFC and hippocampal in the FM group were significantly (p<0.05) lower than those in the control group. These changes were restored (p<0.05) by tianeptine administration (Fig. 6 and Fig. 7). Immunohistochemistry-stained brain images (Fig. 7) revealed that BDNF and p-CREB were differentially expressed among the control, FM, and tianeptine-administered FM groups. Intensity values of immunohistochemical staining between control and FM groups or between FM and FM-TIA groups were significantly (p<0.05) different (Fig. 8).
Previous investigations have implicated that changes in central nervous system circuitry are the underlying mechanisms of stress-induced hyperalgesia [57]. BDNF is one of the most extensively investigated proteins in brain plasticity. It has been proposed that BDNF participates in hypothalamic-pituitary-adrenal (HPA) axis activity by stress [58]. Reduced BDNF expression contributes to structural anomalies and functional impairment in local brain legions. Other studies have shown that a variety of drugs can concurrently increase p-CREB and BDNF levels in rat hippocampus [59]. Increased expression and activation of CREB has been linked to BDNF of hippocampus [31]. Our FM-like animal model showed increased immobility time in TST but decreased mechanical and thermal thresholds based on results of TST, TFT, PWT, and PWL. These changes were accompanied by decreased levels of BDNF expression and phosphorylation of CREB in the hippocampus and mPFC. Mice exhibited significant changes in serum corticosterone levels after 1 h of restraint stress. However, these changes were reversed by tianeptine administration. Tianeptine treatment was able to normalize both behavioral and protein alterations elicited by stress. Results of this study demonstrated that stress-induced corticosterone level might be related to abnormal pain behavior through BDNF-CREB signaling. Depression symptoms are frequently reported to be associated with disturbed glucocorticoid secretion in patients with depression and in many animal models of depression [60]. Therefore, dysfunction of the HPA axis has been investigated through assessment of serum corticosterone level [60]. In this study, FM animal model showed significant increase in serum corticosterone level in response to stress, indicating hyperactivity in the HPA axis, consistent with results of many previous studies [6162].
Antidepressants block changes that contribute to long-term nociceptive sensory plasticity in the spinal cord and limbic regions involved in the modulation of affect [6364]. Recommended pharmacotherapy for neuropathic pain includes the use of some antidepressants such as tricycle antidepressant (TCAs) or serotonin and noradrenaline re-uptake inhibitors [65]. Antidepressant drugs require chronic treatment to control neuropathic pain, suggesting recruitment of secondary downstream mechanisms and long-term molecular mechanisms. In TST of this study, tianeptine improved stress-induced depressive status, in correlation with BDNF and CREB results. In TFT and PWL, tianeptine reversed stress-induced thermal and mechanical hyperalgesia because it increased the latency time in radiant heat application. In the PWT test, tianeptine exerted the maximum threshold in mice. Among various TCAs, amitriptyline is the most studied and the most prescribed TCA clinically. It has efficacy for several neuropathic pain conditions, including postherpetic neuralgia and central pain [66]. Unlike classical analgesics, the effect of antidepressants on neuropathic pain is observed after prolonged treatment [67]. Selective serotonin reuptake inhibitors such as fluoxetine, paroxetine, and citalopram have been used for painful diabetic neuropathy [666869]. The analgesic effect of tianeptine was also confirmed in our animal model, suggesting that tianeptine might have beneficial effect on pain-depression syndrome. Results of the present study confirmed that stressful exposure could impair pain-related symptoms via BDNF-CREB pathway in FM animal model.
In conclusion, tianeptine treatment can rescue repeated stress-induced depression and pain symptoms through activating the BDNF-CREB pathway. Our results provide a potential mechanistic explanation for the pathophysiology of FM with depressive symptoms and enhanced pain.
Figures and Tables
 | Fig. 1Corticosterone concentration changes after stressful paradigm.Corticosterone secretion was induced by restraint stress in FM mice (n=7 per group). Corticosterone level was expressed in concentration of pg/ml. Data are presented as means±SEM (*p<0.05 vs. CON; †p<0.05 vs. FM). FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model.
|
 | Fig. 2Effect of tianeptine on immobility time in mice subjected to tail suspension test.The total duration of immobility during a 6-min period was measured (n=7 per group). Data are presented as means±SEM (*p<0.05 vs. CON; †p<0.05 vs. FM). FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model.
|
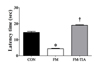 | Fig. 3Effect of tianeptine on nociceptive response via tail flick test in FM model.Latency time of tail flicking away from radiant heat was scored (n=7 per group). Comparison was made between Con and FM groups. Data are presented in second as means±SEM (*p<0.05 vs. CON; †p<0.05 vs. FM). FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model.
|
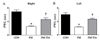 | Fig. 4Effect of tianeptine on nociceptive response in FM model.(A) Results of the right hind paw. (B) Results of the left hind paw. Thermal pain threshold was assessed via plantar test (n=7 per group). Graphs are shown in means±SEM of response frequency to heat stimulation (*p<0.05 vs. CON; †p<0.05 vs. FM). PWL, paw withdrawal time; FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model.
|
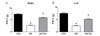 | Fig. 5Effect of tianeptine on nociceptive response in FM model using digital von Frey.(A) Results of the right hind paw. (B) Results of the left hind paw. Withdrawal responses to von Frey filaments from both hind paws were counted and then expressed as an average in gram (n=7 per group). Comparison was made between CON and FM groups. Data are presented as means±SEM (*p<0.05 vs. CON; †p<0.05 vs. FM). PWT, paw withdrawal threshold; FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model.
|
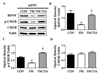 | Fig. 6Western blot analysis of BDNF, p-CREB, and CREB in mPFC.(A) Expression levels of BDNF, p-CREB, and CREB were detected by western blot analysis using anti-β-tubulin antibody as an internal control. Graph demonstrating the ratio of p-CREB or BDNF to Tubb. (B~D) Quantitative analysis of western blot results for BDNF, pCREB, CREB expression levels in the medial prefrontal cortex (*p<0.05 vs. CON; †p<0.05 vs. FM). FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model.
|
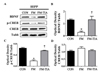 | Fig. 7Western blot analysis of BDNF, p-CREB, and CREB in hippocampus.(A) Expression levels of BDNF, CREB, and CREB were detected by western blot analysis using anti-β-tubulin antibody as an internal control. Graph demonstrating the ratio of p-CREB or BDNF to Tubb quantified using ImageJ software. (B~D) Quantitative analysis of western blot results of BDNF, pCREB, an CREB in the hippocampus (*p<0.05 vs. CON; †p<0.05 vs. FM). FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model.
|
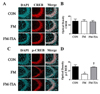 | Fig. 8Patterns of p-CREB expression across the hippocampus in the FM model and tianeptine-administered FM model group.(A) Immunohistochemistry analysis showing p-CREB immunoreactivity in the hippocampus. Confocal microscopy image showed immunofluorescent staining for p-CREB (anti-p-CREB, red, Cy3) with DAPI (blue) in CA3 region of hippocampus. Scale bar, 50 µm. (B) Optical densities of p-CREB signals in immunostained hippocampus sections (*p<0.05 vs. CON; †p<0.05 vs. FM). (C) Confocal microscopy image showing immunofluorescent staining for CREB (anti-CREB, red, Cy3) with DAPI (blue) in the hippocampus. Scale bar, 50 µm. (D) Optical densities of CREB signals in immunostained hippocampus sections (*p<0.05 vs. CON; †p<0.05 vs. FM). Quantitative measurements of p-CREB, CREB, BDNF, and β-tubulin proteins were obtained using ImageJ software (http://imagej.nih.gov/ij). HIPP, hippocampus; FM, Fibromyalgia animal model; CON, non-FM animal model; FM-TIA, Tianeptine administered FM model; p-CREB, phospho CREB; CREB, cAMP response element-binding protein.
|
ACKNOWLEDGEMENTS
The present study was performed with the support of the ‘Cooperative Research Program for Agriculture Science and Technology Development (project no. PJ011582)’ of the Rural Development Administration, Korea.
Notes
Author contributions Author H.J.K. designed and directed the entire project. H.K.K., J.T.K., and Y.O.K. managed literature searches and analyses. J.I. and H.W. took care of animals and assisted most of animal behavior tests. Authors S.W.L., S.L., I.H.C. undertook statistical analysis. Author H.Y.L. performed most of animal behavior tests and protein expression analysis and contributed substantially to the first draft of the manuscript. All authors contributed to and approved the final version of the manuscript.
References
1. Clauw DJ. Fibromyalgia: more than just a musculoskeletal disease. Am Fam Physician. 1995; 52:843–851. 853–854.
2. Rohrbeck J, Jordan K, Croft P. The frequency and characteristics of chronic widespread pain in general practice: a case-control study. Br J Gen Pract. 2007; 57:109–115.
3. Chrousos GP, Gold PW. The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis. JAMA. 1992; 267:1244–1252.
4. Bardin L, Malfetes N, Newman-Tancredi A, Depoortère R. Chronic restraint stress induces mechanical and cold allodynia, and enhances inflammatory pain in rat: Relevance to human stress-associated painful pathologies. Behav Brain Res. 2009; 205:360–366.
5. Nishiyori M, Ueda H. Prolonged gabapentin analgesia in an experimental mouse model of fibromyalgia. Mol Pain. 2008; 4:52.
6. Magariños AM, McEwen BS. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience. 1995; 69:89–98.
7. Zafir A, Banu N. Modulation of in vivo oxidative status by exogenous corticosterone and restraint stress in rats. Stress. 2009; 12:167–177.
8. Goffer Y, Xu D, Eberle SE, D'amour J, Lee M, Tukey D, Froemke RC, Ziff EB, Wang J. Calcium-permeable AMPA receptors in the nucleus accumbens regulate depression-like behaviors in the chronic neuropathic pain state. J Neurosci. 2013; 33:19034–19044.
9. Mutso AA, Radzicki D, Baliki MN, Huang L, Banisadr G, Centeno MV, Radulovic J, Martina M, Miller RJ, Apkarian AV. Abnormalities in hippocampal functioning with persistent pain. J Neurosci. 2012; 32:5747–5756.
10. Norman GJ, Karelina K, Zhang N, Walton JC, Morris JS, Devries AC. Stress and IL-1beta contribute to the development of depressive-like behavior following peripheral nerve injury. Mol Psychiatry. 2010; 15:404–414.
11. De Ryck LM, Raus JC. Superfusion of normal and neoplastic mouse mammary tissue with estrogens. Endocrinology. 1983; 113:399–408.
12. Arnold LM, Bradley LA, Clauw DJ, Glass JM, Goldenberg DL. Multidisciplinary care and stepwise treatment for fibromyalgia. J Clin Psychiatry. 2008; 69:e35.
13. Arnold LM, Goldenberg DL, Stanford SB, Lalonde JK, Sandhu HS, Keck PE Jr, Welge JA, Bishop F, Stanford KE, Hess EV, Hudson JI. Gabapentin in the treatment of fibromyalgia: a randomized, double-blind, placebo-controlled, multicenter trial. Arthritis Rheum. 2007; 56:1336–1344.
14. Arnold LM, Russell IJ, Diri EW, Duan WR, Young JP Jr, Sharma U, Martin SA, Barrett JA, Haig G. A 14-week, randomized, double-blinded, placebo-controlled monotherapy trial of pregabalin in patients with fibromyalgia. J Pain. 2008; 9:792–805.
15. M'Dahoma S, Barthélemy S, Tromilin C, Jeanson T, Viguier F, Michot B, Pezet S, Hamon M, Bourgoin S. Respective pharmacological features of neuropathic-like pain evoked by intrathecal BDNF versus sciatic nerve ligation in rats. Eur Neuropsychopharmacol. 2015; 25:2118–2130.
16. Schmidt-Kastner R, Wetmore C, Olson L. Comparative study of brain-derived neurotrophic factor messenger RNA and protein at the cellular level suggests multiple roles in hippocampus, striatum and cortex. Neuroscience. 1996; 74:161–183.
17. Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry. 2006; 59:1116–1127.
18. Shimizu E, Hashimoto K, Okamura N, Koike K, Komatsu N, Kumakiri C, Nakazato M, Watanabe H, Shinoda N, Okada S, Iyo M. Alterations of serum levels of brain-derived neurotrophic factor (BDNF) in depressed patients with or without antidepressants. Biol Psychiatry. 2003; 54:70–75.
19. McEwen BS. Glucocorticoids, depression, and mood disorders: structural remodeling in the brain. Metabolism. 2005; 54:5 Suppl 1. 20–23.
20. Piccinni A, Marazziti D, Catena M, Domenici L, Del Debbio A, Bianchi C, Mannari C, Martini C, Da Pozzo E, Schiavi E, Mariotti A, Roncaglia I, Palla A, Consoli G, Giovannini L, Massimetti G, Dell'Osso L. Plasma and serum brain-derived neurotrophic factor (BDNF) in depressed patients during 1 year of antidepressant treatments. J Affect Disord. 2008; 105:279–283.
21. Sen S, Duman R, Sanacora G. Serum brain-derived neurotrophic factor, depression, and antidepressant medications: meta-analyses and implications. Biol Psychiatry. 2008; 64:527–532.
22. Miletic G, Miletic V. Increases in the concentration of brain derived neurotrophic factor in the lumbar spinal dorsal horn are associated with pain behavior following chronic constriction injury in rats. Neurosci Lett. 2002; 319:137–140.
23. Guo JQ, Deng HH, Bo X, Yang XS. Involvement of BDNF/TrkB and ERK/CREB axes in nitroglycerin-induced rat migraine and effects of estrogen on these signals in the migraine. Biol Open. 2017; 6:8–16.
24. Bhatt DK, Ramachandran R, Christensen SL, Gupta S, Jansen-Olesen I, Olesen J. CGRP infusion in unanesthetized rats increases expression of c-Fos in the nucleus tractus solitarius and caudal ventrolateral medulla, but not in the trigeminal nucleus caudalis. Cephalalgia. 2015; 35:220–233.
25. Buldyrev I, Tanner NM, Hsieh HY, Dodd EG, Nguyen LT, Balkowiec A. Calcitonin gene-related peptide enhances release of native brain-derived neurotrophic factor from trigeminal ganglion neurons. J Neurochem. 2006; 99:1338–1350.
26. Yajima Y, Narita M, Narita M, Matsumoto N, Suzuki T. Involvement of a spinal brain-derived neurotrophic factor/full-length TrkB pathway in the development of nerve injury-induced thermal hyperalgesia in mice. Brain Res. 2002; 958:338–346.
27. Clauw DJ. Fibromyalgia: an overview. Am J Med. 2009; 122:12 Suppl. 3S–13S.
28. Siuciak JA, Lewis DR, Wiegand SJ, Lindsay RM. Antidepressant-like effect of brain-derived neurotrophic factor (BDNF). Pharmacol Biochem Behav. 1997; 56:131–137.
29. Shirayama Y, Chen AC, Nakagawa S, Russell DS, Duman RS. Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J Neurosci. 2002; 22:3251–3261.
30. Schmidt HD, Duman RS. Peripheral BDNF produces antidepressant-like effects in cellular and behavioral models. Neuropsychopharmacology. 2010; 35:2378–2391.
31. Nibuya M, Morinobu S, Duman RS. Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci. 1995; 15:7539–7547.
32. Nibuya M, Nestler EJ, Duman RS. Chronic antidepressant administration increases the expression of cAMP response element binding protein (CREB) in rat hippocampus. J Neurosci. 1996; 16:2365–2372.
33. McEwen BS, Chattarji S, Diamond DM, Jay TM, Reagan LP, Svenningsson P, Fuchs E. The neurobiological properties of tianeptine (Stablon): from monoamine hypothesis to glutamatergic modulation. Mol Psychiatry. 2010; 15:237–249.
34. Watson PN, Merskey H. Antidepressant analgesics in pain management. Encyclopedia of Pain. 2007. p. 100–104.
35. Uzbay IT, Cinar MG, Aytemir M, Tuglular I. Analgesic effect of tianeptine in mice. Life Sci. 1999; 64:1313–1319.
36. Chu CC, Wang JJ, Chen KT, Shieh JP, Wang LK, Shui HA, Ho ST. Neurotrophic effects of tianeptine on hippocampal neurons: a proteomic approach. J Proteome Res. 2010; 9:936–944.
37. Kim WM, Lee SH, Jeong HJ, Lee HG, Choi JI, Yoon MH. The analgesic activity of intrathecal tianeptine, an atypical antidepressant, in a rat model of inflammatory pain. Anesth Analg. 2012; 114:683–689.
38. Czéh B, Michaelis T, Watanabe T, Frahm J, de Biurrun G, van Kampen M, Bartolomucci A, Fuchs E. Stress-induced changes in cerebral metabolites, hippocampal volume, and cell proliferation are prevented by antidepressant treatment with tianeptine. Proc Natl Acad Sci U S A. 2001; 98:12796–12801.
39. Wood GE, Young LT, Reagan LP, Chen B, McEwen BS. Stress-induced structural remodeling in hippocampus: prevention by lithium treatment. Proc Natl Acad Sci U S A. 2004; 101:3973–3978.
40. Uzbay TI. Tianeptine: potential influences on neuroplasticity and novel pharmacological effects. Prog Neuropsychopharmacol Biol Psychiatry. 2008; 32:915–924.
41. Defrance R, Marey C, Kamoun A. Antidepressant and anxiolytic activities of tianeptine: an overview of clinical trials. Clin Neuropharmacol. 1988; 11:Suppl 2. S74–S82.
42. Guelfi JD, Pichot P, Dreyfus JF. Efficacy of tianeptine in anxious-depressed patients: results of a controlled multicenter trial versus amitriptyline. Neuropsychobiology. 1989; 22:41–48.
43. Wilde MI, Benfield P. Tianeptine. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in depression and coexisting anxiety and depression. Drugs. 1995; 49:411–439.
44. Ejchel-Cohen TF, Wood GE, Wang JF, Barlow K, Nobrega JN, S McEwen B, Trevor Young L. Chronic restraint stress decreases the expression of glutathione S-transferase pi2 in the mouse hippocampus. Brain Res. 2006; 1090:156–162.
45. Magariños AM, Li CJ, Gal Toth J, Bath KG, Jing D, Lee FS, McEwen BS. Effect of brain-derived neurotrophic factor haploinsufficiency on stress-induced remodeling of hippocampal neurons. Hippocampus. 2011; 21:253–264.
46. Nishiyori M, Uchida H, Nagai J, Araki K, Mukae T, Kishioka S, Ueda H. Permanent relief from intermittent cold stress-induced fibromyalgia-like abnormal pain by repeated intrathecal administration of antidepressants. Mol Pain. 2011; 7:69.
47. Steru L, Chermat R, Thierry B, Simon P. The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology (Berl). 1985; 85:367–370.
48. Keyhanfar F, Shamsi Meymandi M, Sepehri G, Rastegaryanzadeh R, Heravi G. Evaluation of antinociceptive effect of pregabalin in mice and its combination with tramadol using tail flick test. Iran J Pharm Res. 2013; 12:483–493.
49. Meymandi MS, Sepehri G, Mobasher M. Gabapentin enhances the analgesic response to morphine in acute model of pain in male rats. Pharmacol Biochem Behav. 2006; 85:185–189.
50. Hargreaves K, Dubner R, Brown F, Flores C, Joris J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain. 1988; 32:77–88.
51. Reddyjarugu B, Pavek T, Southard T, Barry J, Singh B. Analgesic efficacy of firocoxib, a selective inhibitor of cyclooxygenase 2, in a mouse model of incisional pain. J Am Assoc Lab Anim Sci. 2015; 54:405–410.
52. Inoue M, Rashid MH, Fujita R, Contos JJ, Chun J, Ueda H. Initiation of neuropathic pain requires lysophosphatidic acid receptor signaling. Nat Med. 2004; 10:712–718.
53. Rashid MH, Inoue M, Toda K, Ueda H. Loss of peripheral morphine analgesia contributes to the reduced effectiveness of systemic morphine in neuropathic pain. J Pharmacol Exp Ther. 2004; 309:380–387.
54. Morley-Fletcher S, Darnaudery M, Koehl M, Casolini P, Van Reeth O, Maccari S. Prenatal stress in rats predicts immobility behavior in the forced swim test. Effects of a chronic treatment with tianeptine. Brain Res. 2003; 989:246–251.
55. Joo J, Lee S, Nah SS, Kim YO, Kim DS, Shim SH, Hwangbo Y, Kim HK, Kwon JT, Kim JW, Song HY, Kim HJ. Lasp1 is down-regulated in NMDA receptor antagonist-treated mice and implicated in human schizophrenia susceptibility. J Psychiatr Res. 2013; 47:105–112.
56. Merighi A, Salio C, Ghirri A, Lossi L, Ferrini F, Betelli C, Bardoni R. BDNF as a pain modulator. Prog Neurobiol. 2008; 85:297–317.
57. Imbe H, Iwai-Liao Y, Senba E. Stress-induced hyperalgesia: animal models and putative mechanisms. Front Biosci. 2006; 11:2179–2192.
58. Nowacka MM, Paul-Samojedny M, Bielecka AM, Plewka D, Czekaj P, Obuchowicz E. LPS reduces BDNF and VEGF expression in the structures of the HPA axis of chronic social stressed female rats. Neuropeptides. 2015; 54:17–27.
59. Duman RS, Vaidya VA. Molecular and cellular actions of chronic electroconvulsive seizures. J ECT. 1998; 14:181–193.
60. Zunszain PA, Anacker C, Cattaneo A, Carvalho LA, Pariante CM. Glucocorticoids, cytokines and brain abnormalities in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2011; 35:722–729.
61. Grippo AJ, Sullivan NR, Damjanoska KJ, Crane JW, Carrasco GA, Shi J, Chen Z, Garcia F, Muma NA, Van de Kar LD. Chronic mild stress induces behavioral and physiological changes, and may alter serotonin 1A receptor function, in male and cycling female rats. Psychopharmacology (Berl). 2005; 179:769–780.
62. Mizuki I, Sato A, Matsuo A, Suyama Y, Suzuki J, Makita A. Clonal structure, seed set, and self-pollination rate in mass-flowering bamboo species during off-year flowering events. PLoS One. 2014; 9:e105051.
63. Duric V, McCarson KE. Effects of analgesic or antidepressant drugs on pain- or stress-evoked hippocampal and spinal neurokinin-1 receptor and brain-derived neurotrophic factor gene expression in the rat. J Pharmacol Exp Ther. 2006; 319:1235–1243.
64. Kremer M, Yalcin I, Nexon L, Wurtz X, Ceredig RA, Daniel D, Hawkes RA, Salvat E, Barrot M. The antiallodynic action of pregabalin in neuropathic pain is independent from the opioid system. Mol Pain. 2016; 12:1744806916633477.
65. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007; (4):CD005454.
66. Sindrup SH, Otto M, Finnerup NB, Jensen TS. Antidepressants in the treatment of neuropathic pain. Basic Clin Pharmacol Toxicol. 2005; 96:399–409.
67. Sindrup SH, Gram LF, Brøsen K, Eshøj O, Mogensen EF. The selective serotonin reuptake inhibitor paroxetine is effective in the treatment of diabetic neuropathy symptoms. Pain. 1990; 42:135–144.
68. Max MB, Lynch SA, Muir J, Shoaf SE, Smoller B, Dubner R. Effects of desipramine, amitriptyline, and fluoxetine on pain in diabetic neuropathy. N Engl J Med. 1992; 326:1250–1256.
69. Sindrup SH, Bjerre U, Dejgaard A, Brøsen K, Aaes-Jørgensen T, Gram LF. The selective serotonin reuptake inhibitor citalopram relieves the symptoms of diabetic neuropathy. Clin Pharmacol Ther. 1992; 52:547–552.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download