Abstract
The flavonoid quercetin is a low molecular weight compound generally found in apple, gingko, tomato, onion and other red-colored fruits and vegetables. Like other flavonoids, quercetin has diverse pharmacological actions. However, relatively little is known about the influence of quercetin effects in the regulation of ligand-gated ion channels. Previously, we reported that quercetin regulates subsets of nicotinic acetylcholine receptors such as α3β4, α7 and α9α10. Presently, we investigated the effects of quercetin on muscle-type of nicotinic acetylcholine receptor channel activity expressed in Xenopus oocytes after injection of cRNA encoding human fetal or adult muscle-type of nicotinic acetylcholine receptor subunits. Acetylcholine treatment elicited an inward peak current (IACh) in oocytes expressing both muscle-type of nicotinic acetylcholine receptors and co-treatment of quercetin with acetylcholine inhibited IACh. Pre-treatment of quercetin further inhibited IACh in oocytes expressing adult and fetal muscle-type nicotinic acetylcholine receptors. The inhibition of IACh by quercetin was reversible and concentration-dependent. The IC50 of quercetin was 18.9±1.2 μM in oocytes expressing adult muscle-type nicotinic acetylcholine receptor. The inhibition of IACh by quercetin was voltage-independent and non-competitive. These results indicate that quercetin might regulate human muscle-type nicotinic acetylcholine receptor channel activity and that quercetin-mediated regulation of muscle-type nicotinic acetylcholine receptor might be coupled to regulation of neuromuscular junction activity.
Go to : 
References
1. Jensen ML, Schousboe A, Ahring PK. Charge selectivity of the Cys-loop family of ligand-gated ion channels. J Neurochem. 2005; 92:217–225.

2. Dani JA, Bertrand D. Nicotinic acetylcholine receptors and nicotinic cholinergic mechanisms of the central nervous system. Annu Rev Pharmacol Toxicol. 2007; 47:699–729.

3. Boulter J, Evans K, Goldman D, Martin G, Treco D, Heinemann S, Patrick J. Isolation of a cDNA clone coding for a possible neural nicotinic acetylcholine receptor alpha-subunit. Nature. 1986; 319:368–374.
4. Boulter J, Connolly J, Deneris E, Goldman D, Heinemann S, Patrick J. Functional expression of two neuronal nicotinic acetylcholine receptors from cDNA clones identifies a gene family. Proc Natl Acad Sci USA. 1987; 84:7763–7767.

5. Karlin A. Emerging structure of the nicotinic acetylcholine receptors. Nat Rev Neurosci. 2002; 3:102–114.

6. Gotti C, Clementi F. Neuronal nicotinic receptors: from structure to pathology. Prog Neurobiol. 2004; 74:363–396.

7. Boorman JP, Groot-Kormelink PJ, Sivilotti LG. Stoichiometry of human recombinant neuronal nicotinic receptors containing the b3 subunit expressed in Xenopus oocytes. J Physiol. 2000; 529:565–577.
8. Couturier S, Bertrand D, Matter JM, Hernandez MC, Bertrand S, Millar N, Valera S, Barkas T, Ballivet M. A neuronal nicotinic acetylcholine receptor subunit (alpha 7) is developmentally regulated and forms a homo-oligomeric channel blocked by alpha-BTX. Neuron. 1990; 5:847–856.
9. Elgoyhen AB, Johnson DS, Boulter J, Vetter DE, Heinemann S. Alpha 9: an acetylcholine receptor with novel pharmacological properties expressed in rat cochlear hair cells. Cell. 1994; 79:705–715.
10. Gotti C, Hanke W, Maury K, Moretti M, Ballivet M, Clementi F, Bertrand D. Pharmacology and biophysical properties of alpha 7 and alpha 7-alpha 8 alpha-bungarotoxin receptor subtypes immunopurified from the chick optic lobe. Eur J Neurosci. 1994; 6:1281–1291.
11. Mishina M, Takai T, Imoto K, Noda M, Takahashi T, Numa S, Methfessel C, Sakmann B. Molecular distinction between fetal and adult forms of muscle acetylcholine receptor. Nature. 1986; 321:406–411.

12. Karlin A. Emerging structure of the nicotinic acetylcholine receptors. Nat Rev Neurosci. 2002; 3:102–114.

13. Havsteen BH. The biochemistry and medical significance of the flavonoids. Pharmacol Ther. 2002; 96:67–202.

14. Satoh E, Ishii T, Shimizu Y, Sawamura S, Nishimura M. The mechanism underlying the protective effect of the thearubigin fraction of black tea (Camellia sinensis) extract against the neuromuscular blocking action of botulinum neurotoxins. Pharmacol Toxicol. 2002; 90:199–202.

15. Basu S, Chaudhuri T, Chauhan SP, Das Gupta AK, Chaudhury L, Vedasiromoni JR. The theaflavin fraction is responsible for the facilitatory effect of black tea at the skeletal myoneural junction. Life Sci. 2005; 76:3081–3088.

16. Tong JJ. Mitochondrial delivery is essential for synaptic potentiation. Biol Bull. 2007; 212:169–175.

17. Chiang HC, Iijima K, Hakker I, Zhong Y. Distinctive roles of different beta-amyloid 42 aggregates in modulation of synaptic functions. FASEB J. 2009; 23:1969–1977.
18. Moustafa AM, Ahmed SH, Nabil ZI, Hussein AA, Omran MA. Extraction and phytochemical investigation of Calotropis procera: effect of plant extracts on the activity of diverse muscles. Pharm Biol. 2010; 48:1080–1190.
19. Re L, Barocci S, Capitani C, Vivani C, Ricci M, Rinaldi L, Paolucci G, Scarpantonio A, León-Fernández OS, Morales MA. Effects of some natural extracts on the acetylcholine release at the mouse neuromuscular junction. Pharmacol Res. 1999; 39:239–245.

20. Lee BH, Jeong SM, Lee JH, Kim JH, Yoon IS, Lee JH, Choi SH, Lee SM, Chang CG, Kim HC, Han Y, Paik HD, Kim Y, Nah SY. Quercetin inhibits the 5-hydroxytryptamine type 3 receptor-mediated ion current by interacting with pretransmembrane domain I. Mol Cells. 2005; 20:69–73.
21. Lee BH, Lee JH, Yoon IS, Lee JH, Choi SH, Pyo MK, Jeong SM, Choi WS, Shin TJ, Lee SM, Rhim H, Park YS, Han YS, Paik HD, Cho SG, Kim CH, Lim YH, Nah SY. Human glycine alpha1 receptor inhibition by quercetin is abolished or inversed by alpha267 mutations in transmembrane domain 2. Brain Res. 2007; 1161:1–10.
22. Lee BH, Choi SH, Shin TJ, Pyo MK, Hwang SH, Kim BR, Lee SM, Lee JH, Kim HC, Park HY, Rhim H, Nah SY. Quercetin enhances human α7 nicotinic acetylcholine receptor-mediated ion current through interactions with Ca(2+) binding sites. Mol Cells. 2010; 30:245–253.

23. Lee BH, Choi SH, Shin TJ, Pyo MK, Hwang SH, Lee SM, Paik HD, Kim HC, Nah SY. Effects of quercetin on α9α10 nicotinic acetylcholine receptor-mediated ion currents. Eur J Pharmacol. 2011; 650:79–85.

24. Lee BH, Hwang SH, Choi SH, Shin TJ, Kang J, Lee SM, Nah SY. Quercetin inhibits alpha3beta4 nicotinic acetylcholine receptor-mediated ion currents expressed in xenopus oocytes. Korean J Physiol Pharmacol. 2011; 15:17–22.
25. Sine SM, Taylor P. Local anesthetics and histrionicotoxin are allosteric inhibitors of the acetylcholine receptor. Studies of clonal muscle cells. J Biol Chem. 1982; 257:8106–8114.

26. Heidmann T, Oswald RE, Changeux JP. Multiple sites of action for noncompetitive blockers on acetylcholine receptor rich membrane fragments from torpedo marmorata. Biochemistry. 1983; 22:3112–3127.

27. Arias HR. Luminal and non-luminal non-competitive inhibitor binding sites on the nicotinic acetylcholine receptor. Mol Membr Biol. 1996; 13:1–17.

28. Gr⊘nlien JH, Ween H, Thorin-Hagene K, Cassar S, Li J, Briggs CA, Gopalakrishnan M, Malysz J. Importance of M2-M3 loop in governing properties of genistein at the α7 nicotinic acetylcholine receptor inferred from α7/5-HT3A chimera. Eur J Pharmacol. 2010; 647:37–47.
Go to : 
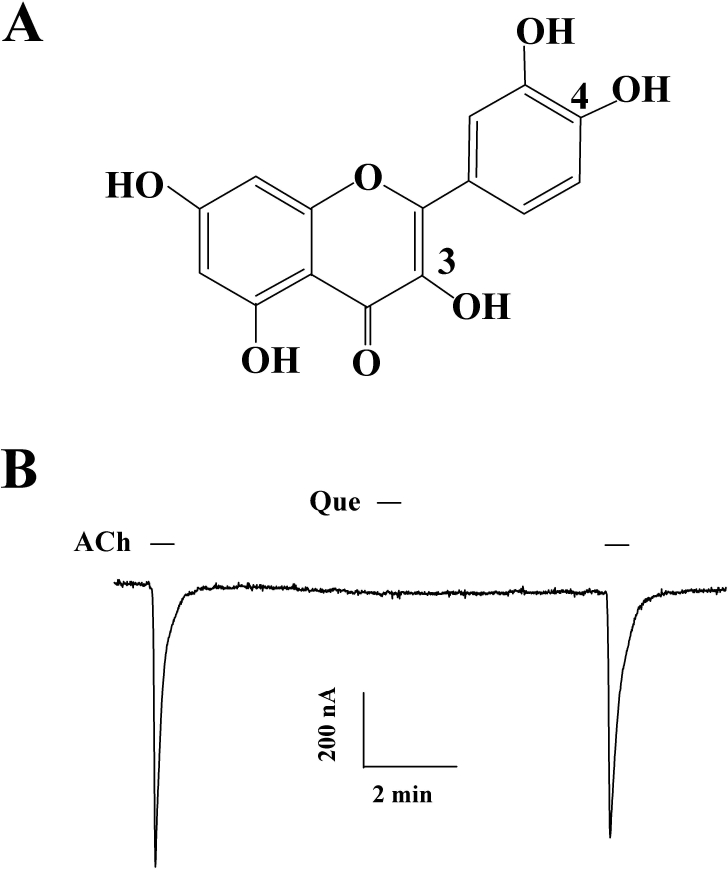 | Fig. 1.Chemical structure of quercetin (A) and its effect in oocytes expressing α1β1δε nicotinic acetylcholine receptors (B). Quercetin had no effect on IACh in oocytes expressing α1β1δε nicotinic acetylcholine receptors. |
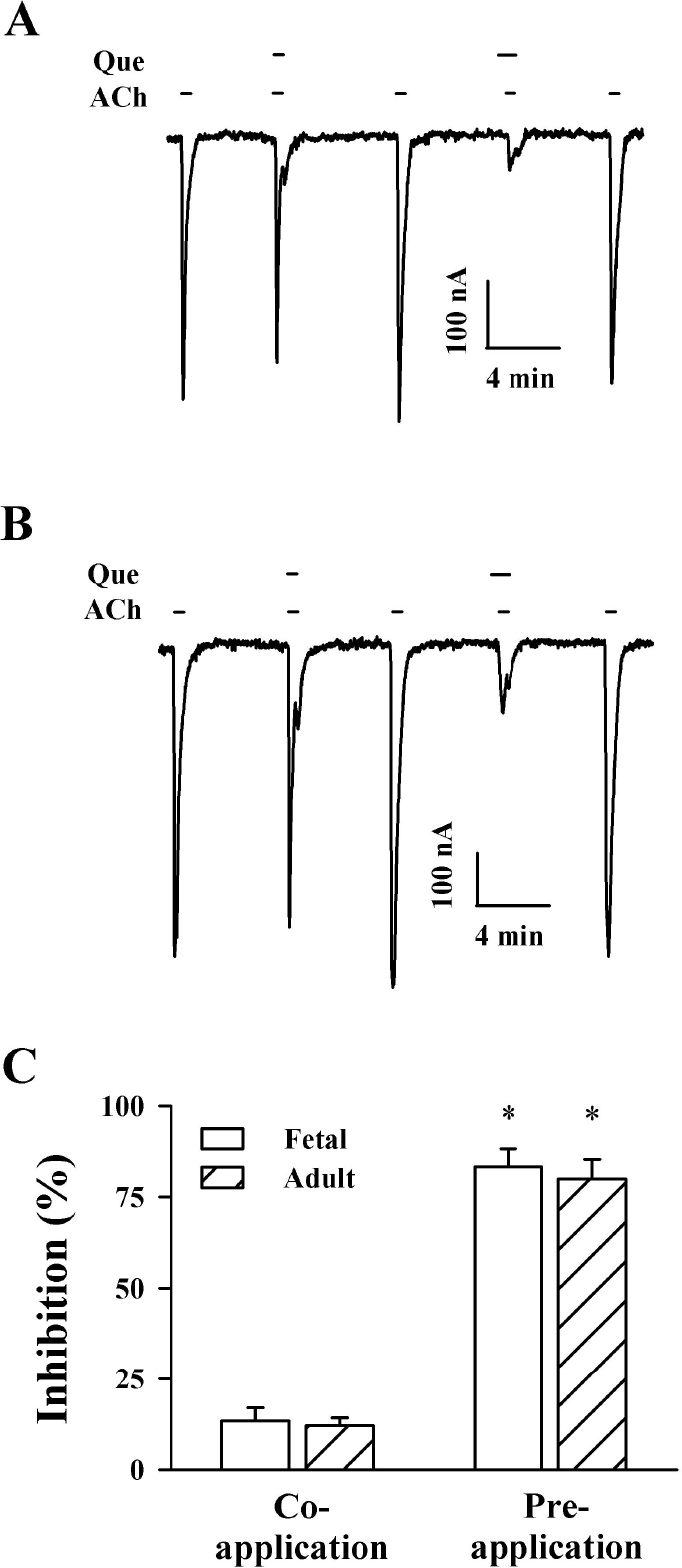 | Fig. 2.Effect of quercetin (Que) on IACh in oocytes expressing α1β1 δγ and α1β1δε muscle-type nicotinic acetylcholine receptors. (A) Acetylcholine (100 μM) was first applied and then acetylcholine was co- or pre-treated with quercetin (100 μM). Thus, co-treatment of quercetin with acetylcholine and pre-treatment of quercetin before acetylcholine application inhibited IACh in oocytes expressing α1β1δγ nicotinic acetylcholine receptors. (B) Acetylcholine (100 μM) was first applied and then acetylcholine was co- or pre-treated with quercetin (100 μM). Thus, co-treatment of quercetin with acetylcholine and pre-treatment of quercetin before acetylcholine application inhibited IACh in oocytes expressing α1β1δε nicotinic acetylcholine receptors. The resting membrane potential of oocytes was about –35 mV and oocytes were voltage-clamped at a holding potential of –80 mV prior to drug application. Traces are representative of six separate oocytes from three different frogs. (C) Summary of % inhibition by quercetin of IACh was calculated from the average of the peak inward current elicited by acetylcholine alone before quercetin and the peak inward current elicited by acetylcholine alone after co- and pre-treatment of quercetin with acetylcholine. Each point represents the mean±S.E.M. (n=9∼12 from three different frogs). |
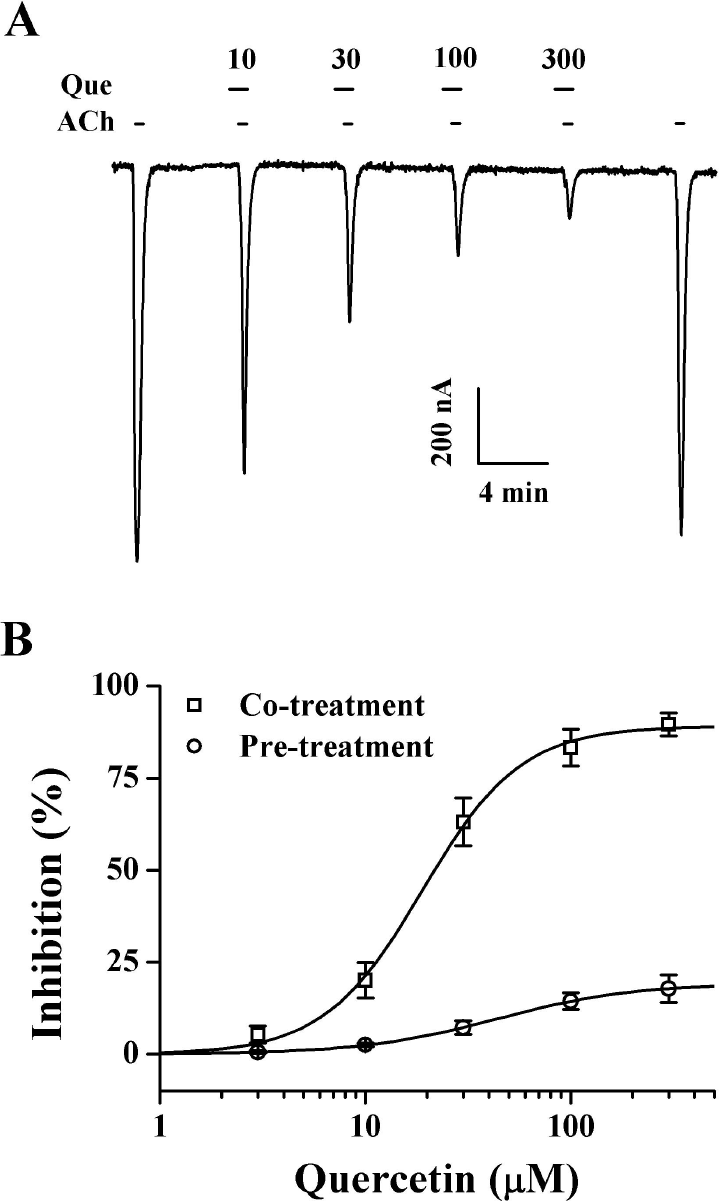 | Fig. 3.Dose-dependent effect of quercetin on IACh in oocytes expressing α1β1δε nicotinic acetylcholine receptors. (A) Acetylcholine (100 μM) was first applied and then acetylcholine was pre-treated with different concentration of quercetin. Thus, co-treatment of quercetin with acetylcholine and pre-treatment of quercetin before acetylcholine application inhibited IACh. The resting membrane potential of oocytes was about –35 mV and oocytes were voltage-clamped at a holding potential of –80 mV prior to drug application. Traces are representative of six separate oocytes from three different frogs. (A) IACh in oocytes expressing α1β1δε nicotinic acetylcholine receptors was elicited at – 80 mV holding potential with indicated time in the presence of 100 μM acetylcholine and then the indicated concentration of quercetin was pre-treated for 30 s before acetylcholine. Traces are representative of 6∼9 separate oocytes from three different frogs. (B) Percent inhibition by quercetin of IACh was calculated from the average of the peak inward current elicited by acetylcholine alone before quercetin and the peak inward current elicited by acetylcholine alone after co-treatment of quercetin with acetylcholine and pre-treatment of quercetin before acetylcholine application. The continuous line shows the curve fitted according to the equation. y/ymax=[Quercetin]/[Quercetin] +K1/2), where ymax, the maximum inhibition (89.1±2.2% and 19.3±1.3 from pre-treatment and co-treatment, respectively, mean±S.E.M.) and K1/2 is the concentration for half-maximum inhibition (18.9±1.3 and 44.3±1.1 μM from pre-treatment and co-treatment, respectively, mean±S.E.M.), and [Quercetin] is the concentration of quercetin. Each point represents the mean±S.E.M. (n=9∼12 from three different frogs). |
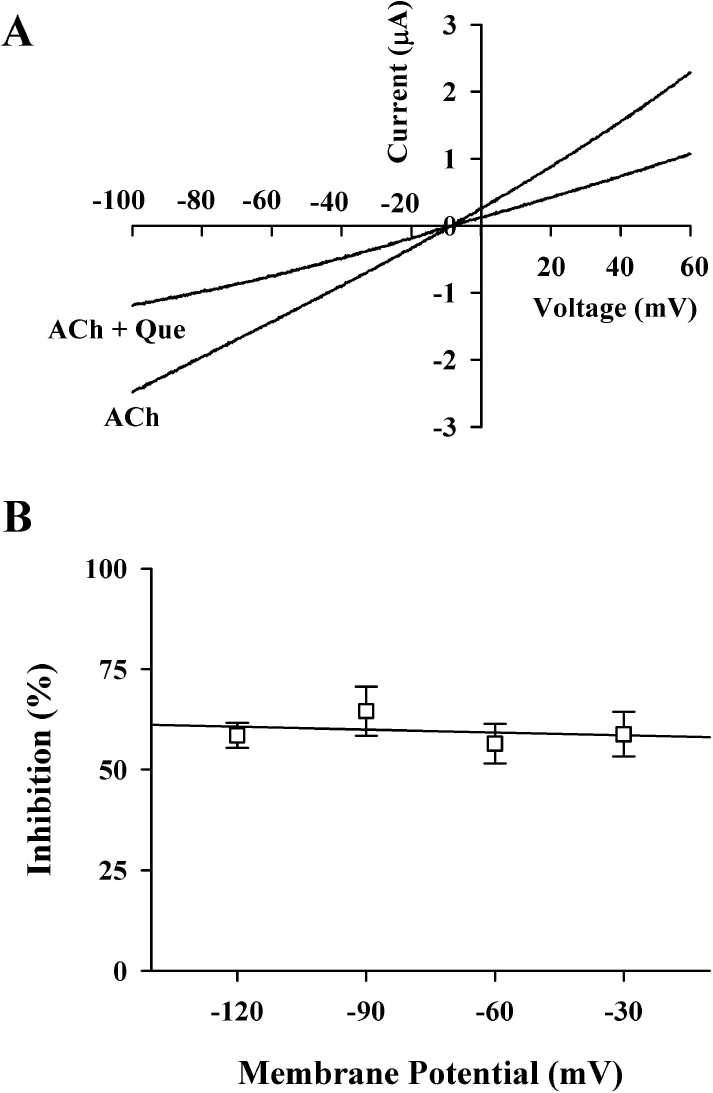 | Fig. 4.Current-voltage relationship and voltage-independent inhibition by quercetin. (A) Current-voltage relationships of IACh inhibition by quercetin in α1β1δε nicotinic acetylcholine receptors. Representative current-voltage relationships were obtained using voltage ramps of –100 to +60 mV for 300 ms at a holding potential of –80 mV. Voltage steps were applied before and after application of 100 μM acetylcholine in the absence or presence of 20 μM quercetin. (B) Voltage-independent inhibition of IACh in the α1β1δε nicotinic acetylcholine receptors by quercetin. The values were obtained from the receptors in the absence or presence of 20 μM quercetin at the indicated membrane holding potentials. |
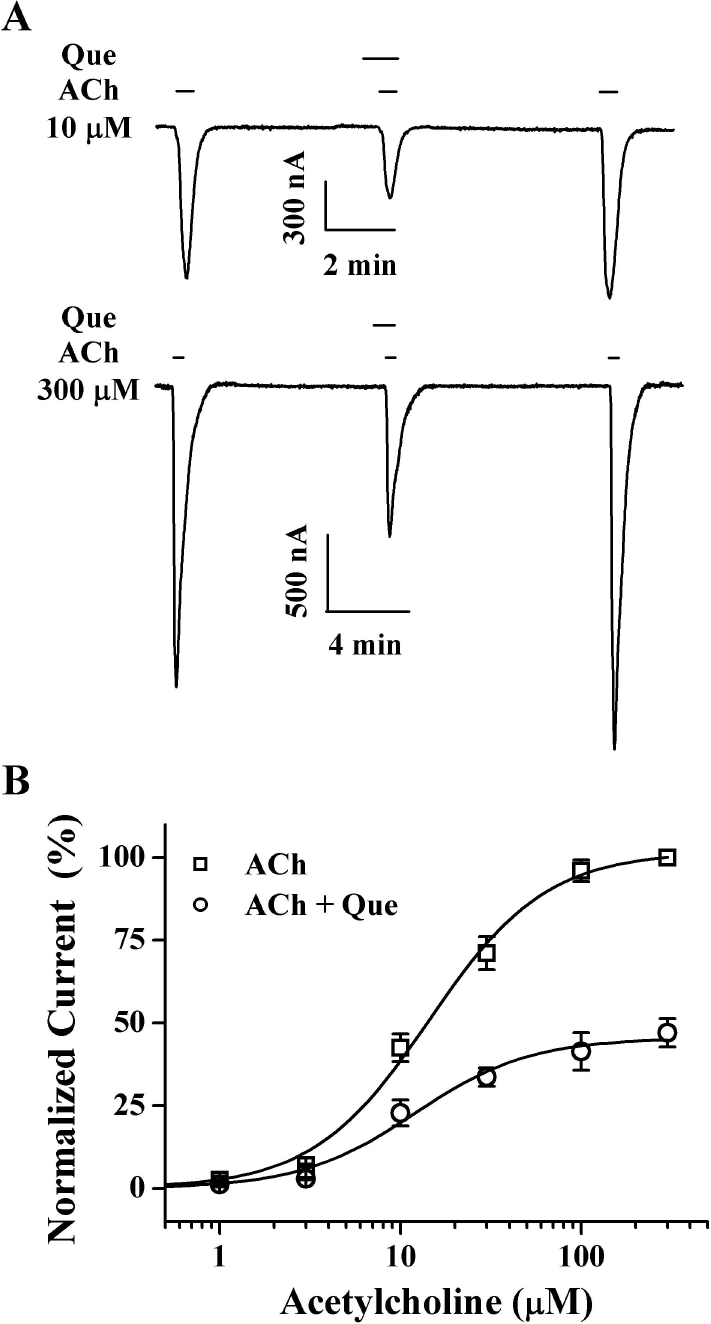 | Fig. 5.Concentration-dependent effects of acetylcholine on quercetin-mediated inhibition of IACh. (A) The representative traces were obtained from α1β1δε nicotinic acetylcholine receptors expressed in oocytes. IACh of the upper and lower panels were elicited from concentration of 10 μM and 300 μM acetylcholine at a holding potential of –80 mV, respectively. (B) Concentration-response relationships for acetylcholine in the α1β1δε nicotinic acetylcholine receptors applied with acetylcholine (1∼300 μM) alone or with acetylcholine plus pre-application of 20 μM quercetin. The IACh of oocytes expressing the α1β1δε nicotinic acetylcholine receptors was measured using the indicated concentration of acetylcholine in the absence (☐) or presence (❍) of 20 μM quercetin. Oocytes were exposed to ACh alone or to ACh with quercetin. Oocytes were voltage-clamped at a holding potential of –80 mV. Each point represents the mean±S.E.M. (n=8 ∼ 12/group). |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download