ABBREVIATIONS
BSA
DPH
PBS
Py-3-Py
RET
SPMV
SPMVTL
SPMVPL
TNBS
INTRODUCTION
METHODS
Materials
Liposomes preparation and TNBS labeling
Fluorescence measurements
Effect of lidocaine·HCl on the structure of the individual monolayers of SPMV, SPMVTL and SPMVPL: selective quenching of Py-3-Py
Determination of annular lipid fluidity in SPMV
Determination of protein distribution in the SPMV llipid bilayers
Effect of lidocaine·HCl on the rotational mobility of bulk SPMV, SPMVTL and SPMVPL
Effect of lidocaine·HCl on the separate monolayers of SPMV, SPMVTL and SPMVPL: selective quenching of DPH
RESULTS
The purity of SPMV
Effect of lidocaine·HCl on the rate and range of lateral mobility in SPMV, SPMVTL and SPMVPL bulk bilayer
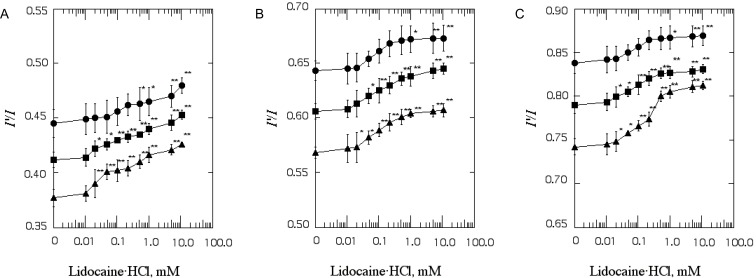 | Fig. 1The effect of lidocaine·HCl on excimer to monomer fluorescence intensity ratio (I'/I) of Py-3-Py in SPMV (A), SPMVTL (B) and SPMVPL (C). The excitation wavelength of Py-3-Py was 330 nm and the I'/I values were calculated from the 480 nm to 379 nm signal ratio. SPMV was treated±4mM TNBS, pH 8.5, at 4℃ for 80 min. SPMVTL and SPMVPL were treated±0.5 mM TNBS, pH 8.5, at 4℃ for 20 min. Py-3-Py was incorporated into SPMV, SPMVTL and SPMVPL and fluorescence measurements were performed at 37℃ (pH 7.4). Untreated (inner and outer monolayers, ▪); TNBS treated (inner monolayer, ▴); calculated for outer monolayer (•) by eq. 3 as described in Materials and Methods. Each point represents the mean±SEM of 5 determinations. An asterisk and double asterisks signify p<0.05 and p<0.01, respectively, compared to control by Student's t-test. |
Effect of lidocaine·HCl on the rate and range of transbilayer asymmetric lateral mobility of SPMV, SPMVTL and SPMVPL monolayers
Effect of lidocaine·HCl on annular lipid fluidity in the SPMV lipid bilayers
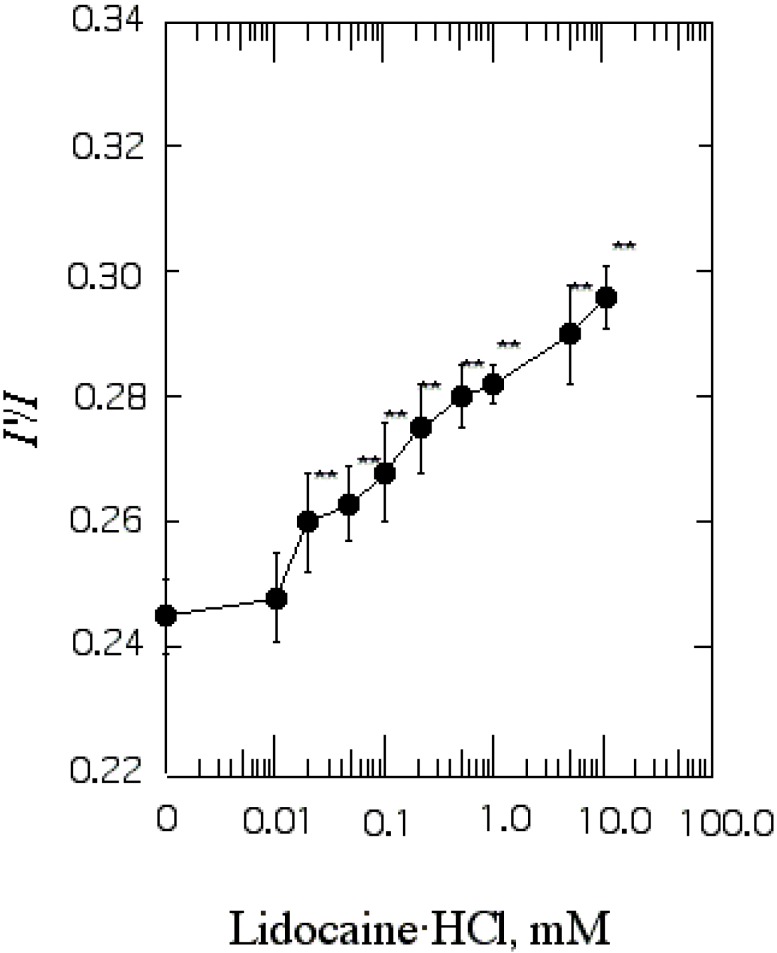 | Fig. 2The effect of lidocaine·HCl on annular lipid fluidity in SPMV. Py-3-Py was excited through RET from tryptophan (excitation wavelength, 286 nm) and the excimer to monomer fluorescence intensity ratio (I'/I) was calculated from the 480 nm to 379 nm signal ratio. Fluorescence measurements were performed at 37℃ (pH 7.4). Each point represents the mean±SEM of 5 determinations. An asterisk and double asterisks signify p<0.05 and p<0.01, respectively, compared to control by Student's t-test. |
Effect of lidocaine·HCl on protein distribution in SPMV
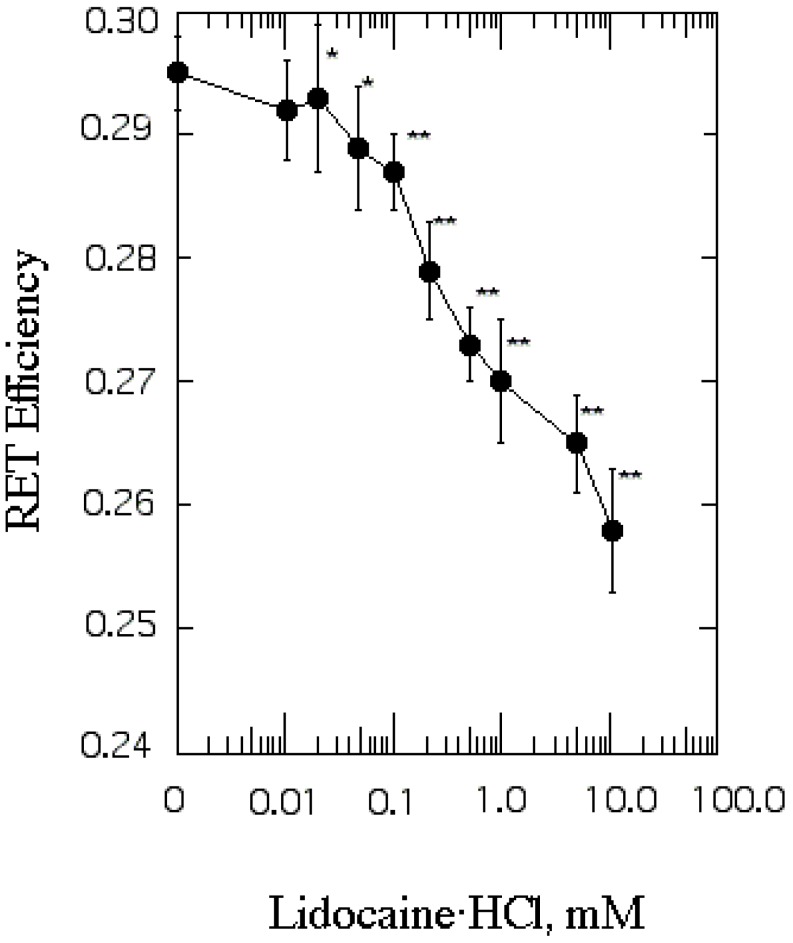 | Fig. 3The effect of lidocaine·HCl on protein distribution in SPMV. Efficiency of RET from tryptophan to Py-3-Py was taken as a measure of protein clustering and calculated by eq. 4. Fluorescence measurements were performed at 37℃ (pH 7.4). Each point represents the mean±SEM of 5 determinations. An asterisk and double asterisks signify p<0.05 and p<0.01, respectively, compared to control by Student's t-test. |
Effect of lidocaine·HCl on the range of rotational mobility of SPMV, SPMVTL and SPMVPL bulk bilayer
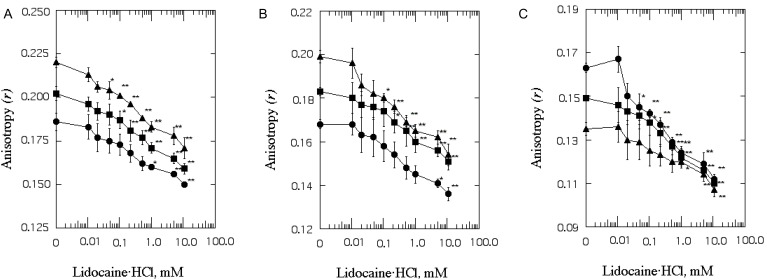 | Fig. 4The effect of lidocaine·HCl on the anisotropy (r) of DPH in SPMV (A), SPMVTL (B) and SPMVPL (C). The excitation wavelength for DPH was 362 nm and fluorescence emission was read at 424 nm. SPMV was treated±2 mM TNBS, pH 8.5, at 4℃ for 40 min. SPMVTL and SPMVPL were treated±0.5 mM TNBS, pH 8.5, at 4℃ for 20 min. DPH was incorporated into SPMV, SPMVTL and SPMVPL and fluorescence measurements were performed at 37℃ (pH 7.4). Untreated (inner and outer monolayers, ▪); TNBS treated (inner monolayer, ▴); calculated for outer monolayer (•) by eq. 7 as described in Materials and Methods. Each point represents the mean±SEM of 5 determinations. An asterisk and double asterisks signify p<0.05 and p<0.01, respectively, compared to control by Student's t-test. |
Effect of lidocaine·HCl on the range of transbilayer asymmetric rotational mobility of SPMV monolayers
Table 1
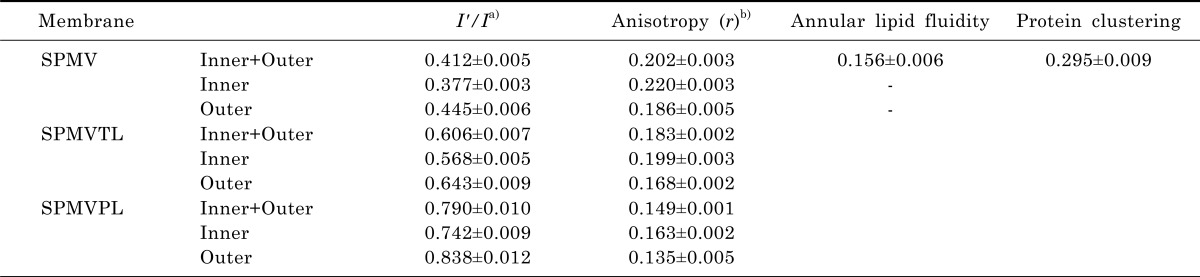
a)SPMV was treated±4 mM TNBS, pH 8.5, at 4℃ for 80 min (I'/I). SPMVTL and SPMVPL were treated±0.5 mM TNBS, pH 8.5, at 4℃ for 20 min (I'/I). Py-3-Py was incorporated and fluorescence measurements were performed at 37℃ (pH 7.4); b)SPMV was treated±2 mM TNBS, pH 8.5, at 4℃ for 40 min (anisotropy). SPMVTL and SPMVPL were treated±0.5 mM TNBS, pH 8.5, at 4℃ for 20 min (anisotropy). DPH was incorporated and fluorescence measurements were performed at 37℃ (pH 7.4).
Values from untreated membranes represent inner+outer monolayer; Values from TNBS treated membranes represent the inner monolayer; Values for the outer monolayer were calculated as described in Materials and Methods. Values are represented as the mean±SEM of 5 determinations.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


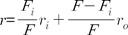
 XML Download
XML Download