INTRODUCTION
METHODS
Primary astrocyte cultures
Induction of chemical ischemia in cultured astrocytes
MTT Assay for cell viability
Lactic dehydrogenase (LDH) assay for cell Cytotoxicity
RNA isolation and real-time PCR
Membrane fraction from cultured astrocytes
Western blotting
Assay of Na-K ATPase specific activity
Statistical analysis and Quantification of PCR and Western blots
RESULTS
Effect of chemical ischemia on the cytotoxicity and viability of cultured astrocytes
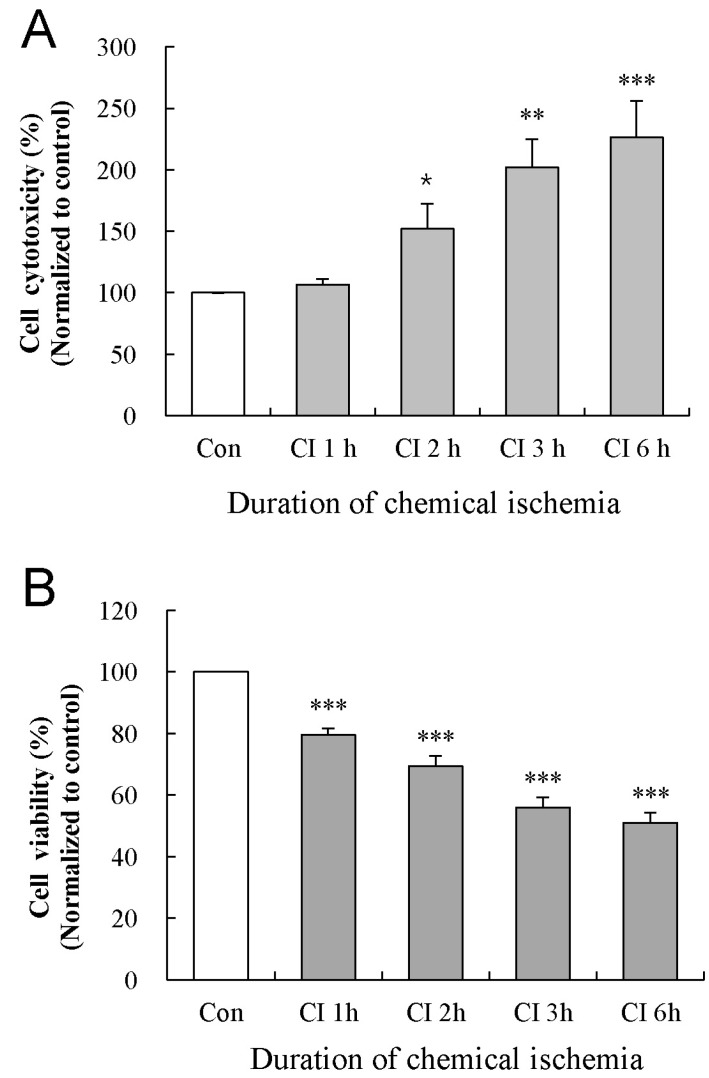 | Fig. 2Effect of chemical ischemia on the cytotoxicity and viability of cultured astrocytes. Cytotoxicity and cell viability was measured by LDH and MTT assays, respectively. The levels of LDH release (A) and MTT reduction (B) were quantified and compared to the control at each time point. Each value indicates the mean±S.E.M. normalized to the control of each time point. Data were obtained from five experiments. *p<0.05, **p<0.01, and ***p<0.001. |
Effects of chemical ischemia on the protein and mRNA expression of the Na-K ATPase α1 subunit in cultured astrocytes
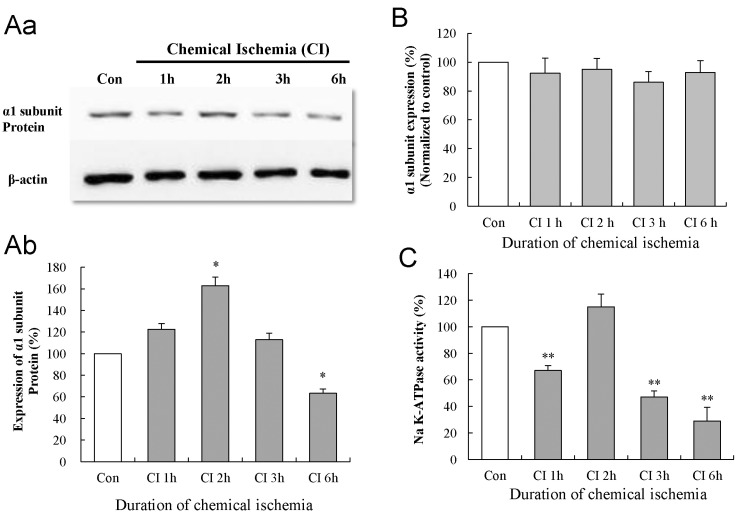 | Fig. 3Effect of chemical ischemia on the expression of Na-K ATPase α1 subunit and Na-K ATPase activity in cultured astrocytes. (A) Western blot analysis shows the expression of Na-K ATPase α1 subunit protein following chemical ischemia for 1, 2, 3, and 6 h. Data were obtained from five experiments. (B) Real-time PCR analysis shows the expression of Na-K ATPase α1 subunit mRNA in cultured astrocytes following chemical ischemia for 1, 2, 3, and 6 h. Data were obtained from four experiments. (C) The activity of Na-K ATPase in the membrane fraction of cultured astrocytes after chemical ischemia for 1, 2, 3, and 6 h. Data were obtained from five experiments. For all experiments, each value indicates the mean±S.E.M. normalized to the control of each time point. *p<0.05, **p<0.01. |
Effects of chemical ischemia on Na-K ATPase activity in cultured astrocytes
Effect of reoxygenation on chemical ischemia-induced cytotoxicity of cultured astrocytes
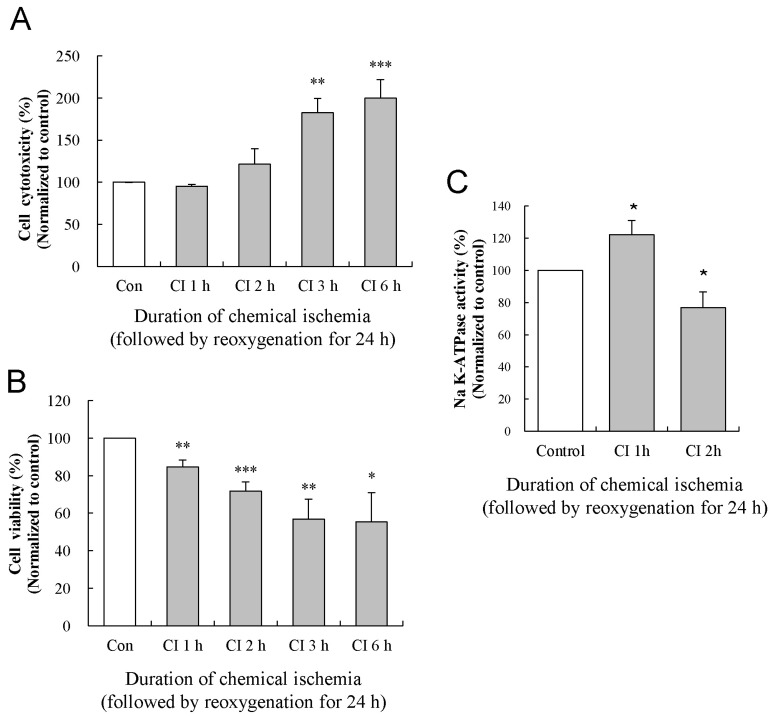 | Fig. 4Effects of reoxygenation on the changes in the cytotoxicity, viability, and activity of the Na-K ATPase. The levels of LDH release (A) and MTT reduction (B) were quantified at 24 h of reoxygenation following chemical ischemia for 1, 2, 3, and 6 h. (C) Activity of Na-K ATPase was measured at 24 h of reoxygenation following chemical ischemia for 1 and 2 h. Data show the mean± S.E.M. of the relative values obtained from five animals. *p<0.05, **p<0.01, and ***p<0.001. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download