INTRODUCTION
METHODS
Animal model for 2d-H
Measurement of pulmonary arterial pressure (PAP) and HPV
Histology
Preparations of isolated PASMCs
Measurement of membrane currents in Patch-clamp experiments
Solutions and drugs
Statistical analysis
RESULTS
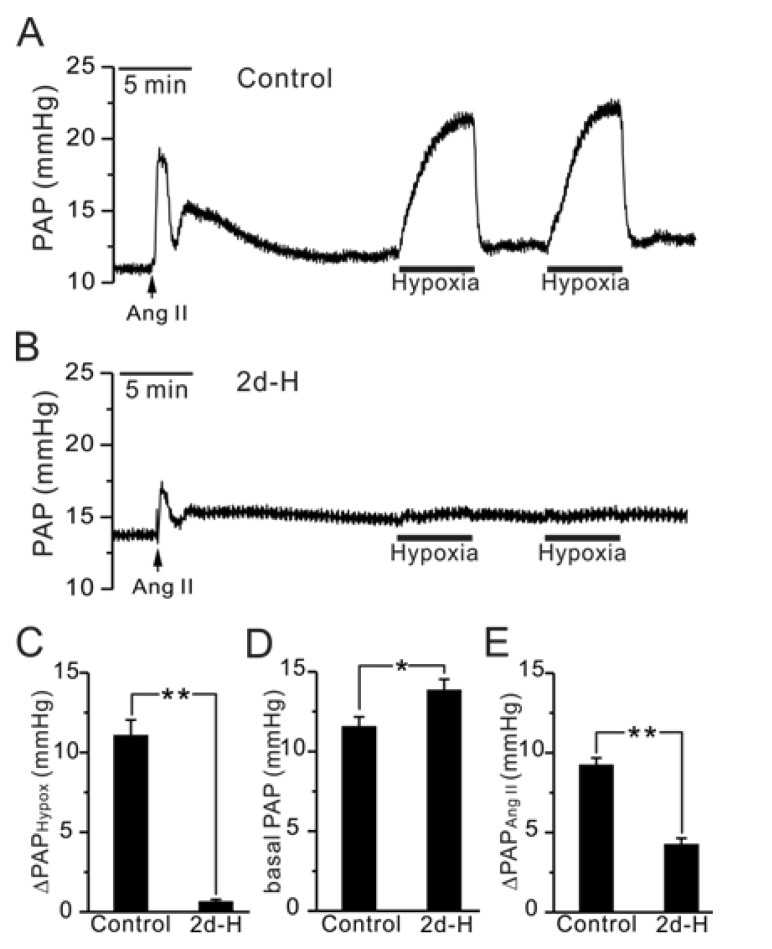 | Fig. 1Comparison of HPV, basal PAP and Ang II-induced ΔPAP between control and 2d-H rats in V/P lungs. (A, B) Representative traces of PAP recording in control (A) and 2d-H rats (B). Hypoxic ventilation (3% PO2)-induced PAP increase (ΔPAPHypox) was abolished in 2d-H. Ang II was initially applied to the perfusate, inducing a transient increase in PAP. (C~E) Summaries of ΔPAPHypoxia (C), basal PAP (D), and AngII-induced ΔPAP (E) are shown as bar graphs comparing the results between control and 2d-H rats (*p<0.05, **p<0.01). |
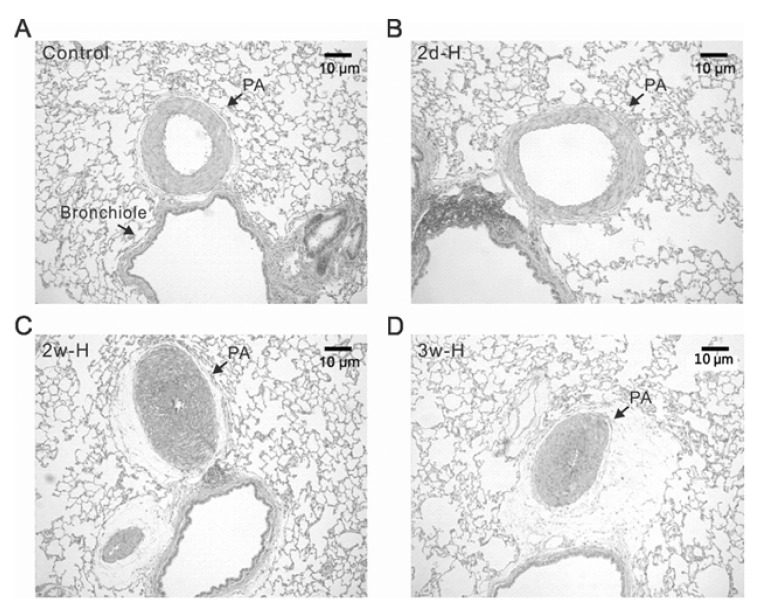 | Fig. 2Differential structural changes of PAs according to the period of hypoxia in rats. Representative histological figures (H&E staining) of lung from control (A), 2 days of hypoxia (2d-H, B), 2 weeks of hypoxia (2w-H, C), and 3 weeks of hypoxia (3w-H, D). The medial thickness of PA was similar between control and 2d-H, whereas the PAs from 2w-H and 3w-H showed marked hypertropic changes with almost obliterated lumen. |
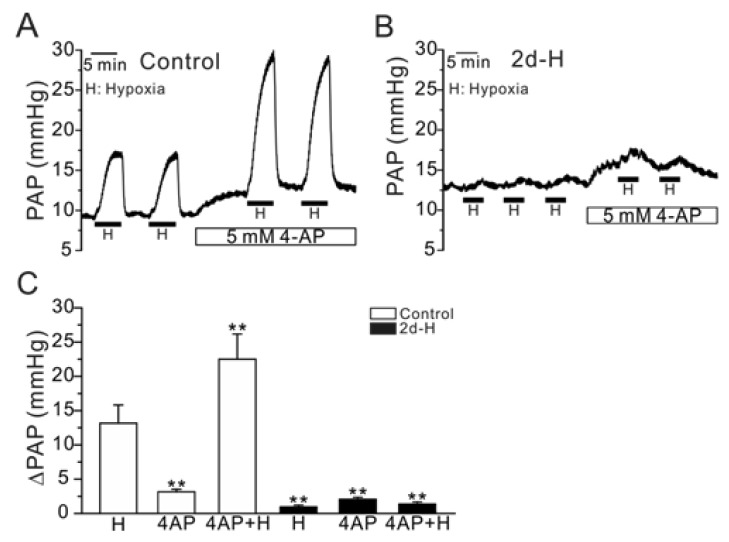 | Fig. 3Effect of 4-AP, a Kv inhibitor, on PAP and HPV (ΔPAPHypoxia) in control and 2d-H rats. (A, B) Representative traces of PAP recording in control and 2d-H rats. Both basal PAP and ΔPAPHypoxia were increased by 5 mM 4-AP in control (A). In 2d-H, however, basal PAP was slightly increased by 4-AP, and ΔPAPHypoxia was not recovered by the 4-AP pretreatment (B). (C) Summaries of the results shown as bar graphs (n=5; control, n=4; 2d-H, **p<0.01). |
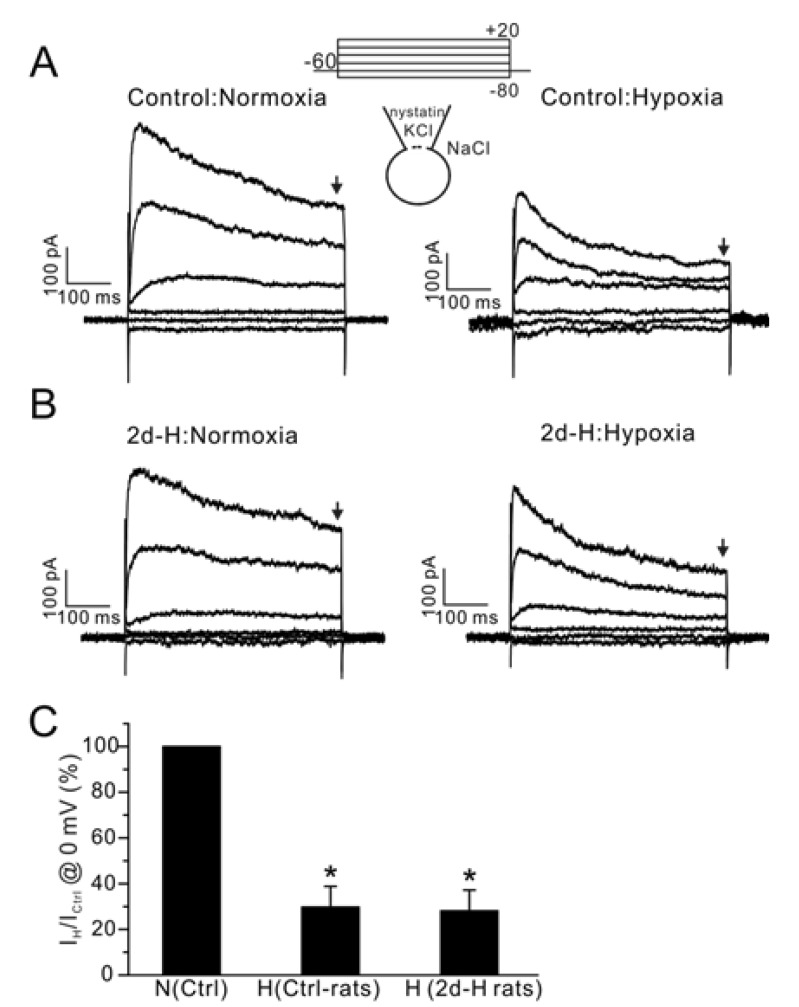 | Fig. 4Effect of acute hypoxia on IKv in PASMCs from control and 2d-H rats. (A, B) Representative current traces obtained by step pulses (between -80 mV and 20 mV, 20 mV interval, 500 ms duration,) from -60 mV of holding potential. The recording was done at 37℃. Acute hypoxia (PO2 3%) decreased the amplitudes of IKv in PASMCs similarly between control and 2d-H PASMCs. (C) Summary of outward current amplitudes at 0 mV normalized to each control (Ctrl-rats; control, H; hypoxia, *p<0.05). The amplitudes of outward currents were measured at the end of step pulses as indicated by downward arrows above (A, B). |
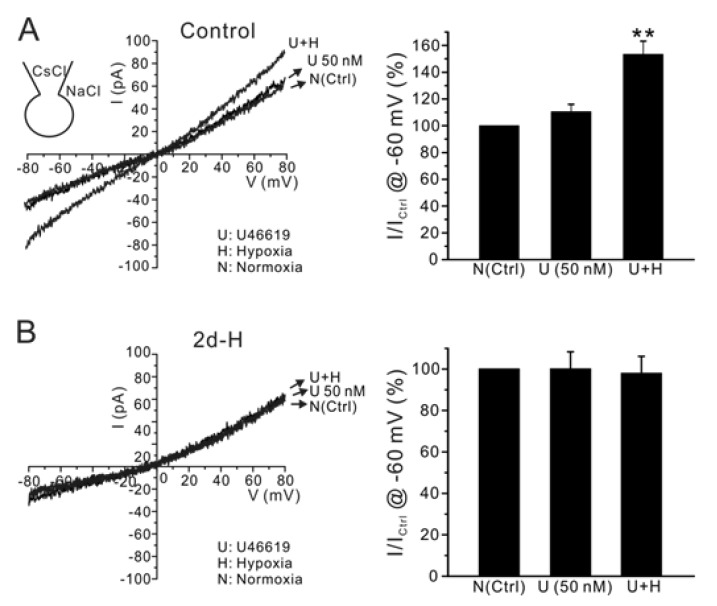 | Fig. 5Effect of acute hypoxia on INSC in PASMCs of control and 2d-H rats. Representative I/V curves of INSC were obtained by ramp pulses (-80 mV~80 mV) with CsCl pipette solution. (A) Application of U46619 alone did not significantly increased INSC, while the additional acute hypoxia significantly increased the conductance. (B) In contrast, INSC was not increased by acute hypoxia in 2d-H rats. Summaries of inward current amplitudes at -60 mV normalized to each control (U; U46619, H; hypoxia) are display as bar graphs in the right panels (**p<0.01). |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download