Abstract
Vascular inflammation process has been suggested to be an important risk factor in the development of atherosclerosis. Recently we reported that induction of peroxisome proliferator-activated receptor-γ (PPAR-γ) selectively inhibits vascular cell adhesion molecule-1 (VCAM-1) but not intercellular cell adhesion molecule-1 (ICAM-1) in tumor necrosis factor (TNF)-α-activated human umbilical vein endothelial cells (HUVEC). In this study, we investigated whether genipin inhibits expression of cellular adhesion molecules, which is relevant to inflammation. Pretreatment with genipin reduced reactive oxygen species (ROS) production and expression of VCAM-1, but not ICAM-1 in TNF-α-activated HUVEC. Genipin dose- and time-dependently increased PPAR-γ expression and inhibited TNF-α-induced phosphorylation of Akt and PKC with different degrees. Finally, genipin prevented TNF-α-induced adhesion of U937 monocytic cells to HUVEC. Taken together, these results indicate that upregualtion of PPAR-γ by genipin selectively inhibits TNF-α-induced expression of VCAM-1, in which regulation of Akt and/or PKC play a key role. We concluded that genipin can be used for the treatment of cardiovascular disorders such as atherosclerosis.
References
1. Ross R. Atherosclerosis–an inflammatory disease. N Engl J Med. 1999; 340:115–126.
2. Fan J, Watanabe T. Inflammatory reactions in the pathogenesis of atherosclerosis. J Atheroscler Thromb. 2003; 10:63–71.

3. Springer TA. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell. 1994; 76:301–314.

4. Modur V, Zimmerman GA, Prescott SM, McIntyre TM. Endothelial cell inflammatory responses to tumor necrosis factor alpha. Ceramide-dependent and -independent mitogen-activated protein kinase cascades. J Biol Chem. 1996; 271:13094–13102.
5. Moon L, Ha YM, Jang HJ, Kim HS, Jun MS, Kim YM, Lee YS, Lee DH, Son KH, Kim HJ, Seo HG, Lee JH, Kim YS, Chang KC. Isoimperatorin, cimiside E and 23-O-acetylshengmanol-3-xyloside from Cimicifugae rhizome inhibit TNF-α-induced VCAM-1 expression in human endothelial cells: involvement of PPAR-γ upregulation and PI3K, ERK1/2, and PKC signal pathways. J Ethnopharmacol. 2011; 133:336–344.

6. Jackson SM, Parhami F, Xi XP, Berliner JA, Hsueh WA, Law RE, Demer LL. Peroxisome proliferator-activated receptor activators target human endothelial cells to inhibit leukocyte-endothelial cell interaction. Arterioscler Thromb Vasc Biol. 1999; 19:2094–2104.

7. Nizamutdinova IT, Jeong JJ, Xu GH, Lee SH, Kang SS, Kim YS, Chang KC, Kim HJ. Hesperidin, hesperidin methyl chalone and phellopterin from Poncirus trifoliata (Rutaceae) differentially regulate the expression of adhesion molecules in tumor necrosis factor-alpha-stimulated human umbilical vein endothelial cells. Int Immunopharmacol. 2008; 8:670–678.
8. Tsoyi K, Kim WS, Kim YM, Kim HJ, Seo HG, Lee JH, Yun-Choi HS, Chang KC. Upregulation of PTEN by CKD712, a synthetic tetrahydroisoquinoline alkaloid, selectively inhibits lipopoly-saccharide-induced VCAM-1 but not ICAM-1 expression in human endothelial cells. Atherosclerosis. 2009; 207:412–419.

9. Kim SJ, Kim JK, Lee DU, Kwak JH, Lee SM. Genipin protects lipopolysaccharide-induced apoptotic liver damage in D-galactosamine-sensitized mice. Eur J Pharmacol. 2010; 635:188–193.

10. Lee SJ, Oh PS, Lim KT. Hepatoprotective and hypolipidaemic effects of glycoprotein isolated from Gardenia jasminoides ellis in mice. Clin Exp Pharmacol Physiol. 2006; 33:925–933.

11. Koo HJ, Song YS, Kim HJ, Lee YH, Hong SM, Kim SJ, Kim BC, Jin C, Lim CJ, Park EH. Antiinflammatory effects of genipin, an active principle of gardenia. Eur J Pharmacol. 2004; 495:201–208.

12. Desideri G, Ferri C. Endothelial activation. Sliding door to atherosclerosis. Curr Pharm Des. 2005; 11:2163–2175.

13. Albelda SM, Smith CW, Ward PA. Adhesion molecules and inflammatory injury. FASEB J. 1994; 8:504–512.

14. Sakai S, Kawamata H, Kogure T, Mantani N, Terasawa K, Umatake M, Ochiai H. Inhibitory effect of ferulic acid and isoferulic acid on the production of macrophage inflammatory protein-2 in response to respiratory syncytial virus infection in RAW264.7 cells. Mediators Inflamm. 1999; 8:173–175.

15. Pasceri V, Wu HD, Willerson JT, Yeh ET. Modulation of vascular inflammation in vitro and in vivo by peroxisome proliferator- activated receptor-gamma activators. Circulation. 2000; 101:235–238.
16. Blaschke F, Caglayan E, Hsueh WA. Peroxisome proliferator-activated receptor gamma agonists: their role as vasoprotective agents in diabetes. Endocrinol Metab Clin North Am. 2006; 35:561–574.

17. Umetani M, Mataki C, Minegishi N, Yamamoto M, Hamakubo T, Kodama T. Function of GATA transcription factors in induction of endothelial vascular cell adhesion molecule-1 by tumor necrosis factor-alpha. Arterioscler Thromb Vasc Biol. 2001; 21:917–922.
18. Nizamutdinova IT, Kim YM, Chung JI, Shin SC, Jeong YK, Seo HG, Lee JH, Chang KC, Kim HJ. Anthocyanins from black soybean seed coats preferentially inhibit TNF-alpha-mediated induction of VCAM-1 over ICAM-1 through the regulation of GATAs and IRF-1. J Agric Food Chem. 2009; 57:7324–7330.
Fig. 2.
Anti-proliferative effect of genipin in HUVECs. HUVECs were treated as indicated concentrations of genipin for 24 h. Cell proliferation was then assessed by MTT assay. The data were expressed as the means±SD of three independent experiments. ††p<0.01 compared with control.
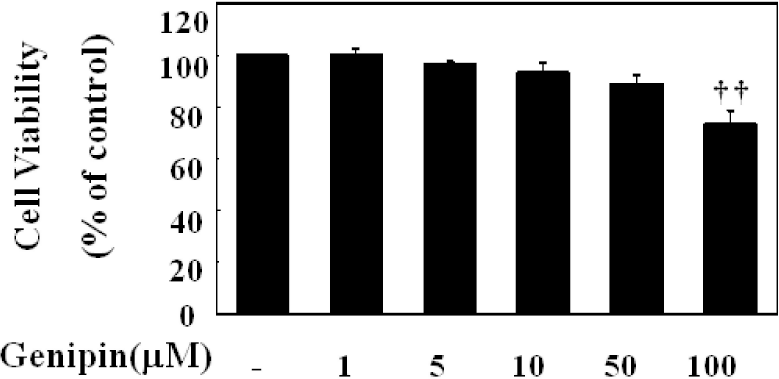
Fig. 3.
Inhibition of ROS production in TNF-α-activated HUVECs. Different concentration of genipin was added 30 min prior to TNF-α and further incubated for 10 min. Cells were suspended and then DCFH-DA was added at the final concentration of 10 μM. Fluorescence was monitored at the excitation and emission wavelength of 485 and 530 nm, respectively using a fluorescence plate reader (50 cycles per 20 s at 37°C). Results were expressed as relative changes to the initial fluorescence. The data were expressed as the means±SD of three independent experiments. ∗∗p<0.01 compared with control. ††p<0.01, compared with TNF-α.
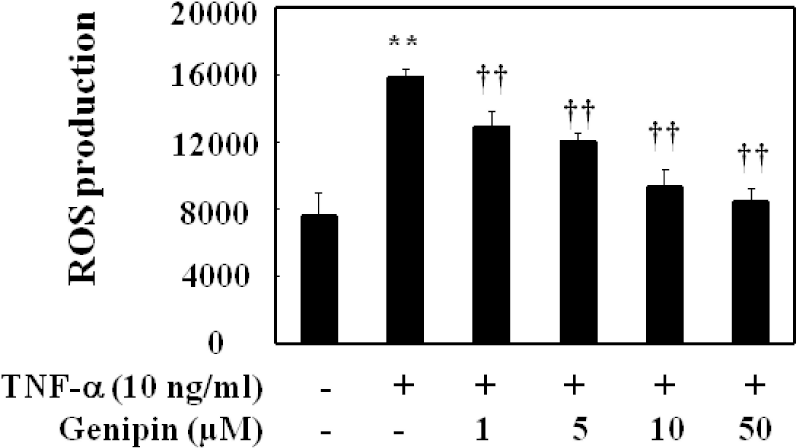
Fig. 4.
Preferential inhibition of TNF-α-mediated induction of VCAM-1 over ICAM-1. HUVECs were pretreated with genipin for 1 h and then treated with TNF-α for 6 h. The protein level of ICAM-1 and VCAM-1 was detected by Western blot analysis, as detailed in Materials and Methods. Data were confirmed by three independent experiments. The expression levels of ICAM-1, VCAM-1 protein were quantified by densitometer. Data are presented as means±SD from three independent experiments. Significance compared with control. ∗∗p<0.01 compared with control. ††p<0.01, compared with TNF-α
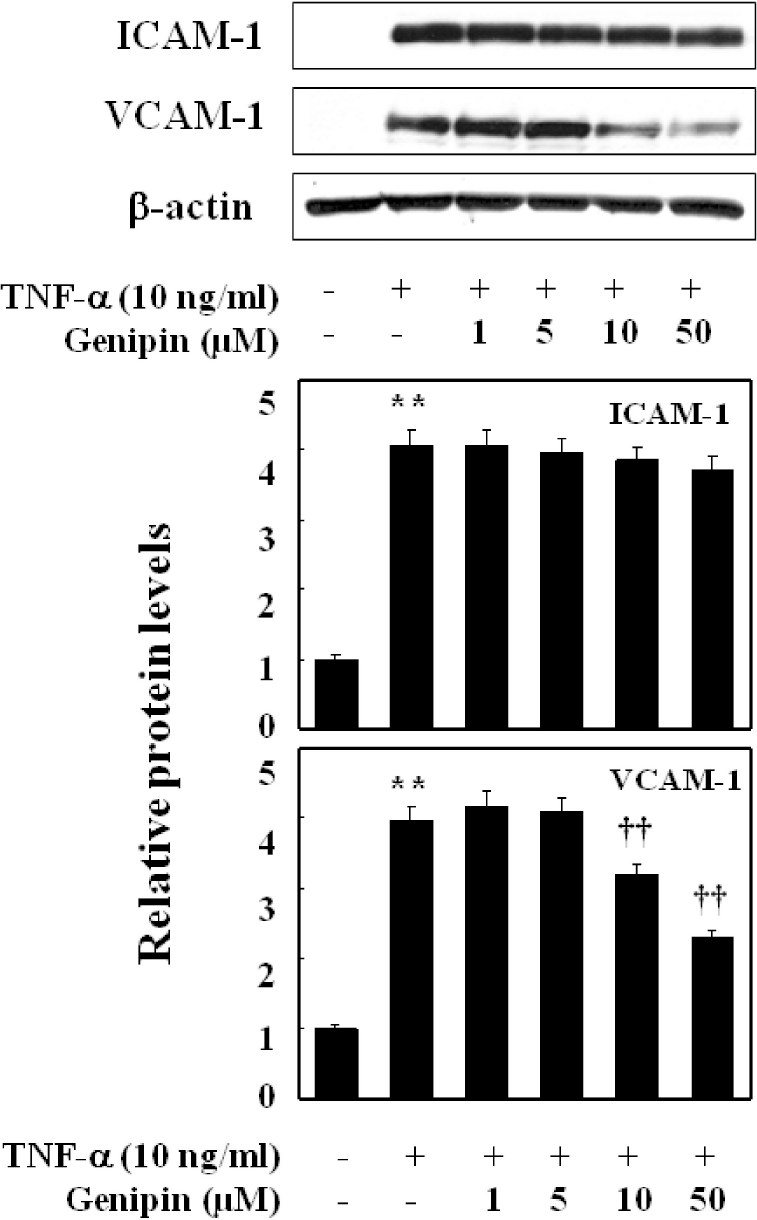
Fig. 5.
Time- and concentration-dependent induction of PPAR-γ protein in TNF-α-induced HUVECs. Cells were treated with different time period with fixed concentration (upper) or different concentration (lower) of genipin. Proteins were isolated and subjected to Western blot for PPAR-γ expression. Data were confirmed by three independent experiments. ∗∗p<0.01 compared with control.
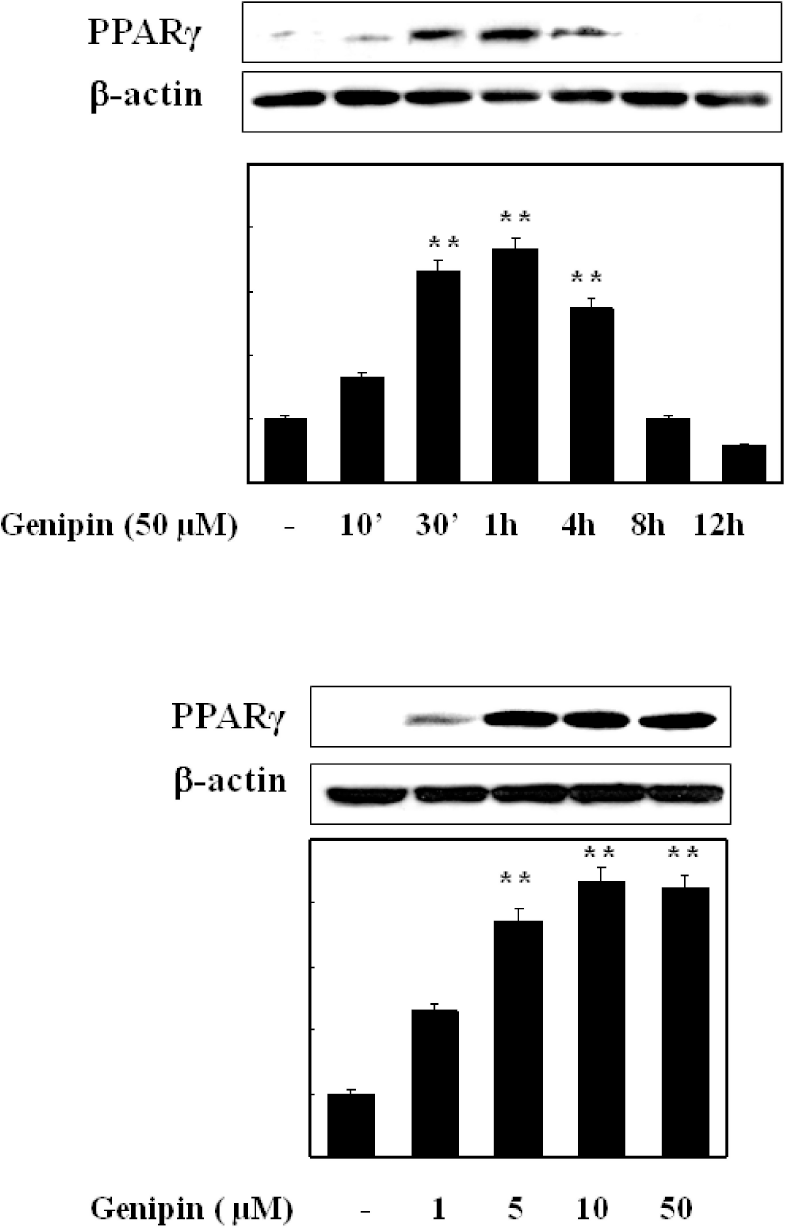
Fig. 6.
Effect on phosphorylation of ERK1/2, Akt, and PKC activation by TNF-α in HUVECs. Cells were pretreated with different concentration of genipin (1, 5, 10 and 50 μM) for 24 h, and then treated with TNF-α for 10 min for detection of phosphor-ERK1/2 and PKC or for 30 min for detection of phosphor-Akt. Cells were extracted and protein level was detected by Western blot analysis (upper). The blot was quantified by using densitometry and represented as % increase of control (lower). ∗∗p<0.01 compared with control. †p<0.05, ††p<0.01, compared with TNF-α, respectively. Data were confirmed by two independent experiments.

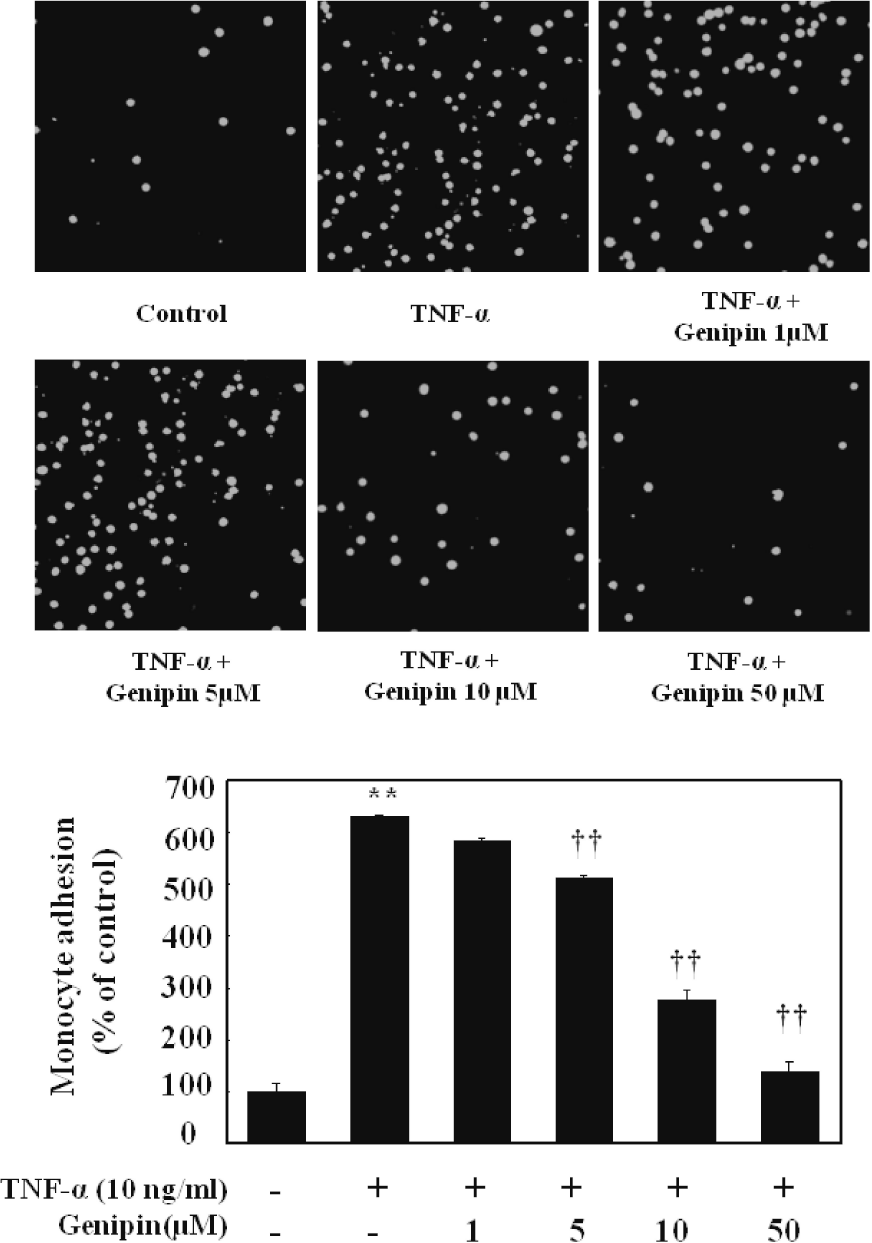
Fig. 7.
Effect on adhesion of monocytes to HUVECs stimulated with TNF-α. Cell were pretreated with different concentration of genipin (1, 5, 10 and 50 μM) and then stimulated with 10 ng/ml TNF-α for 6 h. Thereafter, cells were coincubated with fluorescent labeled monocytic cells for 30 min at 37°C. Monocyte adhesion was presented as images and a percentage of U937 cells bound to TNF-α-untreated cells (control) Data represented mean±SD from three independent experiments. ∗∗p<0.01, compared with control, ††p<0.01 compared with TNF-α, respectively.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download