Abstract
The etiology of most psychiatric disorders is still incompletely understood. However, growing evidence suggests that stress is a potent environmental risk factor for depression and anxiety. In rodents, various stress paradigms have been developed, but psychosocial stress paradigms have received more attention than non-social stress paradigms because psychosocial stress is more prevalent in humans. Interestingly, some recent studies suggest that chronic psychosocial stress and social isolation affects mainly anxiety-related behaviors in mice. However, it is unclear whether chronic non-social stress induces both depression- and anxiety-related phenotypes or induces one specific phenotype in mice. In the present study, we examined the behavioral consequences of three chronic non-social stress paradigms: chronic predictable (restraint) stress (CPS), chronic unpredictable stress (CUS), and repeated corticosterone-HBC complex injection (RCI). Each of the three paradigms induced mild to severe depression/despair-like behaviors in mice and resulted in increased immobility in a tail suspension test. However, anxiety-related phenotypes, thigmotaxis and explorative behaviors, were not changed by the three paradigms. These results suggest that depression- and anxiety-related phenotypes can be dissociated in mouse stress models and that social and non-social stressors might affect brain circuits and behaviors differently.
Recent large-scale epidemiological studies have revealed that major depression and anxiety are the most common psychiatric disorders in humans [1,2]. Although the etiology of depression- and anxiety-related disorders is still incompletely understood, stress is a potent environmental risk factor for these disorders. Chronic exposure to stress induces modifications in brain physiology and neural circuits. Stress triggers the rapid activation of the hypothalamic-pituitary-adrenal (HPA) axis in animals, leading to the release of corticosteroids from the adrenal cortex [3,4,5]. Corticosteroids pass the blood brain barrier and reach various brain regions via the circulation. Binding of corticosteroids to two related receptor molecules, the glucocorticoid receptor and the mineralocorticoid receptor, induces the following structural and functional changes in brain neurons: regression of dendritic processes, loss and shrinkage of dendritic spines, inhibition of neurogenesis, disturbance of synaptic plasticity, and neurodegeneration [6,7,8]. These deleterious effects of corticosteroids on neurons are believed to cause depression- and anxiety-related behavioral changes in animals. In agreement with these observations, the hyperactivity of the HPA axis, hypercortisolemia, or prolonged glucocorticoid administration is associated with depression- and anxiety-related disorders [9,10]. In addition, depression- and/or anxiety-like behaviors can be induced in rodents by various chronic stress paradigms in which animals are exposed to several weeks of repeated physical stressors or social stressors.
Interestingly, however, some chronic stress paradigms induce both anxiety and depression-like behaviors in animals, while others have been reported to increase one specific phenotype [11,12,13,14,15,16]. Considering the high comorbidity of depression and anxiety in humans, the coexistence of these disorders in rodents also seems possible. However, there is a possibility that the different behavioral outcomes among the previous studies were caused by different stress paradigms. This idea raises the question whether distinct types of stressors induce different behavioral changes. A few recent reports on social stress paradigms provide evidence supporting this hypothesis. For example, mice exposed to social defeat or chronic subordinate colony housing displayed increased anxiety behaviors without changes in depression-related behaviors [12,13,14]. In addition, social isolation also produced similar behavioral changes in mice [17]. These findings have led us to examine whether chronic stress without social factors selectively induces depression-like behaviors or both depression- and anxiety-like behaviors in mice. In the present study, we investigated the effects of three non-social stress paradigms on depression- and anxiety-related behaviors. Our study provides experimental evidence that social and non-social chronic stressors differentially affect depression and anxiety. Although psychosocial stress is more prevalent in humans, the dissociation of critical risk factors for depression and anxiety may provide clues to understand the neural mechanisms of psychiatric disorders.
Four-week-old male C57BL/6N mice were purchased from Orient Bio (Gyeonggi, Korea). Upon arrival, the animals were acclimated for 1 week in our specific pathogen-free barrier facility before the experiments began. During the rearing and behavioral experiments, mice were housed in groups of 3~4/cage and monitored daily for signs of bullying, hair loss, or fight wounds. Except for stress sessions, animals were allowed free access to food and water and were maintained on a 12-hour light-dark cycle (lights on 08:00 h) at 23±1℃ with a relative humidity of 50~60%. The maintenance of all animals and related experiments were performed according to institutional guidelines for the care and use of animals in research (SNU-120323-1).
To investigate the effect of chronic non-social stress on depression- and anxiety-like behaviors in mice, male littermates were assigned to experimental or control groups at 35~38 days after birth. Each experimental group was exposed to a different type of chronic non-social stress for 3 weeks, as illustrated in Fig. 1. All of the three stress paradigms have been reported to cause stress responses, including increased corticosterone levels, in rodents [18,19,20,21,22]. For chronic predictable stress (CPS), mice were placed in a well-ventilated 60-ml syringe for 2 hours once per day between 17:00 and 19:00. During the 21 restraint sessions, mice were able to move laterally, but not vertically. For chronic unpredictable stress (CUS), mice were exposed in a random order to a variety of chronic stressors, including a wet cage (12 h), light-dark cycle reversal (24 h), white noise (100 dB, 12 h), cold water swim (10℃, 1 h), restraint (2 h), cage shake (30 rpm, 12 h), and electric foot shocks (10 scrambled shocks with duration of 2 s over 120 min). Animals were subjected to one stressor daily for 21 days. For repeated corticosterone injection (RCI), corticosterone-HBC complex was purchased from Sigma-Aldrich (St. Louis, MO) and dissolved in physiological saline. Mice received subcutaneous injections of the corticosterone-HBC complex (20 mg/kg) once per day for 21 days. The control animals for the repeated corticosterone-HBC injections were administered vehicle. All solutions were sterilized through a 0.22-µm filter before injection.
Each animal was placed in the center of an open field apparatus with opaque walls (40×40×40 cm) in a dimly lit room. The behavior of each mouse was monitored for 30 minutes by video recording. The total distance traveled and time spent in the entire open field and in the center (20×20 cm) were calculated using video tracking software (Ethovision XT, Noldus, Netherlands). To avoid possible abnormal behaviors caused by the tail suspension test, the open field test was performed 1 day before the tail suspension test.
Mice were suspended by their tails from a steel bar using adhesive tape in a chamber with opaque walls. The distance between the floor of the chamber and the steel bar was approximately 40 cm. Mice that climbed onto their tail or fell off during the test were excluded from analysis. The total duration of immobility over the 6 min observation period was scored by an experimenter blinded to the experimental details.
Data were analyzed using IGOR Pro (WaveMetrics, OR, USA) and SPSS (Statistical Package for the Social Sciences, IBM, NY, USA) software. Statistical significance was determined by parametric two-tailed Student's t-tests and nonparametric Mann-Whitney-Wilcoxon tests. All data and error bars are expressed as the mean±s.e.m.
Because weight changes have been observed in rodent models of chronic stress [23,24,25,26], we measured the daily body weight of mice during the stress period. As shown in Fig. 2A, CPS significantly reduced body weight. Repeated restraint caused weight loss during the first week and inhibited body weight gain through the end of the stress paradigm. After 3 weeks, stressed animals gained 0.7% of body weight (22.12±0.30 g and 22.25±0.23 g at weeks 0 and 3, respectively; n=9), whereas the body weight of control mice increased 9.3% (22.05±0.19 g and 24.10±0.21 g at weeks 0 and 3, respectively; n=9). These results suggest a significant interaction between body weight gain and treatment (p<0.001, Student's t-test and Mann-Whitney-Wilcoxon test).
To investigate the relationship between the predictability of stress and the reduction of body weight, we next examined the effect of chronic unpredictable non-social stress on body weight (Fig. 2B). Similar to CPS, weight gain during the 3 weeks was greater in the control animals (15.2%; 21.0±0.66 g and 24.20±0.78 g at weeks 0 and 3, respectively; n=9) than in the stressed animals (6.2%; 20.63±0.68 g and 21.88±0.77 g at weeks 0 and 3, respectively; n=8). This result indicates that CUS reduces body weight gain in mice and that the predictability of stress is not important for its effect on body weight gain (CPS: 92.34±0.96% of control; CUS: 91.05±1.01% of control; p>0.1; Fig. 2D).
We next examined the effect of predictable non-physical stress on body weight. Mice that received RCI showed comparable body weight gain to control mice that received repeated injections of vehicle (Fig. 2C). The control mice (n=10, 19.69±0.52 g and 22.75±1.30 g at weeks 0 and 3, respectively) and the corticosterone-HBC complex-treated mice (n=11, 19.35±0.57 g and 22.16±0.85 g at weeks 0 and 3, respectively) gained 15.5% and 14.5% of their original body weight, respectively. This difference did not reach statistical significance (p>0.1). Therefore, in contrast with CPS or CUS, RCI had no effect on body weight gain (Fig. 2D).
To investigate depression-like behaviors, we assessed immobility/despair behaviors of mice using the tail-suspension test [27]. In this test, immobility is thought to reflect a state of despair and of giving up trying to escape from an uncomfortable situation.
Mice exposed to CPS displayed high levels of immobility in the tail-suspension test (Fig. 3A). During the 6-minute test, the total duration of immobility in the control group and in the stressed group was 82.1±18.7 s and 147.63±13.1 s, respectively (n=8 pairs, p<0.05; Fig. 3D). We next examined the effect of CUS on depression-like behaviors. Similar to repeated restraint, the mice exposed to CUS spent more time immobile compared to the control mice (control: n=9, 126.7±10.4 s; CUS: n=8, 197.3±15.2 s; p<0.01; Fig. 3B, 3D). Consistently, a minute-by-minute analysis of the time spent immobile revealed that the CUS group became immobile significantly sooner than the control mice (Fig. 3B). We further tested RCI, predictable non-physical stress, on depression-like behaviors. As shown in Fig. 3C and Fig. 3D, RCI significantly increased the amount of time mice remained immobile by 42.1% compared to the vehicle-treated control mice (control: n=10, 150.5±29.9 s; RCI: n=11, 213.9±26.4 s; p<0.05; Fig. 3C, 3D). Together, these results suggest that each of the three chronic non-social stress paradigms caused elevated levels of depression-like behaviors in the tail-suspension test, regardless of the predictability.
To test whether chronic non-social stress induces both depression- and anxiety-related phenotypes, we examined anxiety-related behaviors using the open field test [28,29]. In this test, the degree of thigmotaxis, the tendency to remain close to the walls, is considered an index of anxiety in mice [30]. Because anxious animals are less likely to explore new environments, they show enhanced thigmotaxis and reduced locomotor activity in the open field chamber. We analyzed thigmotaxis and locomotor activity by measuring the time spent in the peripheral areas and the distance moved in the open field chamber, respectively.
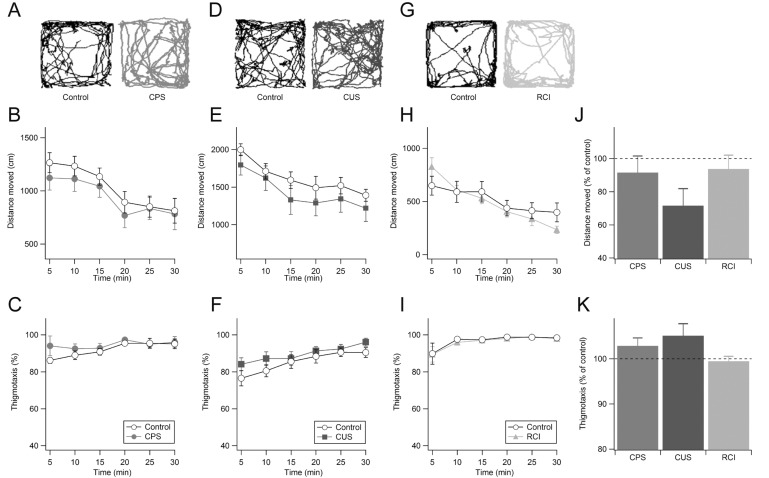
Mice exposed to CPS displayed normal locomotor activity and thigmotaxis levels compared to their control littermates during all test periods (Fig. 4A-C). In addition, when we analyzed the total distance moved (control: n=9, 61.90±4.96 m; CPS: n=9, 56.67±6.22 m; p>0.1; Fig. 4J) and the time spent in the peripheral area (1656.0±37.9 s vs. 1703.0±29.4 s; p>0.1; Fig. 4K) during the 30-minute test periods, we could not find any differences between the two groups. We next examined CUS on anxiety-related behaviors (Fig. 4D). Although mice exposed to CUS showed a tendency to be hypoactive (Fig. 4E) and enhanced thigmotaxis (Fig. 4F), the difference did not reach statistical significance during any test period. When we analyzed the whole test session, we also found no significant effects of CUS on total distance moved (control: n=9, 97.10±5.21 m; CUS: n=8, 88.47±9.03 m; p>0.1; Fig. 4J) and thigmotaxis (1536.0±43.2 s vs. 1614.7±40.6 s; p>0.1; Fig. 4K). Additionally, we examined the effects of RCI on anxiety-related behaviors (Fig. 4G). Again, we did not detect any significant differences in distance moved (Fig. 4H) and time spent in the peripheral areas (Fig. 4I) between the corticosterone- and vehicle-treated groups. As shown in Fig. 4J and Fig. 4K, both groups of mice showed similar levels of exploratory activity (control: n=10, 31.39±4.12 m; RCI: n=11, 29.42±2.62 m; p>0.1) and thigmotaxis (control: 1741.8±18.7 s; RCI: n=11, 1732.3±18.7 m; p>0.1) during the entire test session.
Taken together, these results indicate that each of the three chronic non-social stress paradigms had no effect on anxiety-related behaviors of mice.
Although stress is a risk factor for depression- and anxiety-related disorders, the differential effects of social and non-social stress on these neuropsychiatric disorders are poorly understood. In the present study, we examined the effect of three chronic non-social stress paradigms on mouse behavior. We found that CPS, CUS, and RCI induced depression/despair-like behaviors without affecting anxiety-related behaviors.
According to human studies [31,32,33,34], depression and anxiety show high comorbidity rates. Approximately half of patients with major depressive disorder met the criteria for generalized anxiety disorder, and the majority of patients with generalized anxiety disorder also have major depressive disorder [33,35]. These findings imply that brain circuits associated with depression partly overlap with those of anxiety disorder and that certain types of stress can modify brain circuits associated with both disorders [5,36]. Although the exact brain circuits that mediate depression and anxiety are still unclear, recent human imaging studies identified some brain areas dysregulated in depression and anxiety disorders, including the prefrontal cortex, subgenual cingulate cortex, hippocampus, and amygdala [37,38]. These brain areas are implicated in the regulation of stress responses as well as emotional functions [39]. In mammals, stress activates the HPA axis and results in the secretion of stress hormones (corticosteroids) from the adrenal cortex. Although the neural mechanism that translates stressful stimuli into the activation of the paraventricular nucleus (PVN) in the hypothalamus is still incompletely understood, the hippocampus appears to inhibit the PVN through the ventral subicular projection to PVN-connected GABAergic neurons in the bed nucleus of the stria terminalis (BNST) [5]. In contrast, the amygdala appears to activate hypothalamic areas through excitatory and double GABAergic projections to the PVN [4]. In accordance with this observation, hippocampal stimulation decreases plasma corticosteroids in rats and humans, while stimulation of the amygdala produces the opposite effect [40,41]. Furthermore, overactivation of the amygdala is associated with depression and anxiety [37,38].
In the present study, the three non-social stress paradigms induced a reduction in body weight gain and increased immobility in the tail-suspension test (Fig. 2 and 3). Although decreased weight gain under stress does not necessarily reflect the loss of appetite in animals, the loss of interest in enjoyable activities (anhedonia) is one of the core symptoms of major depressive disorder. In addition, despair (feelings of hopelessness) is the most common symptom of depression. Our study indicates that depression- and anxiety-related circuits modified by stress can be dissociated in the mouse brain, and distinct types of stress affect different brain circuits.
There is a possibility that social stress modifies brain circuits related to anxiety as well as depression, while non-social stress influences depression-related brain circuits. Interestingly, psychosocial stress rather than non-social stress seems more relevant in humans. Thus, the social component of stress might be one of the possible explanations for the high rate of comorbidity between anxiety and depression in human patients. Consistent with this idea, several recent lines of evidence suggest a strong association between social stress and anxiety in rodents. Social isolation, a mild social stress paradigm, selectively induced anxiety-related behavioral changes in mice without affecting depression-like behaviors [17]. In addition, repeated social defeat and chronic subordinate colony housing caused increased anxiety-like behavior in mice, but depression-like behaviors were not changed by these social stress paradigms [12,13,14,15]. However, some experimental evidence argues for a selective effect of chronic social stress on anxiety [11,14,16]. Interestingly, a significant interaction between behavioral changes and time of stress was observed. Stressed animals displayed reduced exploratory activity one week after stress, but anhedonia could be observed 3 weeks after chronic social stress [11]. These results imply that prolonged social stress (more than 3 weeks) might induce depression- as well as anxiety-related behavioral changes in rodents [14,16]. Whether prolonged (more than 4 weeks) non-social stress influences anxiety-related brain circuits and induces anxiety-related behavioral changes as well as depression deserves further investigation.
ACKNOWLEDGEMENTS
This study was supported by a grant from the Korean Health Technology R&D Project, Ministry of Health & Welfare (A120476), by a grant (02-2011-058) from the SNUBH research Fund, by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MESF) [2011-0030119], and by a grant (12182KFDA655) from Ministry of Food and Drug Safety in 2013.
References
1. Kessler RC, Bromet EJ. The epidemiology of depression across cultures. Annu Rev Public Health. 2013; 34:119–138. PMID: 23514317.

2. Kessler RC, Chiu WT, Demler O, Merikangas KR, Walters EE. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005; 62:617–627. PMID: 15939839.

3. aan het Rot M, Mathew SJ, Charney DS. Neurobiological mechanisms in major depressive disorder. CMAJ. 2009; 180:305–313. PMID: 19188629.

4. Herman JP, Figueiredo H, Mueller NK, Ulrich-Lai Y, Ostrander MM, Choi DC, Cullinan WE. Central mechanisms of stress integration: hierarchical circuitry controlling hypothalamo-pituitary-adrenocortical responsiveness. Front Neuroendocrinol. 2003; 24:151–180. PMID: 14596810.

5. López JF, Akil H, Watson SJ. Neural circuits mediating stress. Biol Psychiatry. 1999; 46:1461–1471. PMID: 10599476.

6. Krugers HJ, Hoogenraad CC, Groc L. Stress hormones and AMPA receptor trafficking in synaptic plasticity and memory. Nat Rev Neurosci. 2010; 11:675–681. PMID: 20820185.

7. McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007; 87:873–904. PMID: 17615391.

8. Pittenger C, Duman RS. Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology. 2008; 33:88–109. PMID: 17851537.

9. Bao AM, Meynen G, Swaab DF. The stress system in depression and neurodegeneration: focus on the human hypothalamus. Brain Res Rev. 2008; 57:531–553. PMID: 17524488.

10. Mathew SJ, Price RB, Charney DS. Recent advances in the neurobiology of anxiety disorders: implications for novel therapeutics. Am J Med Genet C Semin Med Genet. 2008; 148C:89–98. PMID: 18412102.

11. Rygula R, Abumaria N, Flügge G, Fuchs E, Rüther E, Havemann-Reinecke U. Anhedonia and motivational deficits in rats: impact of chronic social stress. Behav Brain Res. 2005; 162:127–134. PMID: 15922073.

12. Kinsey SG, Bailey MT, Sheridan JF, Padgett DA, Avitsur R. Repeated social defeat causes increased anxiety-like behavior and alters splenocyte function in C57BL/6 and CD-1 mice. Brain Behav Immun. 2007; 21:458–466. PMID: 17178210.

13. Slattery DA, Uschold N, Magoni M, Bär J, Popoli M, Neumann ID, Reber SO. Behavioural consequences of two chronic psychosocial stress paradigms: anxiety without depression. Psychoneuroendocrinology. 2012; 37:702–714. PMID: 21962377.

14. Keeney AJ, Hogg S. Behavioural consequences of repeated social defeat in the mouse: preliminary evaluation of a potential animal model of depression. Behav Pharmacol. 1999; 10:753–764. PMID: 10780291.

15. Krishnan V, Han MH, Graham DL, Berton O, Renthal W, Russo SJ, Laplant Q, Graham A, Lutter M, Lagace DC, Ghose S, Reister R, Tannous P, Green TA, Neve RL, Chakravarty S, Kumar A, Eisch AJ, Self DW, Lee FS, Tamminga CA, Cooper DC, Gershenfeld HK, Nestler EJ. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell. 2007; 131:391–404. PMID: 17956738.

16. Becker C, Zeau B, Rivat C, Blugeot A, Hamon M, Benoliel JJ. Repeated social defeat-induced depression-like behavioral and biological alterations in rats: involvement of cholecystokinin. Mol Psychiatry. 2008; 13:1079–1092. PMID: 17893702.

17. Kwak C, Lee SH, Kaang BK. Social isolation selectively increases anxiety in mice without affecting depression-like behavior. Korean J Physiol Pharmacol. 2009; 13:357–360. PMID: 19915697.

18. Marin MT, Cruz FC, Planeta CS. Chronic restraint or variable stresses differently affect the behavior, corticosterone secretion and body weight in rats. Physiol Behav. 2007; 90:29–35. PMID: 17023009.

19. Zhao Y1, Ma R, Shen J, Su H, Xing D, Du L. A mouse model of depression induced by repeated corticosterone injections. Eur J Pharmacol. 2008; 581:113–120. PMID: 18184609.

20. Ayensu WK, Pucilowski O, Mason GA, Overstreet DH, Rezvani AH, Janowsky DS. Effects of chronic mild stress on serum complement activity, saccharin preference, and corticosterone levels in Flinders lines of rats. Physiol Behav. 1995; 57:165–169. PMID: 7878112.

21. Pitman DL, Ottenweller JE, Natelson BH. Plasma corticosterone levels during repeated presentation of two intensities of restraint stress: chronic stress and habituation. Physiol Behav. 1988; 43:47–55. PMID: 3413250.

22. Lee B, Sur B, Park J, Kim SH, Kwon S, Yeom M, Shim I, Lee H, Hahm DH. Chronic administration of baicalein decreases depression-like behavior induced by repeated restraint stress in rats. Korean J Physiol Pharmacol. 2013; 17:393–403. PMID: 24227939.

23. Harris RB, Zhou J, Youngblood BD, Rybkin II, Smagin GN, Ryan DH. Effect of repeated stress on body weight and body composition of rats fed low- and high-fat diets. Am J Physiol. 1998; 275:R1928–R1938. PMID: 9843882.
24. Schweizer MC, Henniger MS, Sillaber I. Chronic mild stress (CMS) in mice: of anhedonia, 'anomalous anxiolysis' and activity. PLoS One. 2009; 4:e4326. PMID: 19177164.

25. Gregus A, Wintink AJ, Davis AC, Kalynchuk LE. Effect of repeated corticosterone injections and restraint stress on anxiety and depression-like behavior in male rats. Behav Brain Res. 2005; 156:105–114. PMID: 15474655.

26. Lee JK. Anti-depressant like effect of methyl gallate isolated from acer barbinerve in mice. Korean J Physiol Pharmacol. 2013; 17:441–446. PMID: 24227946.
27. Steru L, Chermat R, Thierry B, Simon P. The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology (Berl). 1985; 85:367–370. PMID: 3923523.

28. Archer J. Tests for emotionality in rats and mice: a review. Anim Behav. 1973; 21:205–235. PMID: 4578750.

29. Prut L, Belzung C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. Eur J Pharmacol. 2003; 463:3–33. PMID: 12600700.

30. Simon P, Dupuis R, Costentin J. Thigmotaxis as an index of anxiety in mice. Influence of dopaminergic transmissions. Behav Brain Res. 1994; 61:59–64. PMID: 7913324.

31. Lamers F, van Oppen P, Comijs HC, Smit JH, Spinhoven P, van Balkom AJ, Nolen WA, Zitman FG, Beekman AT, Penninx BW. Comorbidity patterns of anxiety and depressive disorders in a large cohort study: the Netherlands Study of Depression and Anxiety (NESDA). J Clin Psychiatry. 2011; 72:341–348. PMID: 21294994.
32. Sartorius N, Ustün TB, Lecrubier Y, Wittchen HU. Depression comorbid with anxiety: results from the WHO study on psychological disorders in primary health care. Br J Psychiatry Suppl. 1996; (30):38–43. PMID: 8864147.

33. Gorman JM. Comorbid depression and anxiety spectrum disorders. Depress Anxiety. 1996-1997; 4:160–168. PMID: 9166648.

34. Hirschfeld RM. The comorbidity of major depression and anxiety disorders: recognition and management in primary care. Prim Care Companion J Clin Psychiatry. 2001; 3:244–254. PMID: 15014592.
35. Olfson M, Fireman B, Weissman MM, Leon AC, Sheehan DV, Kathol RG, Hoven C, Farber L. Mental disorders and disability among patients in a primary care group practice. Am J Psychiatry. 1997; 154:1734–1740. PMID: 9396954.

36. Russo SJ, Murrough JW, Han MH, Charney DS, Nestler EJ. Neurobiology of resilience. Nat Neurosci. 2012; 15:1475–1484. PMID: 23064380.

37. Ressler KJ, Mayberg HS. Targeting abnormal neural circuits in mood and anxiety disorders: from the laboratory to the clinic. Nat Neurosci. 2007; 10:1116–1124. PMID: 17726478.

38. Drevets WC. Neuroimaging studies of mood disorders. Biol Psychiatry. 2000; 48:813–829. PMID: 11063977.

39. Fuchs E, Flügge G. Chronic social stress: effects on limbic brain structures. Physiol Behav. 2003; 79:417–427. PMID: 12954436.

40. Dunn JD, Orr SE. Differential plasma corticosterone responses to hippocampal stimulation. Exp Brain Res. 1984; 54:1–6. PMID: 6321219.

41. Rubin RT, Mandell AJ, Crandall PH. Corticosteroid responses to limbic stimulation in man: localization of stimulus sites. Science. 1966; 153:767–768. PMID: 5940897.

Fig. 1
Schematic representation of the experimental design. Experimental groups were exposed to chronic predictable (restraint) stress (CPS), chronic unpredictable stress (CUS), or repeated corticosterone-HBC complex injection (RCI).
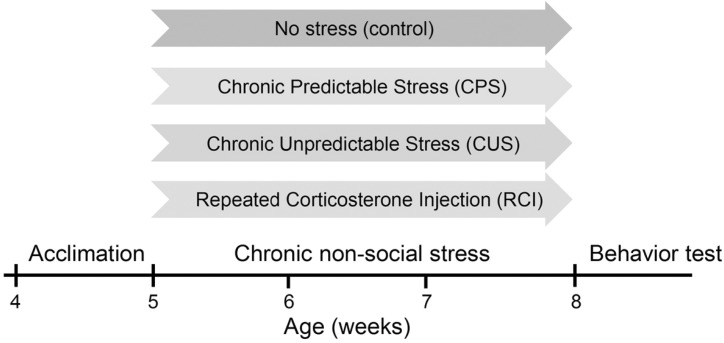
Fig. 2
Effect of 3-week non-social stress on body weight. The body weight of mice during the period of CPS (A), CUS (B), and RCI (C). (D) Body weights of the stressed groups after the stress phase. Data were normalized to each control group (dashed line).

Fig. 3
Chronic non-social stress increases depression-like behaviors. Minute-by-minute analysis of the time spent immobile during the tail suspension test in mice exposed to CPS (A), CUS (B), and RCI (C). (D) The total time spent immobile during the 6-minute test in each stress group was normalized to each control group (dashed line).
Fig. 4
Normal locomotor activity and thigmotaxic behaviors in mice with chronic non-social stress. Examples of exploratory paths (A), distance moved (B), and thigmotaxis (C) from the CPS and control groups are shown. Thigmotaxis levels are expressed as the percent time spent in the periphery. Exploratory paths, distance moved, and thigmotaxis from the CUS (D-F) and RCI (G-I) groups are compared with each control. Total distance moved (J) and thigmotaxis (K) during the 30-minute test were normalized to the control group (dashed line) and summarized.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download