INTRODUCTION
METHODS
Measurement of isometric tension in isolated PAs
PAP measurement in V/P lungs
Preparation of lung slices
Image acquisition and analysis
Solutions and chemicals
Statistical analysis
RESULTS
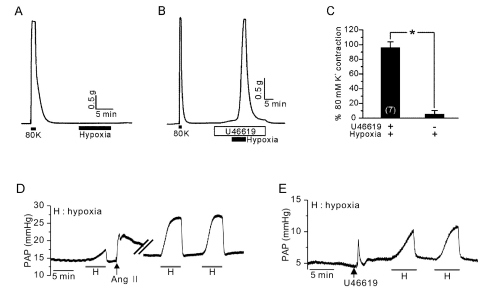 | Fig. 1Essential role of TXA2 on HPV in PAs. (A) Hypoxia (3% Po2) alone did not induce an increased tension of PAs. (B) In the presence of U46619 (10 nM), a strong contraction of PAs occurred in response to hypoxia. (C) Summaries of PAs tone normalized to 80K-induced contraction were shown as a bar graph (mean±SEM, n=7). *Indicates statistically significant difference from the control value (p<0.05). (D) Induction of HPV in V/P lungs. The increase in pulmonary arterial pressure (PAP, mmHg) was observed by hypoxic ventilation. In the same lung, Ang II induced a transient PAP increase, which augmented the HPV response. (E) A pretreatment with U46619 also induced a transient PAP increase, and HPV was also observed afterwards. |
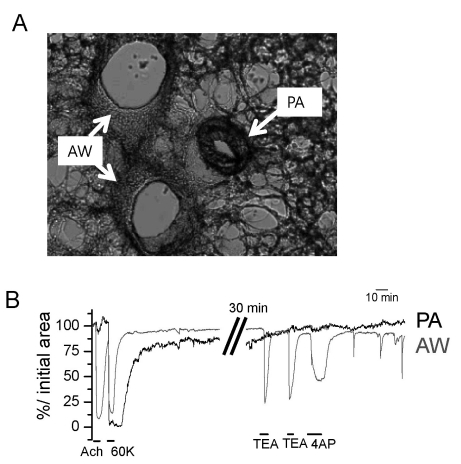 | Fig. 2Identification of PA and AW constrictions in rat PCLS. (A) A representative image of rat PCLS demonstrating the AW and PA. (B) Functional discrimination of PA and AW according to differential responses to ACh and K+ channel blockers. AW showed strong constriction to ACh (10 µM), tetraethylammonium (TEA, 2 mM) and 4-aminopyridine (4AP, 5 mM) while PA showed no response to the concomitant application of the above agents. |
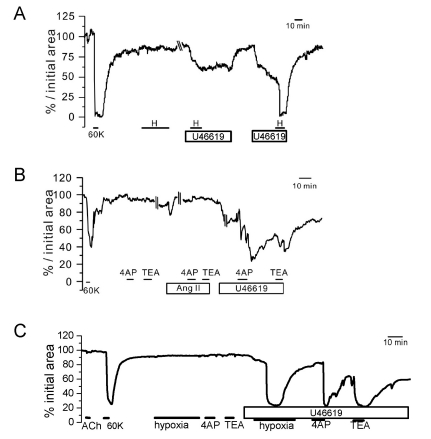 | Fig. 3Responses of PA to hypoxia and K+ channel blockers under pretone conditions in rat PCLS. Three representative cases of relative luminal area (% initial area) changes in PAs. In all cases (A~C), the K+ channel blockers (TEA and 4AP) induced reversible constriction only under the pretreatment with 5 nM U46619 while not with 100 nM Ang II (B). Also, the hypoxic constriction was observed under the pretreatment with 5 nM U46619 (A, C). |
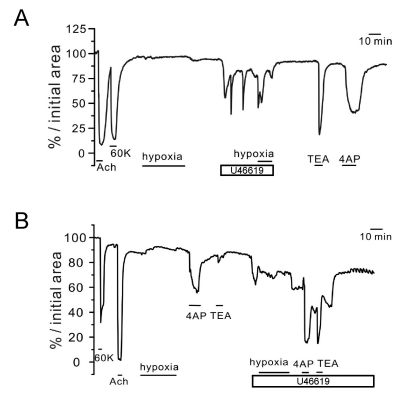 | Fig. 4Responses of AW to hypoxia and K+ channel blockers under pretone conditions in rat PCLS. Two representative cases of luminal area (% initial area) changes in AWs. The strong contractile response to 10 µM ACh was confirmed along with the response to 60 K. Hypoxia alone had no effect (A) or induced a slight dilation (B) of AW. Application of 5 nM U46619 induced oscillatory or tonic constrictions that were not affected (A) or partly inhibited by hypoxia (B). Different from PAs, both 4AP and TEA induced constriction of AW in the absence of U46619 pretreatment. |
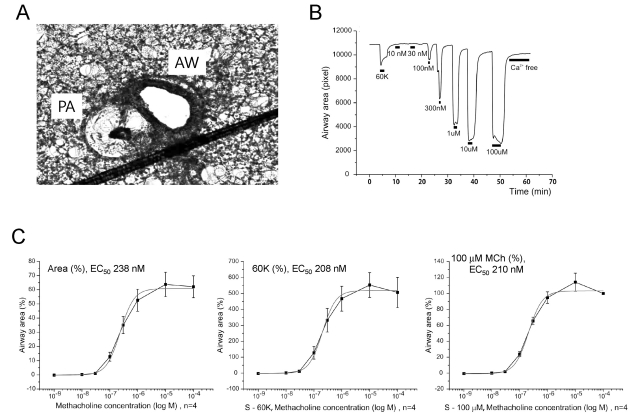 | Fig. 5Response of AW to metacholine (MCh) in mouse PCLS. (A) A representative image of rat PCLS demonstrating the AW and PA. Note the relatively narrow lumen of PA in the mouse PCLS. (B) Concentration-dependent reversible constrictions of mouse AW. The maximum constriction induced by 100 µM MCh was markedly larger (>five fold) than the 60K-constriction. (C) Normalized AW constrictions to different concentrations of MCh were averaged and fitted to logistic functions. Each panel was obtained by different type of normalization criteria; relative change from the maximum luminal area (left), normalized against 60K constriction (middle), and normalized to maximum constriction by 100 µM MCh (right). Regardless of the normalization criteria, the half-effective concentration (EC50) of MCh located at similar ranges. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download