ABBREVIATIONS
TEA
4-AP
OXT
RMP
KCa channel
KV channel
K2P channels
TASK
TREK
KRB
MES
pHo
TTX
ATR
NIOK
VDCCL
INTRODUCTION
METHODS
Tissue preparation for isometric contraction
Immunohistochemical labeling of K2P channel (TASK-2)
Tissue preparation for sharp electrode recording
Solution and drugs
Statistics
RESULTS
Characterization of oxitocin (OXT)-induced contraction of uterine circular muscle
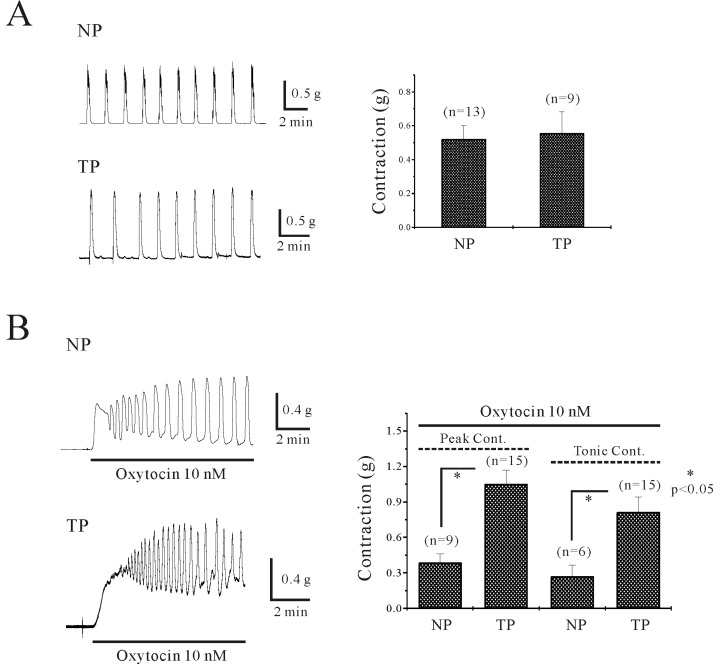 | Fig. 1Spontaneous and oxitocin (OXT)-induced contraction in uterine circular muscle of mouse. (A) Uterine circular muscle shows spontaneous phasic contraction in both non-pregnant (0.5 g) and pregnant myometrium (0.6 g). (B) OXT produced initial and tonic contractions however tonic component was not persistent in non-pregnant uterine circular muscle. Initial contractions by OXT were 0.4 g (non-pregnant) and 1.1 g (pregnant). However, tonic contraction were 0.3 g (non-pregnant) and 0.8 g (pregnant) (p<0.05). Data was summarized in right panel. Asterisks show a statistical significance (p<0.05). |
Effect of quinidine on spontaneous electrical activity of uterine circular muscle
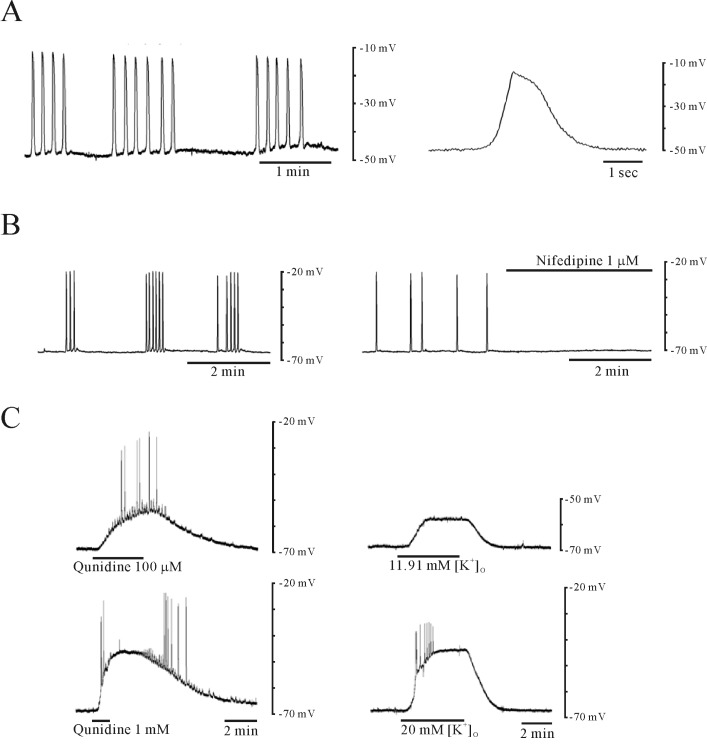 | Fig. 2Effect of quinidine on spontaneous electrical activity of murine myometrium. (A) Electrical responses were recorded from circular smooth muscle cells of the mouse uterus. In intact tissues, circular muscle cells were spontaneously active with generation of plateau-type action potentials. (B) The spontaneous action potentials were inhibited by nifedipine. (C) Application of quinidine and high K+ depolarized the membrane, in a concentration-dependent manner, with generation of spike potentials. |
Effect of quinidine and extracellular acidosis on isometric contraction of uterine circular muscle
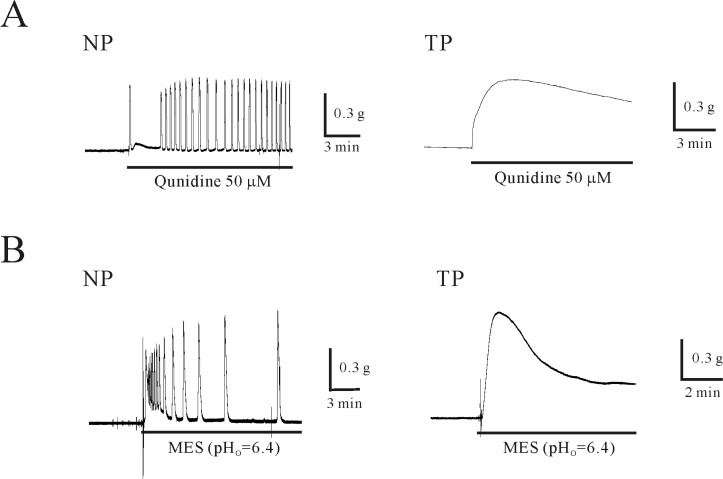 | Fig. 3Effect of quinidine and extracellular acidosis on isometric contraction of uterine circular muscle of mouse. (A) Quinidine (50 µM) produced phasic contraction of non-pregnant myometrium. Quinidine-induced tonic contraction of pregnant myometrium was increased compared to that of non-pregnant myometrium. (B) Extracellular acidosis (pHo=6.4) produced contraction in non-pregnant myometrium. This contraction was also increased compared to that of non-pregnant myometrium. |
Effect of tetraethylammonium (TEA) and 4-aminopyridine (4-AP) which inhibits of Ca2+-activated K+ (KCa) channel and voltage-dependent K+ (KV) channel on uterine circular muscle of mouse
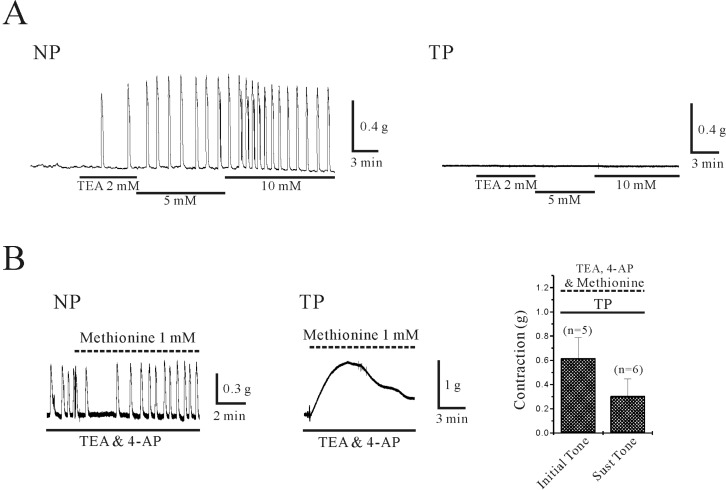 | Fig. 4Effect of TEA and 4-AP on isometric contraction of uterine circular muscle of mouse. (A) TEA (2~5 mM) produced concentration-dependent phasic contraction in non-pregnant uterine circular muscle. However, it did not show any effect in pregnant uterine circular muscle (right panel). (B) In the presence of TEA and 4-AP, effect of stretch-dependent K2P channels (TREK-1) inhibitor (L-methionine) was studied. L-methionine (1 mM) produced contractions that spontaneously decayed to near baseline values within 10~15 min in pregnant uterine circular muscle. However, it did not show significant effect in non-pregnant uterine circular muscle. Data from pregnant uterine circular muscle was summarized in right panel. |
Regulation of murine pregnant myometrial contractility by TASK-2 channel inhibitors
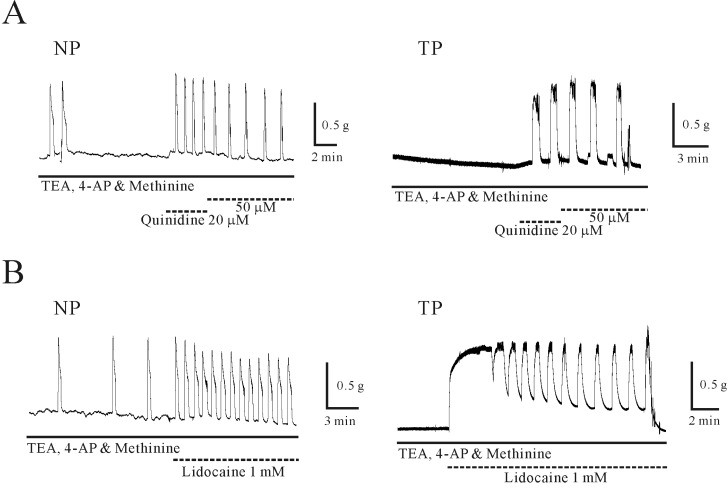 | Fig. 5Regulation of contraction of uterine circular muscle by TASK-2 channel inhibitors in the presence of TEA, 4-AP and L-methionine. The TASK-2 channel inhibitors produced contractions in uterine circular muscle. (A, B) Adding quinidine or lidocaine in the presence of L-methionine produced further robust contractions of uterine circular muscle. Contraction by TASK-2 channel inhibitors was increased in pregnant uterine circular muscle. |
Identification of TASK-2 by immunohistochemistry in uterine circular muscle of mouse
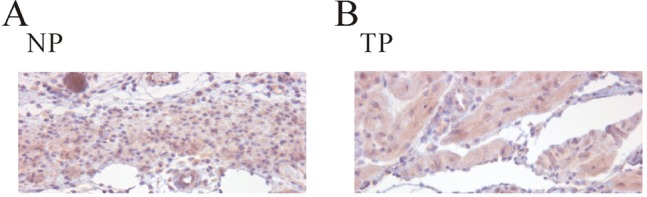 | Fig. 6Identification of TASK-2 channels by immunohistochemistry in uterine circular muscle. We tried to elucidate expression and increase of TASK-2 channels during pregnancy using immunohistochemistry. (A, B) Immunoreactivity for TASK-2 channel antibody in pregnant uterine circular muscle was more even compared to that of non-pregnant uterine circular muscle. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download