INTRODUCTION
METHODS
Tissue preparation
Contraction measurements
Western blot analysis
Chemicals and antibodies
Statistics
RESULTS
Effect of cardamonin on contractions of endothelium-denuded aortas induced by the full RhoA/Rho-kinase activators fluoride and phenylephrine
 | Fig. 2Effect of cardamonin on fluoride-induced vascular contraction in denuded (A) or intact (B) muscles.Each ring was equilibrated in organ bath solution for 30~60 min before relaxation responses to cardamonin were measured. Data are expressed as the means of 3~5 experiments with vertical lines representing SEMs. *p<0.05, **p<0.01, presence versus absence of cardamonin.
|
 | Fig. 3Effect of cardamonin on phenylephrine-induced vascular contraction.Each ring was equilibrated in organ bath solution for 30~60 min before relaxation responses to cardamonin were measured. Data are expressed as the means of 3~5 experiments with vertical lines representing SEMs. *p<0.05, **p<0.01, presence versus absence of cardamonin.
|
Effect of cardamonin on contractions of denuded aortas induced by a phorbol ester as a MEK activator
 | Fig. 4Effect of cardamonin on phorbol ester-induced vascular contraction.Each ring was equilibrated in organ bath solution for 30~60 min before relaxation responses to cardamonin were measured. Data are expressed as the means of 3~5 experiments with vertical lines representing SEMs. *p<0.05, **p<0.01, presence versus absence of cardamonin.
|
Effect of cardamonin on the level of MYPT1 phosphorylation at Thr-855
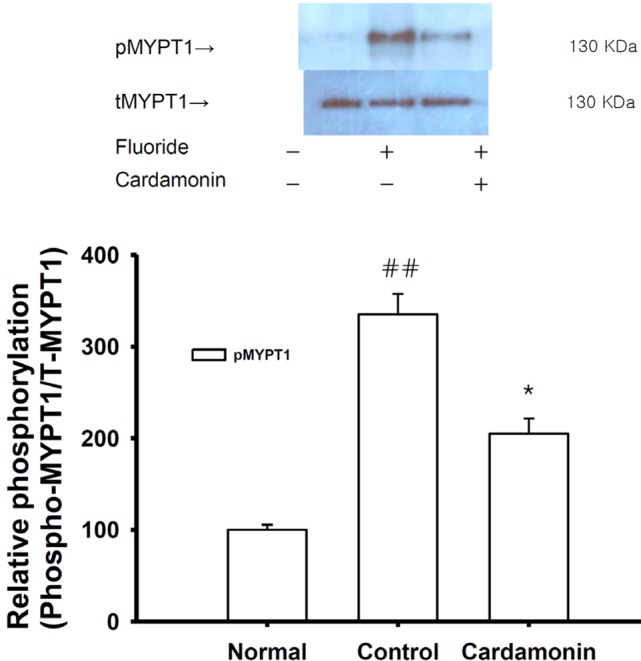 | Fig. 5Effect of cardamonin on fluoride-induced increases in phospho-MYP T1 levels.Phospho-MYPT1 protein levels were significantly decreased in rapidly-frozen cardamonin-treated rat aorta in the absence of endothelium compared to vehicle-treated rat aorta precontracted with fluoride. The upper panel shows a typical blot and the lower panel shows average densitometry results for relative levels of phospho-MYPT1. Data are expressed as the means of 3~5 experiments with vertical lines representing SEMs. *p<0.05, ##p<0.01, versus control or normal group respectively. Carda: 0.1 mM cardamonin; Fluoride: 6 mM sodium fluoride.
|
Effect of cardamonin on levels of ERK1/2 phosphorylation at Thr-202/Tyr-204
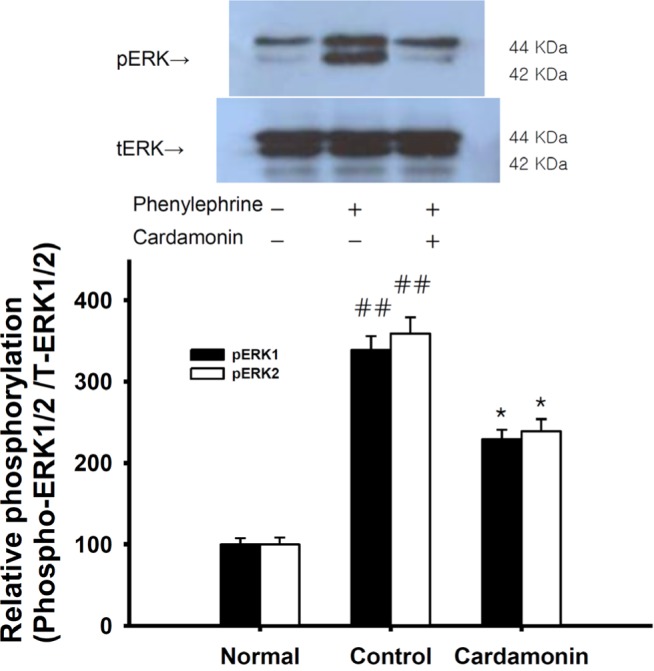 | Fig. 6Effect of cardamonin on the phenylephrine-induced increase in phospho-ERK1/2 level.Phospho-ERK1/2 protein level was decreased in rapidly-frozen cardamonin-treated rat aortas in the absence of endothelium than vehicle-treated rat aortas precontracted with a phorbol ester. The upper panel shows a typical blot and the lower panel shows average densitometry results for relative levels of phospho-ERK1/2. Data are expressed as the means of 3-5 experiments with vertical lines representing SEMs. ##p<0.01, versus normal group respectively. Cardamonin: 0.1 mM cardamonin; Phenylephrine: 1 µM phenylephrine.
|
Effect of cardamonin on the level of LC20 phosphorylation
 | Fig. 7Effect of cardamonin on the fluoride-induced increase in phospho-MLC20 level.Phospho-MLC20 levels expressed as a percentage of total MLC20 were significantly lower in rapidly-frozen cardamonin-treated rat aorta in the absence of endothelium than vehicle-treated rat aorta precontracted with fluoride (6 mM). Data are expressed as the means of 3~5 experiments with vertical lines representing SEMs. *p<0.05, ##p<0.01, versus control or normal group respectively. Cardamonin: 0.1 mM cardamonin; Fluoride: 6 mM sodium fluoride.
|




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download