1. Bloomfield SA, Völgyi B. The diverse functional roles and regulation of neuronal gap junctions in the retina. Nat Rev Neurosci. 2009; 10:495–506. PMID:
19491906.

2. Werblin FS, Dowling JE. Organization of the retina of the mudpuppy, Necturus maculosus. II. Intracellular recording. J Neurophysiol. 1969; 32:339–355. PMID:
4306897.

3. Kaneko A. Receptive field organization of bipolar and amacrine cells in the goldfish retina. J Physiol. 1973; 235:133–153. PMID:
4778132.

4. Naka KI, Nye PW. Role of horizontal cells in organization of the catfish retinal receptive field. J Neurophysiol. 1971; 34:785–801. PMID:
5097157.

5. Marchiafava PL. Horizontal cells influence membrane potential of bipolar cells in the retina of the turtle. Nature. 1978; 275:141–142. PMID:
692683.

6. Arai I, Tanaka M, Tachibana M. Active roles of electrically coupled bipolar cell network in the adult retina. J Neurosci. 2010; 30:9260–9270. PMID:
20610761.

7. Cook JE, Becker DL. Gap junctions in the vertebrate retina. Microsc Res Tech. 1995; 31:408–419. PMID:
8534902.

8. Marc RE, Liu WL, Muller JF. Gap junctions in the inner plexiform layer of the goldfish retina. Vision Res. 1988; 28:9–24. PMID:
3414003.

9. Saito T, Kujiraoka T. Characteristics of bipolar-bipolar coupling in the carp retina. J Gen Physiol. 1988; 91:275–287. PMID:
3373179.
10. DeVries SH, Schwartz EA. Modulation of an electrical synapse between solitary pairs of catfish horizontal cells by dopamine and second messengers. J Physiol. 1989; 414:351–375. PMID:
2558170.

11. DeVries SH, Schwartz EA. Hemi-gap-junction channels in solitary horizontal cells of the catfish retina. J Physiol. 1992; 445:201–230. PMID:
1380084.

12. Lu C, McMahon DG. Modulation of hybrid bass retinal gap junctional channel gating by nitric oxide. J Physiol. 1997; 499:689–699. PMID:
9130165.

13. Lasater EM, Dowling JE. Dopamine decreases conductance of the electrical junctions between cultured retinal horizontal cells. Proc Natl Acad Sci USA. 1985; 82:3025–3029. PMID:
3857632.

14. Piccolino M, Neyton J, Gerschenfeld HM. Decrease of gap junction permeability induced by dopamine and cyclic adenosine 3':5'-monophosphate in horizontal cells of turtle retina. J Neurosci. 1984; 4:2477–2488. PMID:
6092564.

15. Hedden WL Jr, Dowling JE. The interplexiform cell system. II. Effects of dopamine on goldfish retinal neurones. Proc R Soc Lond B Biol Sci. 1978; 201:27–55. PMID:
27790.
16. Murakami M, Miyachi EI, Takahadhi KI. Modulation of gap juctions between horizontal cells by second messengers. Prog Retinal Eye Res. 1995; 14:197–221.
17. McMahon DG, Brown DR. Modulation of gap-junction channel gating at zebrafish retinal electrical synapses. J Neurophysiol. 1994; 72:2257–2268. PMID:
7533830.

18. Baldridge WH, Ball AK, Miller RG. Dopaminergic regulation of horizontal cell gap junction particle density in goldfish retina. J Comp Neurol. 1987; 265:428–436. PMID:
3693614.

19. Stell WK. Inputs to bipolar cell dendrites in goldfish retina. Sens Processes. 1978; 2:339–349. PMID:
755290.
20. Saito T, Kondo H, Toyoda J. Rod and cone signals in the on-center bipolar cell: their different ionic mechanisms. Vision Res. 1978; 18:591–595. PMID:
664343.

21. Malchow RP, Yazulla S. Separation and light adaptation of rod and cone signals in the retina of the goldfish. Vision Res. 1986; 26:1655–1666. PMID:
3617507.

22. Hughes A, Saszik S, Bilotta J, Demarco PJ Jr, Patterson WF 2nd. Cone contributions to the photopic spectral sensitivity of the zebrafish ERG. Vis Neurosci. 1998; 15:1029–1037. PMID:
9839967.

23. Hood DC, Finkelstein MA. Boff KR, Kaufman L, Thomas JP, editors. Sensitivity to light. Handbook of Perception and Human Performance. 1986. New York: Wiley;p. 5-1–5-66.
24. Kim SH, Jung CS. The role of the pattern edge in goldfish visual motion detection. Korean J Physiol Pharmacol. 2010; 14:413–417. PMID:
21311683.

25. Liu CR, Xu L, Zhong YM, Li RX, Yang XL. Expression of connexin 35/36 in retinal horizontal and bipolar cells of carp. Neuroscience. 2009; 164:1161–1169. PMID:
19778581.

26. O'Brien J, al-Ubaidi MR, Ripps H. Connexin 35: a gap-junctional protein expressed preferentially in the skate retina. Mol Biol Cell. 1996; 7:233–243. PMID:
8688555.
27. Condorelli DF, Parenti R, Spinella F, Trovato Salinaro A, Belluardo N, Cardile V, Cicirata F. Cloning of a new gap junction gene (Cx36) highly expressed in mammalian brain neurons. Eur J Neurosci. 1998; 10:1202–1208. PMID:
9753189.

28. Veruki ML, Hartveit E. Meclofenamic acid blocks electrical synapses of retinal AII amacrine and on-cone bipolar cells. J Neurophysiol. 2009; 101:2339–2347. PMID:
19279153.

29. Pan F, Mills SL, Massey SC. Screening of gap junction antagonists on dye coupling in the rabbit retina. Vis Neurosci. 2007; 24:609–618. PMID:
17711600.

30. Tomita T, Yanagida T. Origins of the ERG waves. Vision Res. 1981; 21:1703–1707. PMID:
7336605.

31. Koriyama Y, Yasuda R, Homma K, Mawatari K, Nagashima M, Sugitani K, Matsukawa T, Kato S. Nitric oxide-cGMP signaling regulates axonal elongation during optic nerve regeneration in the goldfish in vitro and in vivo. J Neurochem. 2009; 110:890–901. PMID:
19457064.
32. Villani L, Guarnieri T. Localization of nitric oxide synthase in the goldfish retina. Brain Res. 1996; 743:353–356. PMID:
9017268.

33. Liepe BA, Stone C, Koistinaho J, Copenhagen DR. Nitric oxide synthase in Müller cells and neurons of salamander and fish retina. J Neurosci. 1994; 14:7641–7654. PMID:
7527846.
34. Yazulla S, Lin ZS. Differential effects of dopamine depletion on the distribution of [
3H]SCH 23390 and [
3H]spiperone binding sites in the goldfish retina. Vision Res. 1995; 35:2409–2414. PMID:
8594810.
35. Harsanyi K, Mangel SC. Activation of a D2 receptor increases electrical coupling between retinal horizontal cells by inhibiting dopamine release. Proc Natl Acad Sci USA. 1992; 89:9220–9224. PMID:
1357661.

36. Van Buskirk R, Dowling JE. Isolated horizontal cells from carp retina demonstrate dopamine-dependent accumulation of cyclic AMP. Proc Natl Acad Sci USA. 1981; 78:7825–7829. PMID:
6278491.

37. Lasater EM. Retinal horizontal cell gap junctional conductance is modulated by dopamine through a cyclic AMP-dependent protein kinase. Proc Natl Acad Sci USA. 1987; 84:7319–7323. PMID:
2823257.

38. Huppé-Gourgues F, Coudé G, Lachapelle P, Casanova C. Effects of the intravitreal administration of dopaminergic ligands on the b-wave amplitude of the rabbit electroretinogram. Vision Res. 2005; 45:137–145. PMID:
15581915.

39. Hedden WL Jr, Dowling JE. The interplexiform cell system. II. Effects of dopamine on goldfish retinal neurones. Proc R Soc Lond B Biol Sci. 1978; 201:27–55. PMID:
27790.
40. Dowling JE, Ehinger B. Synaptic organization of the dopaminergic neurons in the rabbit retina. J Comp Neurol. 1978; 180:203–220. PMID:
207745.

41. Dowling JE, Ehinger B. The interplexiform cell system. I. Synapses of the dopaminergic neurons of the goldfish retina. Proc R Soc Lond B Biol Sci. 1978; 201:7–26. PMID:
27792.
42. Hampton CK, Redburn DA. Autoradiographic analysis of 3H-glutamate, 3H-dopamine, and 3H-GABA accumulation in rabbit retina after kainic acid treatment. J Neurosci Res. 1983; 9:239–251. PMID:
6133958.
43. Yazulla S, Zucker CL. Synaptic organization of dopaminergic interplexiform cells in the goldfish retina. Vis Neurosci. 1988; 1:13–29. PMID:
2908724.

44. Djamgoz MB, Wagner HJ. Localization and function of dopamine in the adult vertebrate retina. Neurochem Int. 1992; 20:139–191. PMID:
1304857.

45. Negishi K, Teranishi T, Kato S. The dopamine system of the teleost fish retina. Prog Ret Res. 1990; 9:1–48.
46. Ribelayga C, Cao Y, Mangel SC. The circadian clock in the retina controls rod-cone coupling. Neuron. 2008; 59:790–801. PMID:
18786362.

47. Shen Y, Zhang AJ, Yang XL. Uncoupling of horizontal cells alters the receptive fields of retinal bipolar cells. Neuroreport. 2003; 14:2159–2162. PMID:
14625439.

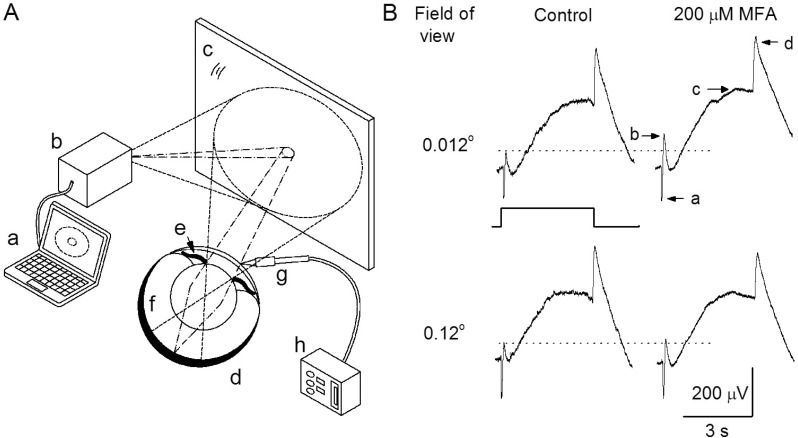
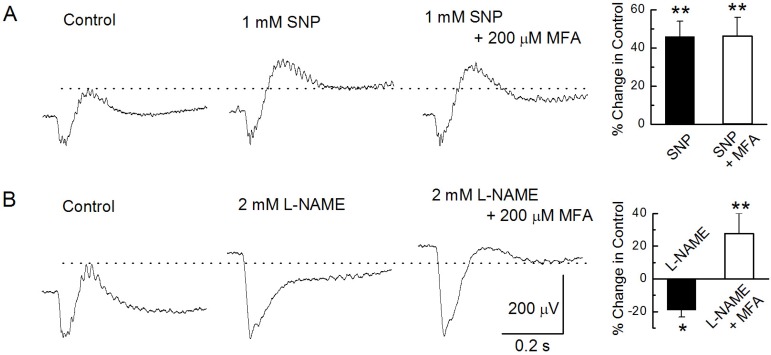
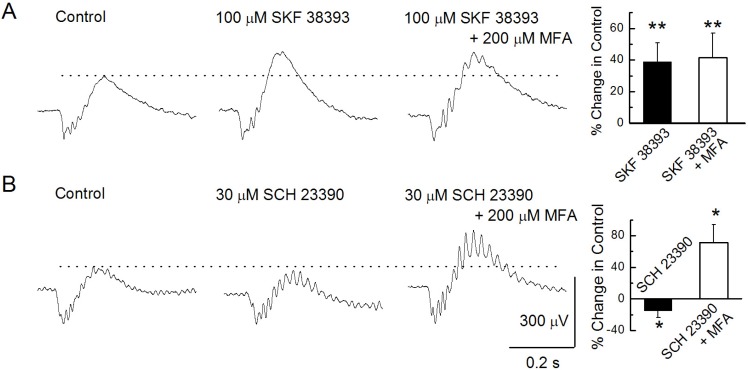
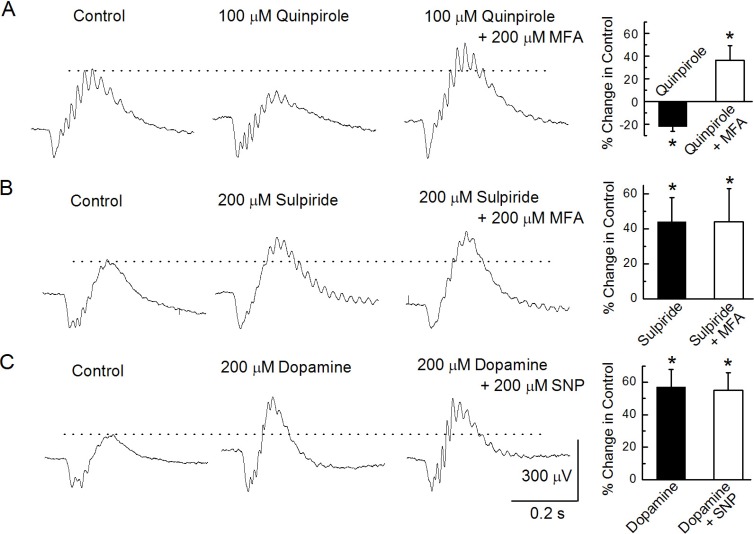




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download