Abstract
Gastrointestinal motility consists of phasic slow-wave contractions and the migrating motor complex (MMC). Eupatilin (Stillen®) has been widely used to treat gastritis and peptic ulcers, and various cytokines and neuropeptides are thought to be involved, which can affect gastrointestinal motility. We performed a study to identify the effects of eupatilin on lower gastrointestinal motility with electromechanical recordings of smooth muscles in the human ileum and colon. Ileum and colon samples were obtained from patients undergoing bowel resection. The tissues were immediately stored in oxygenated Krebs-Ringer's bicarbonate solution, and conventional microelectrode recordings from muscle cells and tension recordings from muscle strips and ileal or colonic segments were performed. Eupatilin was perfused into the tissue chamber, and changes in membrane potentials and contractions were measured. Hyperpolarization of resting membrane potential (RMP) was observed after administration of eupatilin. The amplitude, AUC, and frequency of tension recordings from circular and longitudinal smooth muscle strips and bowel segments of the ileum and colon were significantly decreased after admission of eupatilin. Eupatilin elicited dose-dependent decreases during segmental tension recordings. In conclusion, eupatilin (Stillen®) showed inhibitory effects on the human ileum and colon. We propose that this drug may be useful for treating diseases that increase bowel motility, but further studies are necessary.
Gastrointestinal motility consists of phasic slow-wave contractions and the migrating motor complex (MMC) in various animal models. Slow waves originate from electronic membrane potential changes in interstitial cells of Cajal (ICC) and generate spontaneous and rhythmic phasic contractions. The enteric nervous system affects the generation and migration of the MMC, which consists of periodic mass movements. The phasic contractions act to mix materials in the intestine and the MMC advances material from the oral to the anal end of the intestine [1,2,3]. Human gastrointestinal motility is thought to be similar, but more complex. For example, three-dimensional networks of ICCs or ICCs in line with the septa (ICC-SEP) might generate effective contractions of thick smooth muscles [4,5,6,7].
Disorders related to gastrointestinal motility might be caused by abnormalities of ICCs or of the enteric nervous system. There have been many efforts to develop a specific drug to treat diseases of increasing or decreasing gastrointestinal motility [8]. Irritable bowel syndrome (IBS) is the most common functional bowel disease, which frequently presents with abnormal bowel motility. Lifestyle modification is still the recommended treatment because effective drugs to improve dysmotility and relieve symptoms are not widely available [9,10,11].
Eupatilin (Stillen®) is an ethanol extract from an oriental herb, Artemisiaeasiatica Nakai. Eupatilin has already been widely used to treat gastritis and peptic ulcers. Eupatilin has anti-inflammatory and anti-oxidative cytoprotective effects against gastric mucosal damage and enhances regeneration of damaged mucosa [12,13]. Various cytokines and neuropeptides that can affect gastrointestinal motility are thought to be involved in the mechanism of action [14,15,16]. However, there have been no reports about the effects of eupatilin on human gastrointestinal motility.
We performed this study to identify the effects of eupatilin on human lower gastrointestinal motility using in vitro techniques, including electromechanical recordings of smooth muscles excised from the human ileum and colon.
Human ileum and colon samples were obtained immediately from operations for non-obstructive bowel diseases. There was no greater resection of the bowel than medically necessary because the amount of tissue needed for the experiments was very small. This study was approved by the Institutional Review Board of the Clinical Research Institute of the Seoul National University Hospital (IRB approval number: H-0603-071-170).
After resection of the bowel, 4×2 cm ileal or colonic segments were removed from the resected sections. Specimens were immediately placed into oxygenated Krebs-Ringer solution (KRB). The KRB contained (in mM) 120.4 NaCl, 5.9 KCl, 15.5 NaHCO3, 11.5 glucose, 1.2 MgCl2, 1.2 NaH2PO4 and 2.5 CaCl2. This solution had a pH of 7.3~7.4 at 37.5℃ when bubbled to equilibrium with 97% O2 and 3% CO2.
Tissues were transferred to a Petri dish coated with Sylgard (Dow corning Co., USA) and pinned downed in a dissecting dish. The muscles were cut parallel to the longitudinal muscle (LM) fibers with a knife consisting of a pair of sharp parallel scalpel blades set 1.5 mm apart and turned on side to expose a cross section of all of the muscle layers in the electrophysiological chamber. The chamber was constantly perfused with prewarmed, preoxygenated KRB solution. The temperature was maintained at 37.5±0.5℃. The muscles were equilibrated for at least 2 hours before experiments began. Conventional microelectrode recordings were performed using sharp microelectrodes filled with 3 M KCl [6]. Membrane potentials were measured with a high-input resistance electrometer and outputs were displayed on an oscilloscope. Outputs were measured using pClamp software R (version 9.0. Axon Instruments, Foster City, CA, USA) and Origin Software (MicroCal Software, Northampton, MA, USA) programs. Resting membrane potential (RMP) (mV), wave and spike amplitude (mV), and the frequency (/min) of the slow waves were analyzed (Fig. 1A).
Ileal and colonic tissues were pinned down on a Petri dish coated with Sylgard with the mucosal side facing upward. The mucosal and submucosal layers were gently removed with a pair of scissors. Longitudinal and circular muscle (CM) bundles were obtained by sharp dissection. The size of the muscle strips were 2 mm in width and 1 cm in length.
For contractile activity recordings, the muscle strips were attached with a suture to an isometric strain gauge (World precision Instruments, Sarasota, FL, USA) in a tissue chamber perfused with pre-warmed, preoxygenated KRB solution. One end of the muscle strips was anchored to an isometric strain gauge and the other end was fixed to a steel bar [6]. The temperature was maintained at 37.5±0.5℃. The muscle strips were equilibrated for at least 2 hours before experiments began, and a resting force of 9.8 mN (1 g) was applied. The mechanical signals were digitized and recorded by Acknowledge software (Biopac Systems, Inc, Goleta, CA) for data analysis. Frequency (/min), amplitude (mN) and the area under the curve (AUC, sec×mN/min) of contraction waves were analyzed. In the muscle strip recordings, the area under the curve was defined as the integrated area under a single wave (Fig. 1B).
Ileal or colonic segments, with the mucosa and submucosa intact, were prepared by cutting the whole layer of a segment parallel to longitudinal muscle. The size of the segments was 4 cm in length and 2 cm in width. A stainless steel rod was place parallel to the longitudinal muscle in the organ bath. CM tension was recorded at three (proximal, middle, distal) sites and LM tension was recorded by perpendicular traction using sutures placed at each site. Ileal or colonic segments were suspended in a tissue chamber perfused with pre-warmed, preoxygenated KRB solution. A stainless steel rod (10 cm long, 3 mm in diameter) was placed parallel to the longitudinal muscle. CM tension was recorded at three sites; proximal, mid, and distal sites, each 1cm apart. Stainless steel spring clips (7×18 mm) were attached to both ends of the sutured muscle. Each clip was attached to an isometric force transducer. Another clip was attached to the distal end of the colon flap, which effectively recorded the distal LM tension [6]. Temperature was maintained at 37.5±0.5℃. The muscles were equilibrated for at least 2 hours before experiments began. A resting force of 9.8 mN (1 g) was applied to each measuring site. The tension of each measuring sites was measured using an isometric strain gauge. Frequency (/min), amplitude (mN), and AUC of contraction waves were measured by Acknowledge software. The propagation patterns were evaluated by analyzing sequences of contractions detected at each site during recording. The patterns of propagation were classified as antegrade, which describes propagation from the proximal to distal site, and retrograde, which describes propagation from the distal to proximal site. A mixed pattern of propagation was also defined as contractions without consistent sequences. For evaluating the propagations in a recording of the ileal segment, we analyzed 10 propagations of waves (Fig. 1C).
After regular waves were detected in each of the experiments, eupatilin which is the extract from Stillen® (Dong-A Pharmaceutical Co. Korea) was perfused into tissue chamber. The eupatilin is a single compound with 344.31 of molecular weight. We calculated the concentrations of eupatilin by the molecular weight, and investigated the changes of the smooth muscle contractile activities at various concentrations (1, 2.5, 5 µM). We analyzed 10 waves before and after administration of eupatilin at each level.
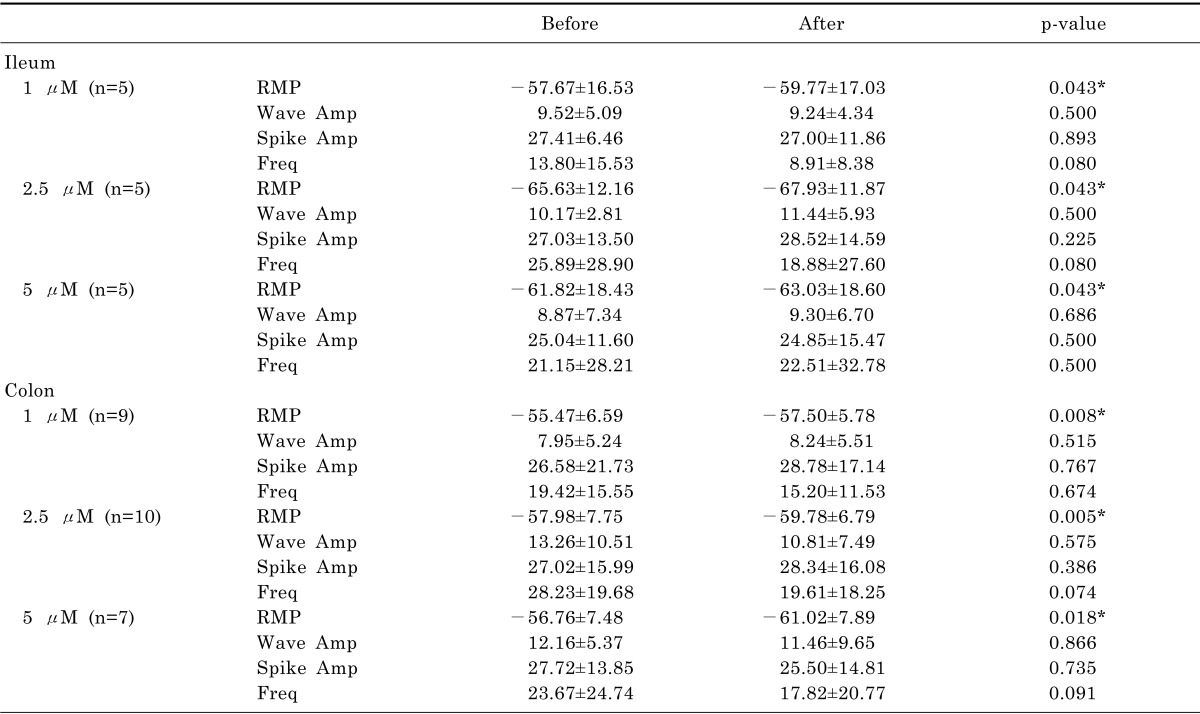
Slow waves in ileum and colon tissues were detected by intracellular recording (Fig. 2). Administration of eupatilin significantly hyperpolarized the RMP at ileal smooth muscle cell at 1 µM (-57.67±16.53 vs -59.77±17.03, p=0.043). The hyperpolarization of RMP was also observed after administration of 2.5 (-65.63±12.16 vs -67.93±11.87, p=0.043) and 5 µM (-61.82±18.43 vs -63.03±18.60, p=0.043). However, the wave amplitude, spike amplitude and frequency were not significantly changed after administration of eupatilin, and electrical activity remained present. Eupatilin induced hyperpolarization of the RMP of colonic smooth muscle cell at 1 µM (-55.47±6.59 vs -57.50±5.78, p=0.008). The hyperpolarization of RMP was also induced at 2.5 (-57.98±7.75 vs -59.78±6.79, p=0.005) and 5 µM (-56.76±7.48 vs -61.02±7.89, p=0.018). The wave amplitude, spike amplitude and frequency were not changed. Detailed analyses of intracellular recordings of membrane potential are described in Table 1.
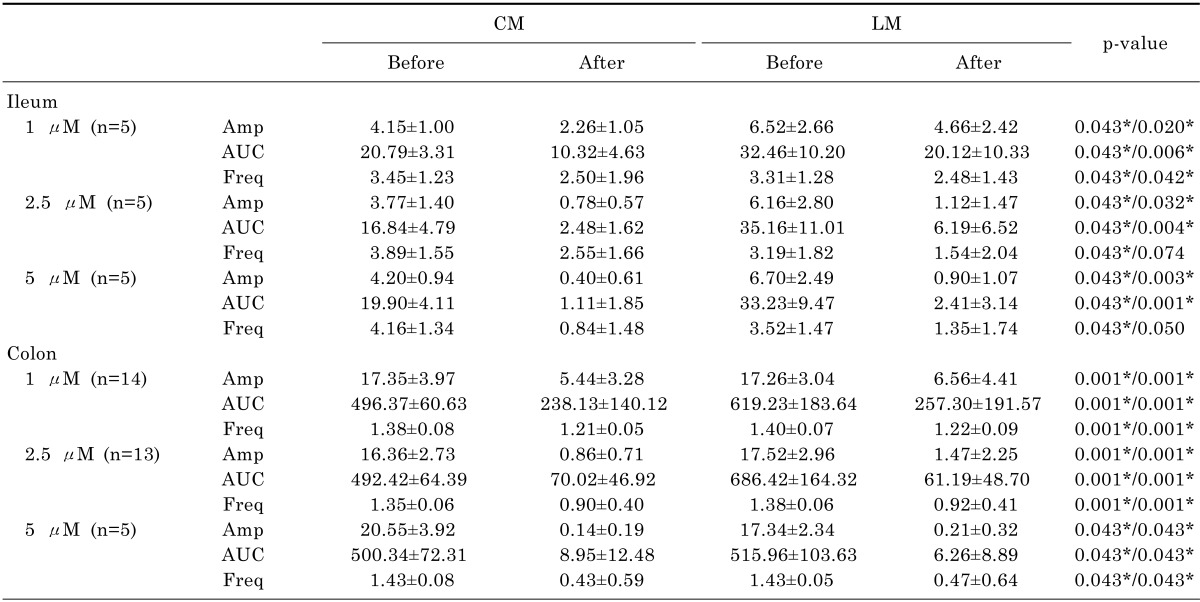
Contractile waves were detected by smooth muscle strip tension recordings in CM and LM. The contractions of smooth muscle strips were reduced after administration of eupatilin in ileum and colon samples (Fig. 3). The amplitude was significantly decreased after administration of 1 µM of eupatilin in ileal CM (4.15±1.00 vs 2.26±1.05, p=0.043) and LM (6.52±2.66 vs 4.66±2.42, p=0.020). The AUC was also decreased at 1 µM of eupatilin in ileal CM (20.79±3.31 vs 10.32±4.63, p=0.043) and LM (32.46±10.20 vs 20.12±10.33, p=0.006). The frequency was decreased at 1 µM of eupatilin in ileal CM (3.45±1.23 vs 2.50±1.96, p=0.043) and LM (3.31±1.28 vs 2.48±1.43, p=0.042). The amplitude, AUC and frequency were also decreased significantly after administration of 2.5 and 5 µM of eupatilin in ileal CM and LM (p<0.050). Eupatilin significantly reduced the amplitude of colonic CM (17.35±3.97 vs 5.44±3.28, p=0.001) and LM (17.26±3.04 vs 6.56±4.41, p=0.001) at 1 µM. The AUC was also decreased at 1 µM of eupatilin in colonic CM (496.37±60.63 vs 238.13±140.12, p=0.001) and LM (619.23±183.64 vs 257.30±191.57, p=0.001). The frequency was decreased at 1 µM of eupatilin in colonic CM (1.38±0.08 vs 1.21±0.05, p=0.001) and LM (1.40±0.07 vs 1.22±0.09, p=0.001). The amplitude, AUC and frequency were also decreased significantly after administration of 2.5 and 5 µM of eupatilin in colonic CM and LM (p<0.050). Detailed analyses of the smooth muscle strip tension recordings are described in Table 2.
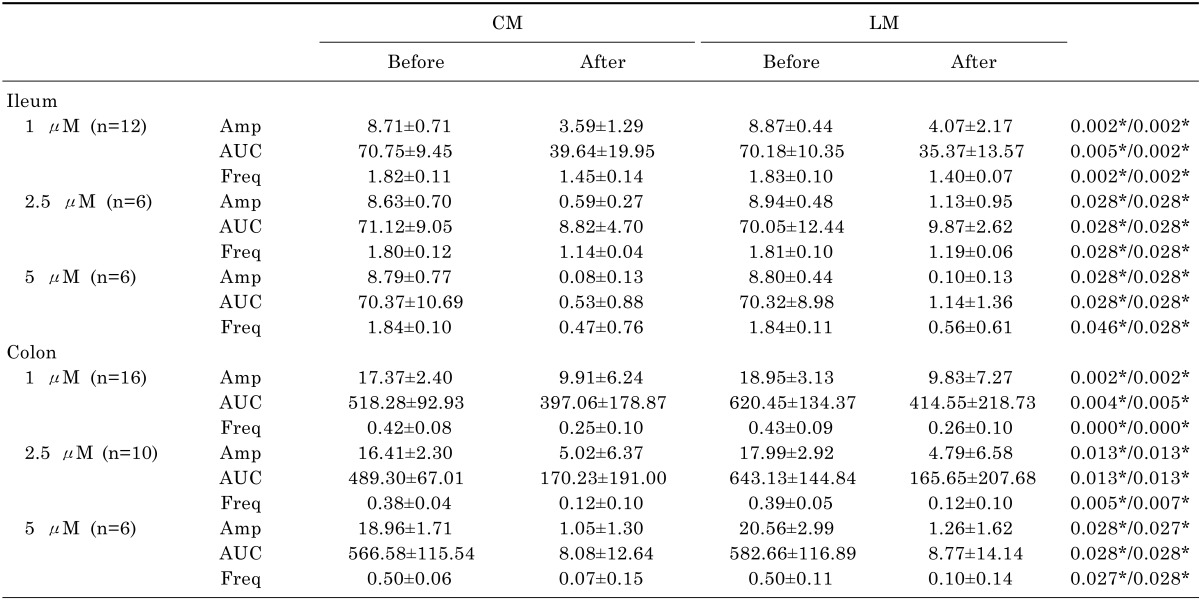
Contractile waves were detected in bowel segments using tension recordings at proximal, middle, and distal sites of CM and LM. The contractions of bowel segments were reduced after administration of eupatilin in both the ileum and the colon (Fig. 4). The amplitude was significantly decreased after administration of 1 µM of eupatilin in CM (8.71±0.71 vs 3.59±1.29, p=0.002) and LM of ileal segment (8.87±0.44 vs 4.07±2.17, p=0.002). The AUC was decreased at 1 µM of eupatilin in CM (70.75±9.45 vs 39.64±19.95, p=0.005) and LM of ileal segment (70.18±10.35 vs 35.37±13.57, p=0.002). The frequency was also decreased after administration of 1 µM of eupatilin in CM (1.82±0.11 vs 1.45±0.14, p=0.002) and LM of ileal segment (1.83±0.10 vs 1.40±0.07, p=0.002). The amplitude, AUC and frequency were decreased significantly after administration of 2.5 and 5 µM of eupatilin in CM and LM of ileal segment (p<0.050). Eupatilin reduced the amplitude of CM (17.37±2.40 vs 9.91±6.24, p=0.002) and LM of colonic segment (18.95±3.13 vs 9.83±7.27, p=0.002) at 1 µM. The AUC was also decreased at 1 µM of eupatilin in CM (518.28±92.93 vs 397.06±178.87, p=0.004) and LM of colonic segment (620.45±134.37 vs 414.55±218.73, p=0.005). The frequency was decreased at 1 µM of eupatilin in CM (0.42±0.08 vs 0.25±0.10, p=0.001) and LM of colonic segment (0.43±0.09 vs 0.26±0.10, p=0.001). The amplitude, AUC and frequency were also decreased significantly after administration of 2.5 and 5 µM of eupatilin in CM and LM of colonic segment (p<0.050). Detailed analyses of bowel segment tension recordings are also described in Table 3. Eupatilin induced a dose-dependent decrease of the amplitude, AUC and frequency at ileal and colonic segment (Fig. 5).
In ileal segment, antegrade propagation was observed 4.00±1.00, and retrograde propagation was 3.20±0.45 times per 10 waves. Mixed patterns were identified 2.80±1.30 times per 10 waves. After administration of eupatilin, antegrade propagation was 3.40±1.14, and retrograde propagation was observed 4.00±1.22 times per 10 waves. Mixed patterns were 2.60±1.52 per 10 waves, and there were no differences of propagation patterns after administration of eupatilin (n=5, p>0.050). In colonic segment, antegrade propagation was identified 6.00±0.71, and retrograde propagation was 1.40±0.55 times per 10 waves. Mixed patterns were observed 2.60±0.55 times per 10 waves. After administration of eupatilin, antegrade propagation was 5.20±0.84, and retrograde propagation was observed 1.60±0.55 times per 10 waves. Mixed patterns were 3.20±0.45 per 10 waves, and there were no differences of propagation patterns after administration of eupatilin (n=5, p>0.050).
Here, we report that eupatilin (Stillen®) decreases human lower gastrointestinal motility. The RMP was hyperpolarized after administration of eupatilin in intracellular electrophysiologic recordings of ileum and colon samples. Eupatilin inhibited excitability by stabilizing the membrane potential and affected contractions of gastrointestinal smooth muscle. Tension recordings of smooth muscle strips and ileal or colonic segments also showed decreased contractility. Eupatilin has long been used to treat nonspecific gastric symptoms such as abdominal pain or diarrhea in oriental medicine. It has been reported to be protective against gastritis or peptic ulcers in experimental or clinical studies, and has been widely used as a tablet drug, Stillen®. The mucoprotective effects of eupatilin are mediated by increased mucus and prostaglandin (PG) secretion from the gastric mucosa and a the cytoprotective action is mediated by nitric oxide (NO) [17]. Eupatilin also has been reported to reduce the activation of human neutrophils induced by Helicobacter pylori [18] and suppress tumor proliferation by inducing cell cycle arrest [19,20,21]. However, the effects of eupatilin on gastrointestinal motility have not been previously studied, although there was a study showing non-toxic effects on the contractions of guinea pig ileum tissue [14].
PGs are autocrine or paracrine lipid mediators produced by cyclooxygenase or lipooxygenase in fatty acid metabolism, and have various actions on smooth muscle. PGF2a and PGI2 increase gastrointestinal smooth muscle contractions by increasing the release of Ach from cholinergic nerve terminals. PGE2 inhibits smooth muscle contractions through specific receptors, such as EP2 or EP4 [22,23]. Eupatilin is known to increase the release of PGE2 [13], which might be one of the reasons for the observed relaxation of gastrointestinal smooth muscle contractions. However, some studies reported that PGE2 depolarized membrane potential and enhanced excitability by increasing acetylcholine release [24]. NO is also involved in the cytoprotective effects of eupatilin on gastric mucus cells [15]. NO contributes to the maintenance of mucosal integrity, which might be mediated by vasodilatation and increased blood flow, or by inhibition of neutrophil-platelet interactions that lead to the formation of thrombosis and mucosal hemorrhages. Excessive NO could be cytotoxic, leading to uncontrolled vasodilatation, hypotension, damage to microvascular integrity of mucosa and increased free radicals [25]. NO has restrictive effects on gastrointestinal smooth muscle contractions by reducing noncholinergic and nonadrenergic neurotransmitter release and altering calcium concentrations in the ICC and smooth muscle cells [26]. PG or NO released from the mucosa might be responsible to decrease gastrointestinal motility in this study. But the reduction of contractions was observed in tension recordings of smooth muscle strips, in which mucosa and submucosa were removed. Another mechanism for direct action to smooth muscle cells can be involved in inhibition of contractions. Further studies are necessary to identify the relationships between of Stillen® or eupatilin, neuropeptides, and their effects on gastrointestinal motility.
IBS is the most common gastrointestinal disorder, characterized by abdominal discomfort with altered bowel function and increasing or decreasing intestinal motility. IBS are classified with 4 subtypes by predominant stool pattern, whether it is constipation or diarrhea [9]. It has been reported that IBS with constipation was associated with prolonged gut transit times and IBS with diarrhea was also related to intestinal dysmotility [27]. Although many therapies, such as increased dietary fiber, antidiarrheals, antispasmodics, probiotics, or antidepressants, have been used to relieve IBS symptoms, there have not been strong evidence of long-term efficacy [28,29]. As altered levels of 5-Hydroxytryptamine (HT) are related to bowel motility disorders, some effective drugs that act on 5-HT receptors have been introduced, and alosetron is a 5-HT3 receptor antagonist that can improve the symptoms of IBS with diarrhea [30,31]. It inhibited MMC in murine small and large bowels in an in vitro study [11]. Although many 5-HT receptor related drugs have been withdrawn due to their side effects, alosetron has current Food and Drug Administration (FDA) approval. We found that Stillen® has inhibitory effects on human lower gastrointestinal motility, and it may improve symptoms related to increased bowel motility, and could also be used to treat IBS with diarrhea.
This study has some limitations. First, we used normal human bowel tissue, but experiments on bowels with IBS might allow us to better to verify the effects of Stillen® in disease. However, it is not easy to obtain IBS tissues because IBS is not treated by surgery. Second, the effects of Stillen® are not exactly those of eupatilin, so further experiments using eupatilin are necessary to explain a detailed mechanism of action. Third, as this was an in vitro study, further clinical trials for in patients should be performed to confirm the effects of the drug in IBS.
In conclusion, eupatilin (Stillen®) showed inhibitory effects on the human ileum and colon measured by intracellular recordings and smooth muscle and bowel segment tension recordings. We suggest that eupatilin (Stillen®) could be used for treating diseases with increased bowel motility, but further studies are necessary.
ACKNOWLEDGEMENTS
Seung-Bum Ryoo and Heung-Kwon Oh were equally contributed this study. And this study was supported by a grant from the Clinical Research Institute to Seoul National University Hospital provided by the Dong-A, Pharmaceutical Research Institute, Yongin, Korea. (Grant No: 0620100430). The statistical analysis was made by the help of the Medical Research Collaborating Center (MRCC) at Seoul National University Hospital Biomedical Research Institute.
References
1. Sanders KM. A case for interstitial cells of Cajal as pacemakers and mediators of neurotransmission in the gastrointestinal tract. Gastroenterology. 1996; 111:492–515. PMID: 8690216.

2. Fida R, Lyster DJ, Bywater RA, Taylor GS. Colonic migrating motor complexes (CMMCs) in the isolated mouse colon. Neurogastroenterol Motil. 1997; 9:99–107. PMID: 9198085.

3. Park KJ, Hennig GW, Lee HT, Spencer NJ, Ward SM, Smith TK, Sanders KM. Spatial and temporal mapping of pacemaker activity in interstitial cells of Cajal in mouse ileum in situ. Am J Physiol Cell Physiol. 2006; 290:C1411–C1427. PMID: 16381798.

4. Lee HT, Hennig GW, Fleming NW, Keef KD, Spencer NJ, Ward SM, Sanders KM, Smith TK. The mechanism and spread of pacemaker activity through myenteric interstitial cells of Cajal in human small intestine. Gastroenterology. 2007; 132:1852–1865. PMID: 17484879.

5. Lee HT, Hennig GW, Fleming NW, Keef KD, Spencer NJ, Ward SM, Sanders KM, Smith TK. Septal interstitial cells of Cajal conduct pacemaker activity to excite muscle bundles in human jejunum. Gastroenterology. 2007; 133:907–917. PMID: 17678922.

6. Choe EK, Moon JS, Moon SB, So IS, Park KJ. Electromechanical characteristics of the human colon in vitro: is there any difference between the right and left colon? Int J Colorectal Dis. 2010; 25:1117–1126. PMID: 20544209.

7. Kim BJ, Park KJ, Kim HW, Choi S, Jun JY, Chang IY, Jeon JH, So I, Kim SJ. Identification of TRPM7 channels in human intestinal interstitial cells of Cajal. World J Gastroenterol. 2009; 15:5799–5804. PMID: 19998500.
8. Huizinga JD, Thuneberg L, Vanderwinden JM, Rumessen JJ. Interstitial cells of Cajal as targets for pharmacological intervention in gastrointestinal motor disorders. Trends Pharmacol Sci. 1997; 18:393–403. PMID: 9357324.

9. Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional bowel disorders. Gastroenterology. 2006; 130:1480–1491. PMID: 16678561.

10. Kellow JE, Phillips SF. Altered small bowel motility in irritable bowel syndrome is correlated with symptoms. Gastroenterology. 1987; 92:1885–1893. PMID: 3569764.

11. Bush TG, Spencer NJ, Watters N, Sanders KM, Smith TK. Effects of alosetron on spontaneous migrating motor complexes in murine small and large bowel in vitro. Am J Physiol Gastrointest Liver Physiol. 2001; 281:G974–G983. PMID: 11557518.

12. Seol SY, Kim MH, Rew JS, Choi MG. A phase III clinical trial of stillen(TM) for erosive gastritis. Korean J Gastrointest Endosc. 2004; 28:230–236.
13. Oh TY, Lee JS, Ahn BO, Cho H, Kim WB, Kim YB, Surh YJ, Cho SW, Lee KM, Hahm KB. Oxidative stress is more important than acid in the pathogenesis of reflux oesophagitis in rats. Gut. 2001; 49:364–371. PMID: 11511558.

14. Lee EB, Cheon SA, Lee ES, Kim KK, Ko ST, Yu KJ, Shin DS, Kang SY, Kim SH, Sohn MH. General pharmacology of artemisia extract powder, DA-9601. J Appl Pharmacol. 1996; 4:174–183.
15. Oh TY, Ahn BO, Ko JI, Ryu BK, Son MW, Kim SH, Kim WB, Lee EB. Studies on Protective effect of da-9601, an artemisiae herba extract, against ethanol-induced gastric mucosal damage and its mechanism. J Appl Pharmacol. 1997; 5:202–210.
16. Oh TY, Ryu BK, Ko JI, Ahn BO, Kim SH, Kim WB, Lee EB, Jin JH, Hahm KB. Protective effect of DA-9601, an extract ofArtemisiae Herba, against naproxen-induced gastric damage in arthritic rats. Arch Pharm Res. 1997; 20:414–419. PMID: 18982482.

17. Ahn BO, Ryu BK, Ko JI, Oh TY, Kim SH, Kim WB, Yang J, Lee eB, Hahm KB. Beneficial effect of DA-9601, an extract of artemisiae herba, on animals models of inflammatory bowel disease. J Appl Pharmacol. 1997; 5:165–173.
18. Lee JJ, Han BG, Kim MN, Chung MH. The inhibitory effect of eupatilin on Helicobacter pylori-induced release of leukotriene D4 in the human neutrophils and gastric mucosal cells. Korean J Physiol Pharmacol. 1997; 1:573–580.
19. Seo HJ, Park KK, Han SS, Chung WY, Son MW, Kim WB, Surh YJ. Inhibitory effects of the standardized extract (DA-9601) of Artemisia asiatica Nakai on phorbol ester-induced ornithine decarboxylase activity, papilloma formation, cyclooxygenase-2 expression, inducible nitric oxide synthase expression and nuclear transcription factor kappa B activation in mouse skin. Int J Cancer. 2002; 100:456–462. PMID: 12115530.
20. Kim DH, Na HK, Oh TY, Kim WB, Surh YJ. Eupatilin, a pharmacologically active flavone derived from Artemisia plants, induces cell cycle arrest in ras-transformed human mammary epithelial cells. Biochem Pharmacol. 2004; 68:1081–1087. PMID: 15313404.

21. Kim MJ, Kim DH, Na HK, Oh TY, Shin CY, Surh Ph D Professor YJ. Eupatilin, a pharmacologically active flavone derived from Artemisia plants, induces apoptosis in human gastric cancer (AGS) cells. J Environ Pathol Toxicol Oncol. 2005; 24:261–269. PMID: 16393120.

22. Fukunaga Y, Mine Y, Yoshikawa S, Takeuchi T, Hata F, Yagasaki O. Role of prostacyclin in acetylcholine release from myenteric plexus of guinea-pig ileum. Eur J Pharmacol. 1993; 233:237–242. PMID: 8467869.

23. Xiao ZL, Biancani P, Behar J. Effects of progesterone on motility and prostaglandin levels in the distal guinea pig colon. Am J Physiol Gastrointest Liver Physiol. 2009; 297:G886–G893. PMID: 20501437.

24. Manning BP, Sharkey KA, Mawe GM. Effects of PGE2 in guinea pig colonic myenteric ganglia. Am J Physiol Gastrointest Liver Physiol. 2002; 283:G1388–G1397. PMID: 12388206.

25. Konturek SK, Konturek PC. Role of nitric oxide in the digestive system. Digestion. 1995; 56:1–13. PMID: 7895925.

26. Boeckxstaens GE, Pelckmans PA, Bult H, De Man JG, Herman AG, Van Maercke YM. Non-adrenergic non-cholinergic relaxation mediated by nitric oxide in the canine ileocolonic junction. Eur J Pharmacol. 1990; 190:239–246. PMID: 1981752.

27. DuPont AW, Jiang ZD, Harold SA, Snyder N, Galler GW, Garcia-Torres F, DuPont HL. Motility abnormalities in irritable bowel syndrome. Digestion. 2014; 89:119–123. PMID: 24503633.

28. American College of Gastroenterology Task Force on Irritable Bowel Syndrome, Brandt LJ, Chey WD, Foxx-Orenstein AE, Schiller LR, Schoenfeld PS, Spiegel BM, Talley NJ, Quigley EM. An evidence-based position statement on the management of irritable bowel syndrome. Am J Gastroenterol. 2009; 104(Suppl 1):S1–35. PMID: 19521341.
29. Chey WY, Jin HO, Lee MH, Sun SW, Lee KY. Colonic motility abnormality in patients with irritable bowel syndrome exhibiting abdominal pain and diarrhea. Am J Gastroenterol. 2001; 96:1499–1506. PMID: 11374689.

30. Atkinson W, Lockhart S, Whorwell PJ, Keevil B, Houghton LA. Altered 5-hydroxytryptamine signaling in patients with constipation-and diarrhea-predominant irritable bowel syndrome. Gastroenterology. 2006; 130:34–43. PMID: 16401466.
31. Ford AC, Brandt LJ, Young C, Chey WD, Foxx-Orenstein AE, Moayyedi P. Efficacy of 5-HT3 antagonists and 5-HT4 agonists in irritable bowel syndrome: systematic review and meta-analysis. Am J Gastroenterol. 2009; 104:1831–1843. quiz 1844. PMID: 19471254.

Fig. 1
Schematic drawing of experiments. (A) Conventional microelectrode recording. (a) resting membrane potential (RMP), (b) slow wave amplitude, (c) spike amplitude. (B) Isometric tension recordings from circular and longitudinal muscle strips. (a) amplitude, (b) interval to calculate frequency, (c) area under the curve (AUC). (C) Isometric tension recordings from circular and longitudinal muscles from ileal or colonic segments (M, mucosa; SM, submucosa; CM, circular muscle; LM, longitudinal muscle; prox, proximal; mid, middle; dist, distal).
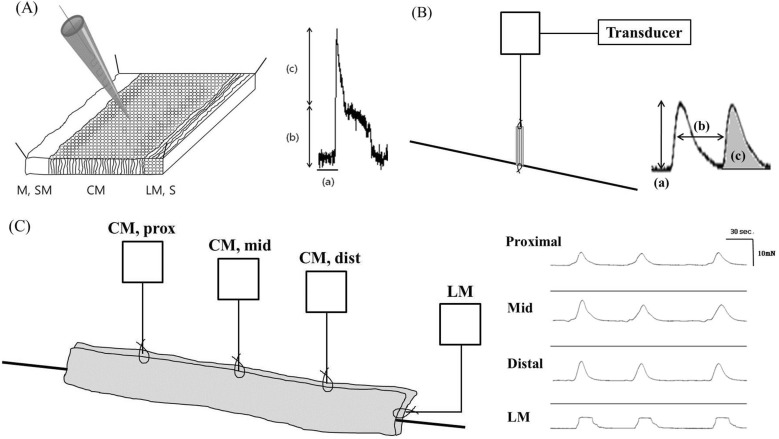
Fig. 2
The resting membrane potential was significantly hyperpolarized after administration of eupatilin in intracellular recordings of
the ileum (A) and colon (B). The horizontal black lines are the level of RMPs.
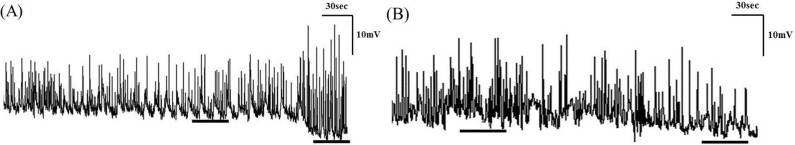
Fig. 3
The amplitude, AUC, and frequency were significantly decreased after administration of eupatilin in tension recordings of ileal (A) and colonic (B) smooth muscle strips.
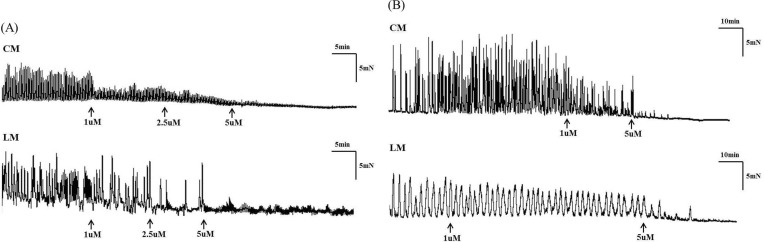
Fig. 4
The amplitude, AUC, and frequency were significantly decreased after administration of eupatilin in tension recordings of ileal (A) and colonic (B) segments.
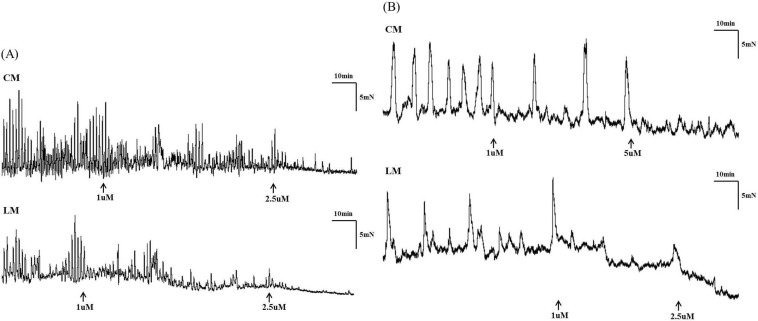
Fig. 5
Eupatilin induced a dose-dependent decrease in segmental tension recordings. The amplitude, AUC, and frequency of ileum (A) and colon (B) muscle contractions decreased as eupatilin increased from 0 to 1, 2.5 and 5 µM (▪ circular muscle, □ longitudinal muscle).
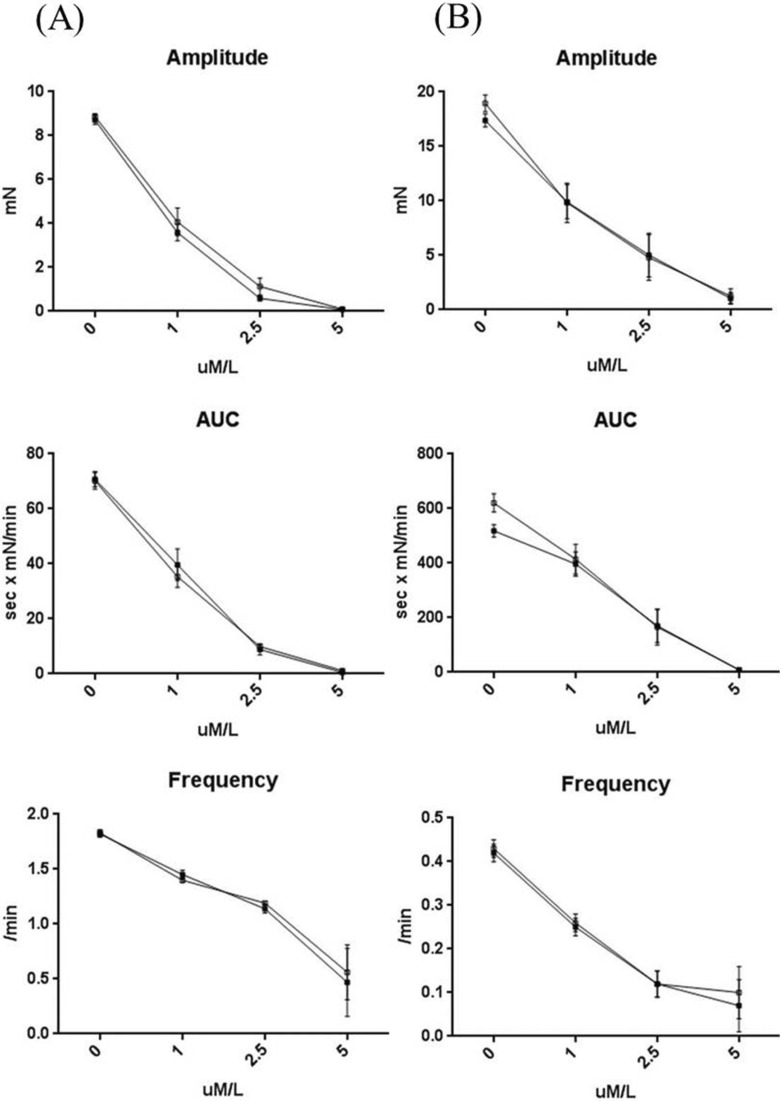
Table 1
Intracellular recordings of membrane potential changes before and after administration of eupatilin




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download