Abstract
It has been shown that QGC isolated and purified from Rumecis folium found protective effects of gastritis and esophagitis which EXT is an ethanol extract of it. We examined acute toxicity and the general pharmacological action of QGC EXT to search for any side effects of it in rats, mice, guinea pigs, and cats. In a single dose toxicity study, QGC EXT didn't show toxicological effects in rats and mice, and the LD50 was over 5 g/kg in both animals, and there were also no changes in weight, feed and water intake during these toxicological experimental periods. We examined the general pharmacological action on central controlled behavior responses, and peripheral organs including blood pressure, heart rate, respiration and gastrointestinal system, We found that there were no significant changes in body temperature, locomotors activity, stereotyped behaviors, sleeping time, and convulsion. In other studies, writhing reaction, normal body temperature, there did not appear to be any changes. The large intestine movement and electrical field stimulation-induced contraction was not changes by its EXT. In addition, the influences on blood pressure, heart rates, and respiration by QGC EXT were not found. These results indicate that QGC EXT may be very safe as a new drug, since its LD50 was very high over 5 g/kg and any side effects were not found.
Flavonoids, which are secondary metabolites in plants, are considered relatively non-toxic bioactive substances and have diverse biological effects, such as anti-inflammatory, anti-oxidant, anti-allergic, hepatoprotective, anti-thrombotic, anti-viral and anti-carcinogenic activities [1,2]. Among the flavonoids, quercetin has been given special attention since it is an antioxidant which efficiently scavenges highly reactive biological species such as peroxynitrite and the hydroxyl radical [3]. Rumecis folium is a family of polygonaceae and this plant was used for disinfestations, treating diarrhea, fever, edema, jaundice and constipation in traditionally oriental medicine. In the previous study performed. QGC (Quercetin-3-O-β-D-Glucuronopyranoside) was isolated from a Rumecis folium through several steps, it QGC had a more potent effect than quercetin on the inhibition of experimental acute and chronic gastritis and ethanol-induced gastritis in SD rats in vivo [4].
The QGC EXT was extracted from Rumecis folium by ethanol. In previous studies, QGC EXT revealed protective effects on indomethacin induced gastric damage [5]. In the present study, to perform part of a preclinical evaluation of QGC EXT, the acute toxicity and general pharmacological effects of QGC EXT on general behavior, central nervous system, digestive system, smooth muscles, cardiovascular and respiratory systems were investigated.
Male Sprague-Dawley rats (200 g), guinea pigs (300 g), mice (30 g), or cats (3.5 kg) were used for the experiments. Animals were housed in a controlled room (temperature of 22±20℃, relative humidity of 50±5%), were maintained in a light controlled room and were given solid diet and tap water ad libitum. All animals were fasted over night before the experiment. This experiment was approved by the Institutional Animal Care and Use Committee of the Chung-Ang University Medical School.
QGC EXT was separated and extracted from Rumex aquaticus, phenylephrine, chlorpromazine, acetic acid, naproxen, zolazepam, strychnine, valproic acid, BaSO4, acetylcholine, urethane and other materials were purchased from Sigma Chemical Co. (St. Louis, Mo, USA).
Drugs were administered orally at a dose rate of 0.5, 1, 2, 3, 5 g/kg in both sexes, respectively. Over the course of 2 weeks, observations regarding the variation of physical states, weights, intake of feed & water, once a day or more by naked eyes were taken. The above parameters were measured before and after treatments. Dead animals were displayed on 4th, 7th, 10th, 14th day of observation. To evaluate the maximum tolerated dose, consecutive doses for 4 days (1, 2, and 3 g/kg) were administrated orally in rats. The number of death animal was recorded. Autopsies were performed and checked hematological findings, main organs weights and other phenomena (diarrhea, constipation, stool state, stomach mucosa, etc).
The methods used were based on the procedures described by Irwin [6] and Lee [7]. The effect of QGC EXT was observed on locomotor activity, writhing response, fighting, convulsion, tremor, exophthalmos, ptosis, piloerection, tail elevation, traction, motor incoordination, muscle tone, catalepsy, righting reflex, pain response, pinna reflex, skin color, respiration, lacrimation, salivation, diarrhea, vocalization and death were observed. One night-fasted animals were administrated with the drugs, General actions about 24 items were indicated as "+" or "-" after specific times. There were no changes on the 24 parameter of the behavior.
Mice were put on the Rotarod (Ugo Basile, ITALY) to discipline them. Rota rod rotation stick was upside down and rotated 5 times per minute described by Dunham et al. [8]. The time it took to fall down was measured and if the animal fell down within 2 minutes, we judged that motion hindrance occurred. Chlorpromazine 5 mg/kg was used as a positive control.
We put mice into the cage and beaker and adjusted them for 30 minutes Koster et al. [9]. Normal saline-diluted with 0.6% acetic acid was injected into the abdominal cavity by 0.1 ml/10 g for writhing reaction (repetition of stretching, squirining, abdominal constriction response). The number of writhing reactions was recorded 10 minutes and 20 minutes after the test. The drug was administrated before acetic acid in mice administration. Considering the effective maintenance time from the preliminary experiment, administration route, and methods, we observed the writhing reaction at the maximum-effect time. 500 mg/kg naproxen was administered as a positive control group.
In each group 10 mice were used. According to the Randall and Selitto [10] method, 20% yeast suspension was injected into the sole and the sample was administrated orally 2 hours later. Using the Randall-Selitto (UGO Basile, Italy) machine, Analgesy meter's needle was put on the foot of mice, and then we started a motor. When we increased the pressure of the needle gradually, the weight that made mice pull out their foot became the threshold for pain. QGC EXT 10, 30, 50 mg/kg was administrated orally to each group, and then the values after 1 hr, 2 hrs were measured. 500 mg/kg naproxen was used as a control.
Zolazepam at 12.5 mg/kg was injected into the abdominal cavity after the administration of QGC EXT in animals through the oral route. The index losing more than 5 seconds of righting reflex, estimated the start time of sleeping and duration of sleeping. Chlorpromazine 20 mg/kg was injected into the abdominal cavity as the positive control group.
Strychnine (1.5 mg/kg) was injected in abdominal cavity 30 min after the drug was administered [11,12]. We watched for symptoms and compared them with the control group. The incidence of Tonic Flexion (TF), Tonic Extension (TE), Clonic convulsion (CL) and mortality were measured. Valproic acid 50 mg/kg was used as the positive control.
Temperature was measured through the rectal route. Temperature was measured 0.5, 1.0, 2.0 and 3.0 hrs after the drug was administered. Chlorpromazine (20 mg/kg) was used as the positive control group.
1 hour before the experiment, we chose animals which did not have diarrhea. 30 minutes after oral administration of the drug, 25% BaSO4 suspension (0.1 ml/10 g) was administered. Then we estimated the time of BaSO4 to come out and compared the control group with the test group.
We used cat or guinea pig low esophageal smooth muscle, ileum and relaxed it in a 1 ml organ bath with Krebs solution. The muscle strips were stimulated with a pulse train of 80 V in amplitude and 10 s in duration with a pulse duration of 1 ms through platinum wire electrodes placed longitudinally on either side of the strip. Acetylcholine was treated with various concentrations, and applied to 4 Hz electronic stimulation and measured the contraction using a polygraph chamber (Grass Instrument Co, Quincy, Mass, USA). Single muscle cells were isolated according to Sohn et al. [13] and Biancani et al. [14]. Muscle strips were incubated overnight in a normal potassium-HEPES buffer containing collagenase. The next day we incubated the tissue in a water bath at 31℃ for 30 min. After incubation, the digested tissue was poured out over a 360-µm Nitex filter mesh, rinsed in collagenase-free HEPES buffer to remove any trace of collagenase, and then incubated in this solution at 31℃. The cells were allowed to dissociate freely for 10 to 20 min. Suspensions of single muscle cells were harvested by filtration through a 500 µm Nitex filter mesh.
After anesthesia with a urethane polyethylene (PE-10) tube was inserted to the carotid artery and femoral vein, blood pressure and heart rates were tested. We applied the physiological recording system (polygraph, Grass Instrument Co., Quincy, Mass, USA) using a pneumograph to measure respiratory rate. Blood pressure and heart rate was carried out using the tachograph. As a positive control, phenylephrine 10 µg/kg was administered.
Data are expressed as means±SEM. Statistical significance was determined using a two-tailed Student's t-test for paired observations or Chi-square test. A difference was considered as significant when the p value was lower than 0.05. Traces were representative of at least three experiments on three or more muscle preparations.
Drug at a dose rate of 0.5, 1, 2, 3, or 5 g/kg was applied both in male and female experimental animals, respectively. There were no changes in physical states, weights, intake of feed & water, or the recordings about the variation performed once per 3~4 days (Fig. 1 of rats, Fig. 2 of mice). We found that LD50 was larger than 5 g/kg in both mice and rats. Dead animals and these data were recorded on the 1st, 3rd, 5th, 7th, 9th, 11th, 13th, 15th of the observation. We found no dead animals with regards to every dose in rats and mice (Table 1 of rats and Table 2 of mice). In addition, changes of hematological findings, main organ weights and other phenomena (diarrhea, constipation, stool state, stomach mucosa) were not found. To evaluate the maximum tolerated dose, consecutive doses for 4 days (1, 2, and 3 g/kg) were administered orally in rats. The number of dead animal was also recorded. We did not find any differences in hematological findings, main organs weights or other phenomena (data not shown).
The chronic oral administration of QGC EXT (10 mg, 30 mg, 50 mg/kg) caused no observable change in general behavior responses (stereotyped behavior, convulsion, exophthalmos, ptosis, piloerection, tail elevation, traction, motor incoordination, muscle tone, analgesia, abnormal tone, catalepy, righting reflex, pinna reflex, pupil reflex, skin color, respiration, lacrimation, salivation, urination, diarrhea, death during 4 hr periods) of the rats and, compared to the control group, no significant changes in body weight, food intake and utilization of food in treated mice. Both the control and treated rats appeared uniformly healthy at the end of study.
Administration of QGC EXT at doses of 10 mg, 30 mg, 50 mg/kg showed no observable changes in muscle cooperation in mice at 30, 60, 90, 120, 180, 240 minutes. In contrast, chlorpromazine, which was a positive control, showed muscle cooperation disturbance (Table 3).
Administration of QGC EXT at doses of 10 mg, 30 mg, 50 mg/kg showed no observable change in No. of Writhings in comparison with the vehicle group. However, when naproxen (positive control) was treated, the No. of writhing reactions decreased significantly (p<0.05) (Table 4).
Administration of QGC EXT at doses of 10 mg, 30 mg, 50 mg/kg showed no observable change in thresholds in comparison with vehicle. Naproxen (positive control) increased pain thresholds significantly (p<0.05) (Table 5).
Zolazepam increased sleeping time remarkably. Administration of QGC EXT at doses of 10 mg, 30 mg, 50 mg/kg showed no observable change in onset time and sleeping time. Chlorpromazine (positive control) decreased onset time and increased sleeping time significantly (p<0.05) (Table 6).
Strychnine induced CL and TE, and death in all cases. Administration of QGC EXT at doses of 10 mg, 30 mg, 50 mg/kg showed no observable change (Table 7). Valproic acid prevented its convulsion. QGC EXT itself has no convulsive effect (data not shown).
Administration of QGC EXT at doses of 10 mg, 30 mg, 50 mg/kg showed no significant changes in body temperature in mice during 240 minutes periods (Table 8).
Administration of QGC EXT at doses of 10 mg, 30 mg, 50 mg/kg showed no significant changes in large intestine movement in mice during 240 minutes periods (Table 9), it was the same as the vehicle treatment.
Both in cat esophageal smooth muscle and cat LES, the dose of QGC EXT 0.01, 0.03, 0.05 mg/ml did not cause a contraction (Fig. 3). When we treated with 10-7, 10-6, 10-5 M Acetylcholine to esophageal smooth muscle and LES, the concentration-dependent contraction was exhibited. There was no change by treatment with 0.01, 0.03, 0.05 mg/ml QGC EXT (Fig. 4) Electrical stimulation induced contractions of esophageal smooth muscle and relaxation of LES of cats. When QGC EXT was added, there was no significant variation (Table 10). When we used acetylcholine and histamine as agonists, the same result was obtained in guinea pigs (Table 11). The muscle strips were stretched 2.5 gm. for optimal force development and equilibrated. When we treated 10-7 M acetylcholine, the maximum contraction occurred at 30 second, and then the contraction maintained during 20 minutes. When QGC EXT was also added, the contraction was inhibited (Fig. 5) By electrical stimulation, esophageal smooth muscle exhibited the contraction. In contrast, LES showed the relaxation effect. EXT had no effect.
We examined the effect of extracts when the maximum contraction was 100%. There was no effect on its own self like esophagus and LES. So, the response of acetylcholine on the contraction and relaxation was expressed as a percentage.
We confirmed statistically significant vasoconstriction and reflective heart rate lowering effects using adrenergic alpha-1 agonist phenylephrine. When phenylephrine (10 µg/kg i.v.) was administered, the mean blood pressure was increased in 5~10 min and recovered and sustained for several hours (Fig. 6). With regards to heart rate, after administering the same amount of phenylephrine, it decreased and after 5~10 min, it recovered and sustained itself for several hours (Fig. 7). There were no changes in respiration rates after administration of phenylephrine (Fig. 8).
Although medicinal plants may produce several biological activities in humans, generally very little is known about their toxicity and the same applies for QGC EXT. Safety should be the overriding criterion in the selection of medicinal plants for use in healthcare systems [15]. One should, in addition to the use of historical documentation on QGC EXT, also have formal toxicological evaluation of this plant to optimize its safe use as a medicine.
QGC showed potent efficacy on the development of reflux esophagitis and indomethacin-induced gastritis, by the inhibition of gastric secretion and the prevention of oxidative stress [16]. Flavonoids also have gastric antisecretory activity [17,18]. It has been reported that flavonoid compound inhibits gastric H+, K+-ATPase where the inhibition was competitive with respect to ATP [19,20] described the gastric antisecretory activity is as effective as cimetidine in reducing gastric acid secretion. Glucuronide flavonoids have antiulcer and gastroprotective activity. It appeared to have antiulcerogenic properties in rats and guinea pigs; such properties appeared to be of interest with respect to the adverse effect of gastric ulceration, which develops commonly in subjects taking anti-inflammatory drugs [21]. It has been shown that apigenin blocked cytokine-induced expression of intercellular adhesion molecule-1 [22,23] vascular cell adhesion molecule-1, and E-selectin on human endothelial cells.
Quercetin is a natural flavone with various bioactivities. Quercetin was found to be highly efficient at scavenging free radicals in cell-free systems [24] and to be more active in this respect than the traditional antioxidants vitamins C and E [25,26] found that flavonoids, especially an apigenin, blocked the cytokine-induced expressions of intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), and E-selectin on human endothelial cells [27]. Quercetin was also found to be an active anti-inflammatory agent in a rat paw carrageenan model and to reduce contact sensitivity in mice. In a study on gastric secretion, the oral administration of QGC reduced gastric content significantly and dose-dependently, and when QGC inhibited gastric acid output, the flavonoids extensively prevented the development of reflux esophagitis. These results suggest that QGC has inhibitory effects on reflux esophagitis and gastritis in rats, and our findings support the antiulcer, gastroprotective, and gastric anti-secretory activities of QGC. Furthermore, in feline esophageal epithelial cells, QGC was found to have a protective effect on ethanol induced cell damage by inhibiting ROS generation, activation downstream of ERK and downstream signal transduction induced by interleukin-1 [28].
In this study, the extract of QGC was found to be non-toxic in mice and rats when administered orally in doses up to 5 gm/kg. Based on the classification of Loomis and Hayes [29] viz. that substances with LD50 between 500 and 5,000 and between 5,000 and 15,000 mg/kg bodyweight are regarded as being slightly toxic and practically non-toxic, respectively, the present results suggested that GQC EET safety falls between these 2 categories.
There were no effects on the central nervous system, cardiovascular system, gastrointestinal system, and respiratory system. From the data found in this experiment we conclude that QGC EXT did not induce any adverse effects in experimental animals.
Figures and Tables
 | Fig. 1The effect of QGC EXT on the weight (A), feed intake (B), and water intake (C) in rats. Both male and female used 0.5, 1, 2, 3, 5 g/kg in each dose, respectively. Any changes were not found between the treated and control groups. |
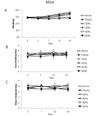 | Fig. 2The effect of QGC EXT on the weight (A), feed intake (B), and water intake (C) in mice. Both male and female used 0.5, 1, 2, 3, 5 g/kg in each dose, respectively. No changes were found among the treated and control groups. |
 | Fig. 3The effect of QGC EXT on esophageal (A) and lower esophageal smooth muscle (B) in cats. The muscle strips were stretched 2.5 g. to bring them to near condition of optimal force development and equilibrated. The contraction did not occur in the dose of QGC EXT 0.01, 0.03, or 0.05 mg/ml. The contraction occurred in the dose of acetylcholine (10-5 M) in esophageal smooth muscle and LES. |
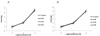 | Fig. 4The dose response curve of acetylcholine on esophageal muscle (A) and lower esophageal sphinctor (B) in cats. Concentration-response curves of acetylcholine were obtained either in the absence and presence of QGC EXT. Treatment of acetylcholine with 10-7, 10-6, 10-5 M to esophageal smooth muscle and LES, the concentration-dependent contraction was exhibited. There was no change by treatment groupo with doses of 0.01, 0.03, 0.05 mg/ml QGC EXT. |
 | Fig. 5The time course of Ach 10-7 M (A), and QGC EXT (B) effect of esophageal smooth muscle of cats. When treated 10-7 M by acetylcholine, the maximum contraction occurred at 30 seconds, and then maintained itself during 20 min. QGC EXT itself did not produce the contraction. |
 | Fig. 6The effect GQC EXT on the changes of mean blood pressure in rats. When administration of QGC was given at doses of 10, 30, 50 mg, there was no change in mean blood pressure. When phenylephrine 10 µg/kg was administered the mean blood pressure increased in 10 minutes and recovered, sustaining itself for several hours. *p<0.05 vs. control (-30 min). |
 | Fig. 7The effect GQC EXT on the changes of heart rates in rat. When administration of QGC was given at doses of 10, 30, 50 mg, there was no change in mean heart rate. When phenylephrine 10 µg/kg was administered, the mean heart rate decreased in 10 minutes and sustained itself for several hours afterwards. *p<0.05 vs. control (-30 min). |
 | Fig. 8The effect of GQC EXT on the changes of respiration rate. When administration of QGC was given at doses of 10, 30, 50 mg, there was no change in respiratory rate in the presence or absence of phenylephrine (10 µg/kg). |
ACKNOWLEDGEMENTS
This study was supported by a grant of the Traditional Korean Medicine R&D project, Ministry for Health & Welfare, Republic of Korea (no. B090062).
References
1. Haenen GR, Paquay JB, Korthouwer RE, Bast A. Peroxynitrite scavenging by flavonoids. Biochem Biophys Res Commun. 1997. 236:591–593.
2. Diplock AT. Defense against reactive oxygen species. Free Radic Res. 1998. 29:463–467.
3. Heijnen CG, Haenen GR, van Acker FA, van der Vijgh WJ, Bast A. Flavonoids as peroxynitrite scavengers: the role of the hydroxyl groups. Toxicol In Vitro. 2001. 15:3–6.
4. Min YS, Lee SE, Hong ST, Kim HS, Choi BC, Sim SS, Whang WK, Sohn UD. The Inhibitory Effect of Quercetin-3-O-beta-D-Glucuronopyranoside on Gastritis and Reflux Esophagitis in Rats. Korean J Physiol Pharmacol. 2009. 13:295–300.
5. Yan XM, Joo MJ, Lim JC, Whang WK, Sim SS, Im C, Kim HR, Lee SY, Kim IK, Sohn UD. The effect of quercetin-3-O-β-D-glucuronopyranoside on indomethacin-induced gastric damage in rats via induction of mucus secretion and down-regulation of ICAM-1 expression. Arch Pharm Res. 2011. 34:1527–1534.
6. Irwin S. Comprehensive observational assessment: Ia. A systematic, quantitative procedure for assessing the behavioral and physiologic state of the mouse. Psychopharmacologia. 1968. 13:222–257.
7. Lee EB. Pharmacological approach of crude drugs. Yakhak Hoeji. 1975. 19:53–59.
8. Dunham NW, Miya TS, Eewards LD. The pharmacological activity of a series of basic esters of mono- and dialkylmalonic acids. J Am Pharm Assoc Am Pharm Assoc (Baltim). 1957. 46:64–66.
9. Koster R, Anderson M, de Beer EJ. Acetic acid for analgesic. 210 screening. Fed Proc. 1959. 18:412–418.
10. Randall LO, Selitto JJ. A method for measurement of analgesic activity on inflamed tissue. Arch Int Pharmacodyn Ther. 1957. 111:409–419.
11. Araki Y, Ueki S. Changes in sensitivity to convulsion in mice with olfactory bulb ablation. Jpn J Pharmacol. 1972. 22:447–456.
12. Swinyard EA, Brown WC, Goodman LS. Comparative assays of antiepileptic drugs in mice and rats. J Pharmacol Exp Ther. 1952. 106:319–330.
13. Sohn UD, Harnett KM, De Petris G, Behar J, Biancani P. Distinct muscarinic receptors, G proteins and phospholipases in esophageal and lower esophageal sphincter circular muscle. J Pharmacol Exp Ther. 1993. 267:1205–1214.
14. Biancani P, Hillemeier C, Bitar KN, Makhlouf GM. Contraction mediated by Ca2+ influx in esophageal muscle and by Ca2+ release in the LES. Am J Physiol. 1987. 253:G760–G766.
15. Tomlinson TM, Akerele O. Medicinal plants their role in health and biodiversity. 1998. Philadelphia: University of Pennsylvania Press.
16. Sohn UD, Cho JH, Song HJ, Sun YH, Hwang WK. Protective effects of Quercetin-3-O-β-D-glucuronopyranoside (QGC) on ethanol-induced cell damage involve inhibitions of ROS generation and downstream activation of the ERK in feline esophageal epithelial cells. M1903. 2009. In : 2009 Digestive Disease Week; May 30th-June 4th; McCormick Place, Chicago, IL, USA.
17. Alvarez A, Pomar F, Sevilla , Montero MJ. Gastric antisecretory and antiulcer activities of an ethanolic extract of Bidens pilosa L. var. radiata Schult. Bip. J Ethnopharmacol. 1999. 67:333–340.
18. Bell NJ, Burget D, Howden CW, Wilkinson J, Hunt RH. Appropriate acid suppression for the management of gastro-oesophageal reflux disease. Digestion. 1992. 51:Suppl 1. 59–67.
19. Murakami S, Muramatsu M, Otomo S. Inhibition of gastric H+, K(+)-ATPase by quercetin. J Enzyme Inhib. 1992. 5:293–298.
20. Parmar NS, Hennings G. The gastric antisecretory activity of 3-methoxy-5,7,3'4'-tetrahydroxyflavan (ME)--a specific histidine decarboxylase inhibitor in rats. Agents Actions. 1984. 15:143–145.
21. Gambhir SS, Goel RK, Das Gupta G. Anti-inflammatory & anti-ulcerogenic activity of amentoflavone. Indian J Med Res. 1987. 85:689–693.
22. Gerritsen ME, Carley WW, Ranges GE, Shen CP, Phan SA, Ligon GF, Perry CA. Flavonoids inhibit cytokine-induced endothelial cell adhesion protein gene expression. Am J Pathol. 1995. 147:278–292.
23. Panés J, Gerritsen ME, Anderson DC, Miyasaka M, Granger DN. Apigenin inhibits tumor necrosis factor-induced intercellular adhesion molecule-1 upregulation in vivo. Microcirculation. 1996. 3:279–286.
24. Rice-Evans CA, Miller NJ, Paganga G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic Biol Med. 1996. 20:933–956.
25. Gordon MH, Roedig-Penman A. Antioxidant activity of quercetin and myricetin in liposomes. Chem Phys Lipids. 1998. 97:79–85.
26. Ishige K, Schubert D, Sagara Y. Flavonoids protect neuronal cells from oxidative stress by three distinct mechanisms. Free Radic Biol Med. 2001. 30:433–446.
27. Gerritsen ME, Carley WW, Ranges GE, Shen CP, Phan SA, Ligon GF, Perry CA. Flavonoids inhibit cytokine-induced endothelial cell adhesion protein gene expression. Am J Pathol. 1995. 147:278–292.
28. Lee SE, Jang HS, Song HJ, Hwang WK, Sohn UD. M1904 Downstream Signal Transduction Induced By Interleukin-1 Beta-Stimulated ROS Generation and Anti-Oxidative Effects of Quercetin-3-O-[beta]-D-Glucuronopyranoside (QGC) in Feline Esophageal Epithelial Cell. Gastroenterology. 2009. 136:A442–A443.
29. Loomis TA, Hayes AW. Loomis's essentials of toxicology. 1996. 4th ed. California: Academic Press.




 PDF
PDF ePub
ePub Citation
Citation Print
Print













 XML Download
XML Download