Abstract
R-type Cav2.3 high voltage-activated Ca2+ channels in peripheral sensory neurons contribute to pain transmission. Recently we have demonstrated that, among the six Cav2.3 isoforms (Cav2.3a∼Cav2.3e), the Cav2.3e isoform is primarily expressed in trigeminal ganglion (TG) nociceptive neurons. In the present study, we further investigated expression patterns of Cav2.3 isoforms in the dorsal root ganglion (DRG) neurons. As in TG neurons, whole tissue RT-PCR analyses revealed the presence of two isoforms, Cav2.3a and Cav2.3e, in DRG neurons. Single-cell RT-PCR detected the expression of Cav2.3e mRNA in 20% (n=14/70) of DRG neurons, relative to Cav2.3a expression in 2.8% (n=2/70) of DRG neurons. Cav2.3e mRNA was mainly detected in small-sized neurons (n=12/14), but in only a few medium-sized neurons (n=2/14) and not in large-sized neurons, indicating the prominence of Cav2.3e in nociceptive DRG neurons. Moreover, Cav2.3e was preferentially expressed in tyrosine-kinase A (trkA)-positive, isolectin B4 (IB4)-negative and transient receptor potential vanilloid 1 (TRPV1)-positive neurons. These results suggest that Cav2.3e may be the main R-type Ca2+ channel isoform in nociceptive DRG neurons and thereby a potential target for pain treatment, not only in the trigeminal system but also in the spinal system.
Go to : 
References
1. Vanegas H, Schaible H. Effects of antagonists to high-threshold calcium channels upon spinal mechanisms of pain, hyperalgesia and allodynia. Pain. 2000; 85:9–18.

2. Wilson SM, Toth PT, Oh SB, Gillard SE, Volsen S, Ren D, Philipson LH, Lee EC, Fletcher CF, Tessarollo L, Copeland NG, Jenkins NA, Miller RJ. The status of voltage-dependent calcium channels in alpha 1E knock-out mice. J Neurosci. 2000; 20:8566–8571.
3. Ikeda M, Matsumoto S. Classification of voltage-dependent Ca2+ channels in trigeminal ganglion neurons from neonatal rats. Life Sci. 2003; 73:1175–1187.
4. Williams ME, Feldman DH, McCue AF, Brenner R, Velicelebi G, Ellis SB, Harpold MM. Structure and functional expression of alpha 1, alpha 2, and beta subunits of a novel human neuronal calcium channel subtype. Neuron. 1992; 8:71–84.
5. Bourinet E, Soong TW, Sutton K, Slaymaker S, Mathews E, Monteil A, Zamponi GW, Nargeot J, Snutch TP. Splicing of alpha 1A subunit gene generates phenotypic variants of P- and Q-type calcium channels. Nat Neurosci. 1999; 2:407–415.
6. Williams ME, Brust PF, Feldman DH, Patthi S, Simerson S, Maroufi A, McCue AF, Velicelebi G, Ellis SB, Harpold MM. Structure and functional expression of an omega-conotoxinsensitive human N-type calcium channel. Science. 1992; 257:389–395.
7. Zhang JF, Randall AD, Ellinor PT, Horne WA, Sather WA, Tanabe T, Schwarz TL, Tsien RW. Distinctive pharmacology and kinetics of cloned neuronal Ca2+ channels and their possible counterparts in mammalian CNS neurons. Neuropharmacology. 1993; 32:1075–1088.
8. Schneider T, Wei X, Olcese R, Costantin JL, Neely A, Palade P, Perez-Reyes E, Qin N, Zhou J, Crawford GD. Molecular analysis and functional expression of the human type E neuronal Ca2+ channel alpha 1 subunit. Receptors Channels. 1994; 2:255–270.
9. Williams ME, Marubio LM, Deal CR, Hans M, Brust PF, Philipson LH, Miller RJ, Johnson EC, Harpold MM, Ellis SB. Structure and functional characterization of neuronal alpha 1E calcium channel subtypes. J Biol Chem. 1994; 269:22347–22357.

10. Vajna R, Schramm M, Pereverzev A, Arnhold S, Grabsch H, Klockner U, Perez-Reyes E, Hescheler J, Schneider T. New isoform of the neuronal Ca2+ channel alpha1E subunit in islets of Langerhans and kidney–distribution of voltage-gated Ca2+ channel alpha1 subunits in cell lines and tissues. Eur J Biochem. 1998; 257:274–285.
11. Schramm M, Vajna R, Pereverzev A, Tottene A, Klockner U, Pietrobon D, Hescheler J, Schneider T. Isoforms of alpha1E voltage-gated calcium channels in rat cerebellar granule cells–detection of major calcium channel alpha1-transcripts by reverse transcription-polymerase chain reaction. Neuroscience. 1999; 92:565–575.
12. Pereverzev A, Leroy J, Krieger A, Malecot CO, Hescheler J, Pfitzer G, Klockner U, Schneider T. Alternate splicing in the cytosolic II-III loop and the carboxy terminus of human E-type voltage-gated Ca2+ channels: electrophysiological characterization of isoforms. Mol Cell Neurosci. 2002; 21:352–365.
13. Matthews EA, Bee LA, Stephens GJ, Dickenson AH. The Cav2.3 calcium channel antagonist SNX-482 reduces dorsal horn neuronal responses in a rat model of chronic neuropathic pain. Eur J Neurosci. 2007; 25:3561–3569.

14. Saegusa H, Kurihara T, Zong S, Minowa O, Kazuno A, Han W, Matsuda Y, Yamanaka H, Osanai M, Noda T, Tanabe T. Altered pain responses in mice lacking alpha 1E subunit of the voltage-dependent Ca2+ channel. Proc Natl Acad Sci USA. 2000; 97:6132–6137.
15. Murakami M, Suzuki T, Nakagawasai O, Murakami H, Murakami S, Esashi A, Taniguchi R, Yanagisawa T, Tan-No K, Miyoshi I, Sasano H, Tadano T. Distribution of various calcium channel alpha(1) subunits in murine DRG neurons and antinociceptive effect of omega-conotoxin SVIB in mice. Brain Res. 2001; 903:231–236.
16. Yusaf SP, Goodman J, Pinnock RD, Dixon AK, Lee K. Expression of voltage-gated calcium channel subunits in rat dorsal root ganglion neurons. Neurosci Lett. 2001; 311:137–141.

17. Fang Z, Park CK, Li HY, Kim HY, Park SH, Jung SJ, Kim JS, Monteil A, Oh SB, Miller RJ. Molecular basis of Cav2.3 calcium channels in rat nociceptive neurons. J Biol Chem. 2007; 282:4757–4764.
18. Kim YH, Park CK, Back SK, Lee CJ, Hwang SJ, Bae YC, Na HS, Kim JS, Jung SJ, Oh SB. Membrane-delimited coupling of TRPV1 and mGluR5 on presynaptic terminals of nociceptive neurons. J Neurosci. 2009; 29:10000–10009.

19. Murray SS, Cheema SS. Constitutive expression of the low-affinity neurotrophin receptor and changes during axotomy-induced death of sensory neurones in the neonatal rat dorsal root ganglion. J Anat. 2003; 202:227–238.

20. Stucky CL, Lewin GR. Isolectin B(4)-positive and -negative nociceptors are functionally distinct. J Neurosci. 1999; 19:6497–6505.

22. Snider WD, McMahon SB. Tackling pain at the source: new ideas about nociceptors. Neuron. 1998; 20:629–632.

23. Nagy JI, Hunt SP. Fluoride-resistant acid phosphatase-containing neurones in dorsal root ganglia are separate from those containing substance P or somatostatin. Neuroscience. 1982; 7:89–97.

24. Michael GJ, Averill S, Nitkunan A, Rattray M, Bennett DL, Yan Q, Priestley JV. Nerve growth factor treatment increases brain-derived neurotrophic factor selectively in TrkA-expressing dorsal root ganglion cells and in their central terminations within the spinal cord. J Neurosci. 1997; 17:8476–8490.

25. Bradbury EJ, Burnstock G, McMahon SB. The expression of P2X3 purinoreceptors in sensory neurons: effects of axotomy and glial-derived neurotrophic factor. Mol Cell Neurosci. 1998; 12:256–268.
26. Murakami M, Nakagawasai O, Suzuki T, Mobarakeh II, Sakurada Y, Murata A, Yamadera F, Miyoshi I, Yanai K, Tan-No K, Sasano H, Tadano T, Iijima T. Antinociceptive effect of different types of calcium channel inhibitors and the distribution of various calcium channel alpha 1 subunits in the dorsal horn of spinal cord in mice. Brain Res. 2004; 1024:122–129.
27. Brown SP, Safo PK, Regehr WG. Endocannabinoids inhibit transmission at granule cell to Purkinje cell synapses by modulating three types of presynaptic calcium channels. J Neurosci. 2004; 24:5623–5631.

28. Dietrich D, Kirschstein T, Kukley M, Pereverzev A, von der Brelie C, Schneider T, Beck H. Functional specialization of presynaptic Cav2.3 Ca2+ channels. Neuron. 2003; 39:483–496.
Go to : 
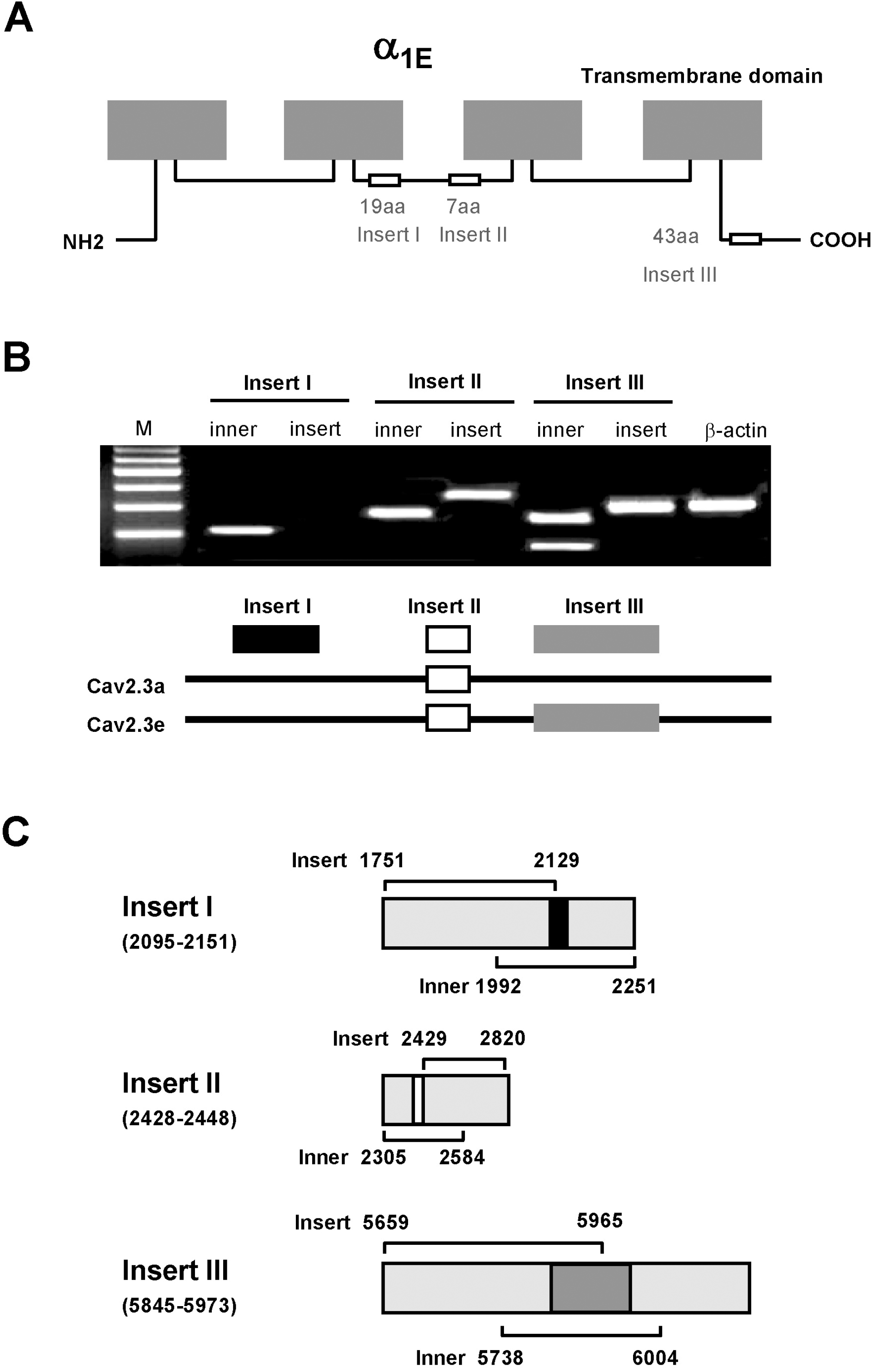 | Fig. 1.(A) Schematic diagram of α1E (Cav2.3) subunit. The structural variations cover two segments of 19 (insert I) and 7 amino acids (insert II) in the loop between domain II and III, and a third segment of 43 amino acids (insert III) in the proximal carboxy terminus. (B) Two Cav2.3 isoforms were found in DRG. Insert I, II and III was analyzed by whole tissue RT-PCR. Cav2.3 isoforms amplified from DRG neurons have insert II, but not insert I, and either lack or contain insert III depending on the isoform. Cav2.3a contains insert II, but not insert I and insert III, while Cav2.3e has insert II and insert III, but not insert I. (C) Illustrated are the locations of the primers designed to detect insert I, insert II and insert III for RT-PCR analysis in relation to the Cav2.3 subunit. |
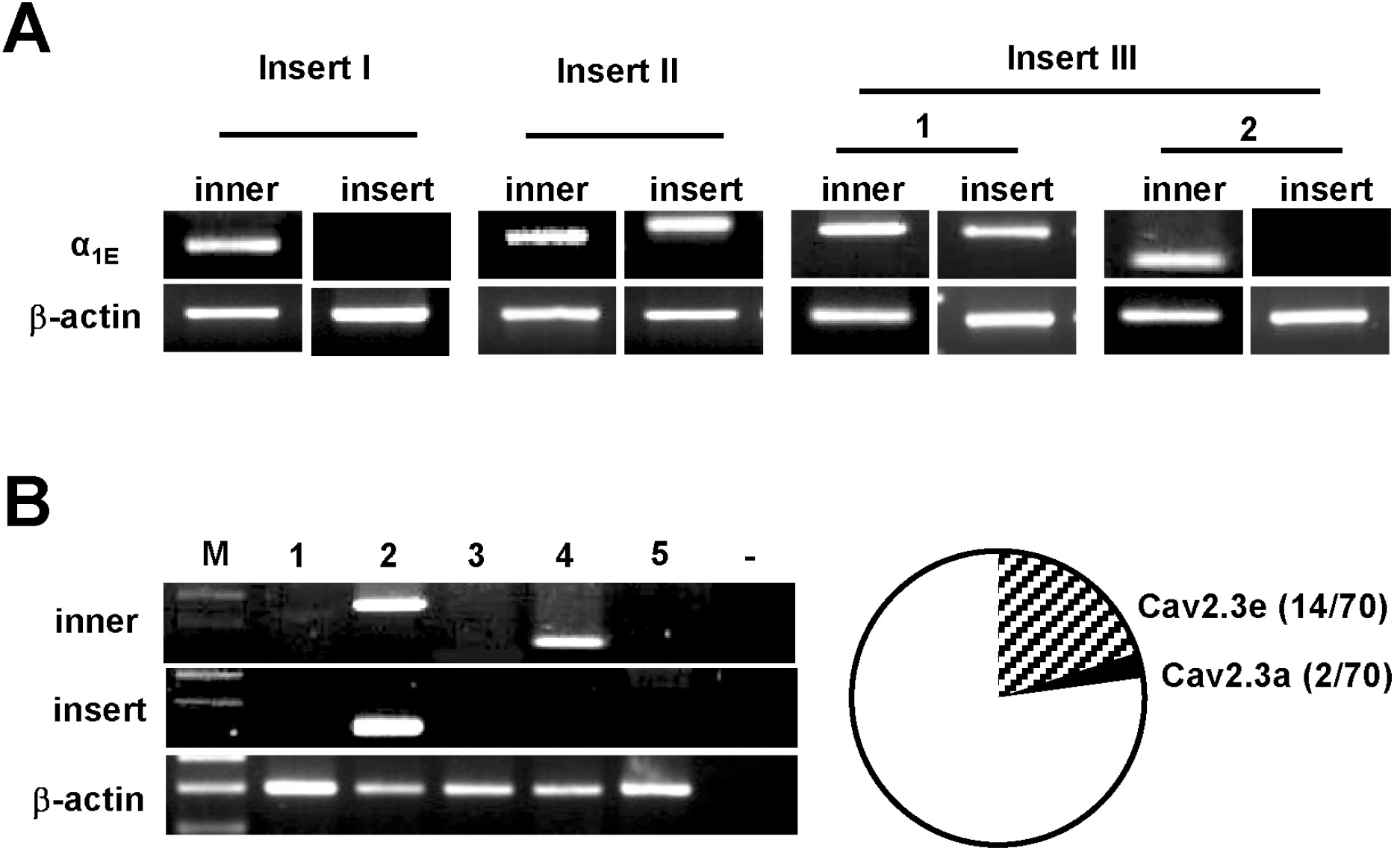 | Fig. 2.Expression of Cav2.3a and Cav2.3e isoforms from individual DRG neurons. (A) Single-cell RT-PCR products amplified with nested primer from DRG neurons. PCR products, with or without insert III (larger, 1 and smaller, 2), were produced in each DRG neuron. (B) Representative gels showing single-cell RT-PCR products amplified using insert III-specific primers. The numbers (1–5) indicate five different neurons examined from single-cell RT-PCR reaction. β-actin was used in each reaction as a positive control. Circle diagram shows the distribution of Cav2.3a and Cav2.3e in DRG neurons (total 70 neurons). |
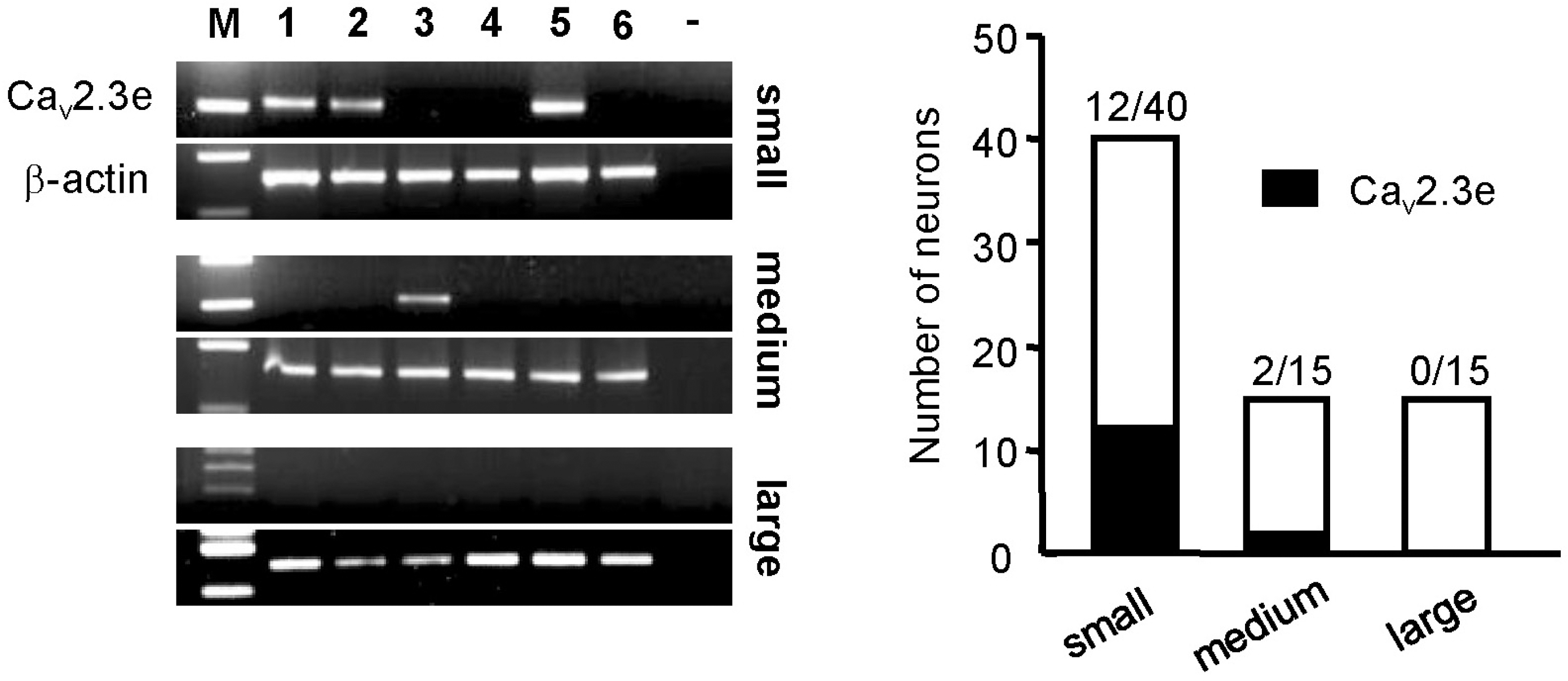 | Fig. 3.Expression pattern of Cav2.3e was analyzed in three groups: small-sized (<16μm), medium-sized (16∼20 μm) and large-sized (20∼30 μm) DRG neurons. Representative gels showing single-cell RT-PCR products obtained from six different neurons. White bars indicate the number of neurons in each group; black bars indicate the number of neurons with expression of Cav2.3e. |
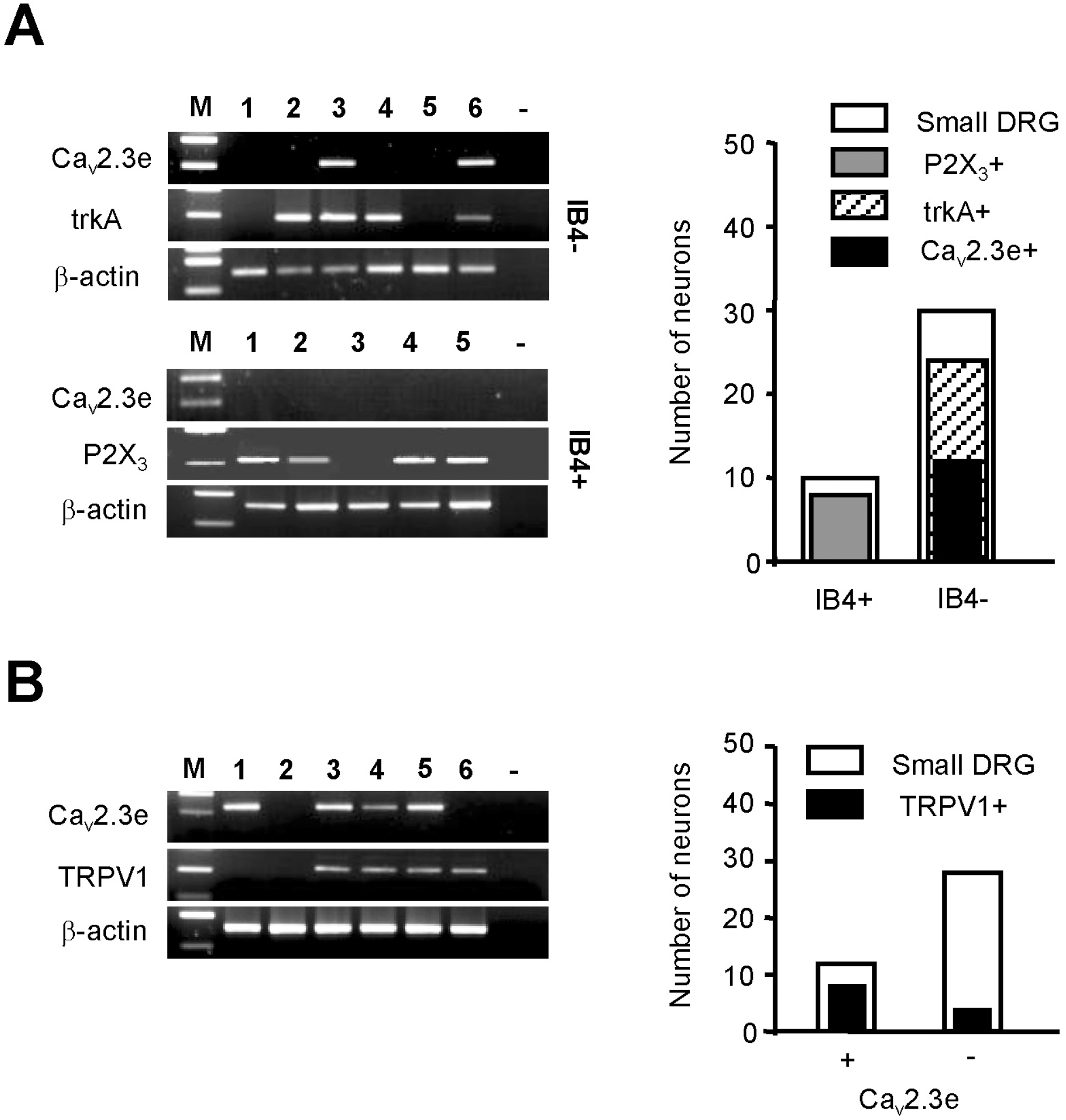 | Fig. 4.(A) Small-sized DRG neurons were divided into IB4-negative and IB4-positive neurons; expression of Cav2.3e was determined in each group with or without trkA and P2X3 expression. The black bar in the graph shows that Cav2.3e is prominent in trkA+/IB4-/TRPV1+ neurons. (B) Expression of Cav2.3e in TRPV1-expressing nociceptive neurons. Representative gels showing RT-PCR products amplified with Cav2.3e, TRPV1, and β-actin-specific primers from six different neurons. The majority of small-sized Cav2.3e-expressing DRG neurons are also TRPV1-positive, though TRPV1 expression is not exclusive to DRG expressing the Cav2.3e subunit (graph; right). |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download