Abstract
We elucidated the distribution of interstitial cells of Cajal (ICC) in human stomach, using cryosection and c-Kit immunohistochemistry to identify c-Kit positive ICC. Before c-Kit staining, we routinely used hematoxylin and eosin (HE) staining to identify every structure of human stomach, from mucosa to longitudinal muscle. HE staining revealed that the fundus greater curvature (GC) had prominent oblique muscle layer, and c-Kit immunostaining c-Kit positive ICC cells were found to have typical morphology of dense fusiform cell body with multiple processes protruding from the central cell body. In particular, we could observe dense processes and ramifications of ICC in myenteric area and longitudinal muscle layer of corpus GC. Interestingly, c-Kit positive ICC-like cells which had morphology very similar to ICC were found in gastric mucosa. We could not find any significant difference in the distribution of ICC between fundus and corpus, except for submucosa where the density of ICC was much higher in gastric fundus than corpus. Furthermore, there was no significant difference in the density of ICC between each area of fundus and corpus, except for muscularis mucosa. Finally, we also found similar distribution of ICC in normal and cancerous tissue obtained from a patient who underwent pancreotomy and gastrectomy. In conclusion, ICC was found ubiquitously in human stomach and the density of ICC was significantly lower in the muscularis mucosa of both fundus/corpus and higher in the submucosa of gastric fundus than corpus.
Go to : 
References
1. Cajal SR. Histologie du système nerveux de l'homme et des vertébrés. 2nd ed.Paris: Maloine;1911. p. 891–942.
2. Faussone-Pellegrini MS, Cortesini C. Ultrastructural features and localization of the interstitial cells of Cajal in the smooth muscle coat of human esophagus. J Submicrosc Cytol. 1985; 17:187–197.
3. Hagger R, Gharaie S, Finlayson C, Kumar D. Regional and transmural density of interstitial cells of Cajal in human colon and rectum. Am J Physiol. 1998; 275:G1309–G1316.

4. Radenkovic G, Savic V, Mitic D, Grahovac S, Bjelakovic M, Krstic M. Development of c-Kit immunopositive interstitial cells of Cajal in the human stomach. J Cell Mol Med. 2010; 14:1125–1134.
5. Romert P, Mikkelsen HB. c-Kit immunoreactive cells of Cajal in the human small and large intestine. Histochem Cell Biol. 1998; 109:195–202.
6. Torihashi S, Horisawa M, Watanabe Y. c-Kit immunoreactive interstitial cells in the human gastrointestinal tract. J Auton Nerv Syst. 1999; 75:38–50.
7. Huizinga JD, Thuneberg L, Kluppel M, Malysz J, Mikkelsen HB, Bernstein A. W/Kit gene required for interstitial cells of Cajal and for intestinal pacemaker activity. Nature. 1995; 373:347–349.
8. Torihashi S, Ward SM, Sanders KM. Development of c-Kit-positive cells and the onset of electrical rhythmicity in murine small intestine. Gastroenterology. 1997; 112:144–155.
9. Nemeth L, Maddeur S, Puri P. Immunolocalization of the gap junction protein connexin 43 in the interstitial cells of Cajal in the normal and Hirschsprung's disease bowel. J Pediatr Surg. 2000; 35:823–828.
10. Klüppel M, Huizinga JD, Malysz J, Bernstein A. Developmental origin and Kit- dependent development of the interstitial cells of Cajal in the mammalian small intestine. Dev Dyn. 1998; 211:60–71.
11. Maeda H, Yamagata A, Nishikawa S, Yoshinaga K, Kobayashi S, Nishi K, Nishikawa S. Requirement of c-Kit for development of intestinal pacemaker system. Development. 1992; 116:369–375.
12. Ördög T, Redelman D, Horváth VJ, Miller LJ, Horowitz B, Sanders KM. Qunatitative analysis by flow cytometry of interstitial cells of Cajal, pacemakers, and mediators of neurotransmission in the gastrointestinal tract. Cytometry A. 2004; 62:139–149.
13. Burns AJ, Lomax AE, Torihashi S, Sanders KM, Ward SM. Interstitial cells of Cajal mediated inhibitory neurotransmission in the stomach. Proc Natl Acad Sci USA. 1996; 93:12008–12013.
14. Goyal RK, Chaudhury A. Mounting evidence against the role of ICC in neurotransmission to smooth muscle in the gut. Am J Physiol Gastrointest Liver Physiol. 2010; 298:G10–G13.

15. Wang XY, Paterson C, Huizinga JD. Cholinergic and nitrergic innervation of ICC-DMP and ICC-IM in the human small intestine. Neurogastroenterol Motil. 2003; 15:531–543.

16. Nakahara M, Isozaki K, Hirota S, Vanderwinden JM, Takakura R, Kinoshita K, Miyagawa J, Chen H, Miyazaki Y, Kiyohara T, Shinomura Y, Matsuzawa Y. Deficiency of Kit-positive cells in the colon of patients with diabetes mellitus. J Gastroenterol Hepatol. 2002; 17:666–670.

17. Vanderwinden JM, Rumessen JJ. Interstitial cells of Cajal in human gut and gastrointestinal disease. Microsc Res Tech. 1999; 47:344–360.

18. Der T, Bercik P, Donnelly G, Jackson T, Berezin I, Collins SM, Huizinga JD. Interstitial cells of Cajal and inflammation-induced motor dysfunction in the mouse small intestine. Gastroenterology. 2000; 119:1590–1599.

19. Popescu LM, Gherghiceanu M, Cretoiu D, Radu E. The connective connection: interstitial cells of Cajal-like cells establish synapses with immunoreactive cells. J Cell Mol Med. 2005; 9:714–730.
20. Hassan S, Kinoshita Y, Kawanami C, Kishi K, Matsushima Y, Ohashi A, Funasaka Y, Maekawa T, He-Yao W, Chuba T. Expression of protooncogene c-Kit and its ligand stem cell factor (SCF) in gastric carcinoma cell lines. Digestive Diseases and Sciences. 1998; 43:8–14.
21. Kunisawa Y, Komuro T. Interstitial cells of Cajal associated with submucosal plexus of the guinea-pig stomach. Neuroscience Letters. 2008; 434:273–276.
22. Torihashi S, Yokoi K, Nagaya H, Aoki K, Fujimoto T. New monoclonal antibody (AIC) identifies interstitial cells of Cajal in the musculature of the mouse gastrointestinal tract. Auton Neurosci. 2004; 113:16–23.

23. Reed J, Ouban A, Schickor FK, Muraca P, Yeatman T, Coppola D. Immunohistochemical staining for c-Kit (CD117) is a rare event in human colorectal carcinoma. Clin Colorectal Cancer. 2002; 2:119–122.
24. Ashman LK. The biology of stem cell factor and its receptor C-kit. Int J Biochem Cell Biol. 1999; 31:1037–1051.
25. Rygaard K, Nakamura T, Spang Thomsen M. Expression of the proto-oncogene c-met and c-Kit and their ligands, hepatocyte growth factor/scatter facter and stem cell factor, in SCLS cell lines and xenografts. Br J Cancer. 1993; 67:37–46.
26. Hibi K, Takahashi T, Sekido Y, Ueda R, Hida T, Ariyoshi Y, Takagi H, Takahashi T. Coexpression of the stem cell factor and the c-Kit genes in small-cell lung cancer. Oncogene. 1991; 6:2291–2296.
27. Fujiwara T, Motoyama T, Ishihara N, Watanabe H, Kumanishi T, Kato K, Ichinose H, Nagatsu T. Characterization of four new cell lines derived from small-cell gastrointestinal carcinoma. Int J Cancer. 1993; 54:965–971.

28. Song G, Hirst GDS, Sanders KM, Ward SM. Regional variation in ICC distribution, pacemaking activity and neural responses in the longitudinal muscle of the murine stomach. J Physiol. 2005; 564:523–540.

29. Hirst GDS, Beckett EAH, Sanders KM, Ward SM. Regional variation in contribution of myenteric and intramuscular interstitial cells of Cajal to generation of slow waves in mouse gastric antrum. J Physiol. 2002; 540:1003–1012.

30. Han J, Shen WH, Jiang YZ, Yu B, He YT, Mei F. Distribution, development and proliferation of interstitial cells of Cajal in murine colon: an immunohistochemical study from neonatal to adult life. Histochem Cell Biol. 2010; 133:163–175.

31. Ibba Manneschi L, Pacini S, Corsani L, Cechi P, Faussone-Pellegrini MS. Interstitial cells of Cajal in the human stomach: distribution and relationship with enteric innervation. Histol Histopathol. 2004; 19:1153–1164.
32. Nemeth L, Puri P. Three-demensional morphology of c-Kit-positive cellular network and nitrergic innervation in the human gut. Arch Pathol Lab Med. 2001; 125:899–904.
33. Burns AJ, Herbert TM, Ward SM, Sanders KM. Interstitial cells of Cajal in the guinea-pig gastrointestinal tract as revealed by c-Kit immunohistochemistry. Cell Tissue Res. 1997; 290:11–20.
34. Horiguchi K, Sanders KM, Ward SM. Enteric motor neurons from synaptic-like junctions with interstitial cells of Cajal in the canine gastric antrum. Cell Tissue Res. 2003; 311:299–313.
35. Faussone-Pellegrini MS, Pantalone D, Cortesini C. An ultra-structural study of the interstitial cells of Cajal of the human stomach. J Submicrosc Cytol Pathol. 1989; 21:439–460.
36. Abrahamsson H. Studies on the inhibitory nervous control of gastric motility. Acta Physiol Scand Suppl. 1973; 390:1–38.
37. Percy WH, Warren JM, Brunz JT. Characteristics of the muscularis mucosae in the acid-secreting region of the rabbit stomach. Am J Physiol. 1999; 276:G1213–G1220.
38. Synnerstad I, Ekblad E, Sundler F, Holm L. Gastric mucosal smooth muscles may explain oscillations in glandular pressure: role of vasocative intestinal peptide. Gastroenterology. 1998; 114:284–294.
39. Moskalewski S, Biernacka-Wawrzonek D, Klimkiewicz J, Zdun R. Venous outflow system in rabbit gastric mucosa. Folia Morphol (Warsz). 2004; 63:151–157.
40. Fawcett DW. A Textbook of Histology. 12th ed.New York, London: Chapman & Hall;1994. p. 617–651.
Go to : 
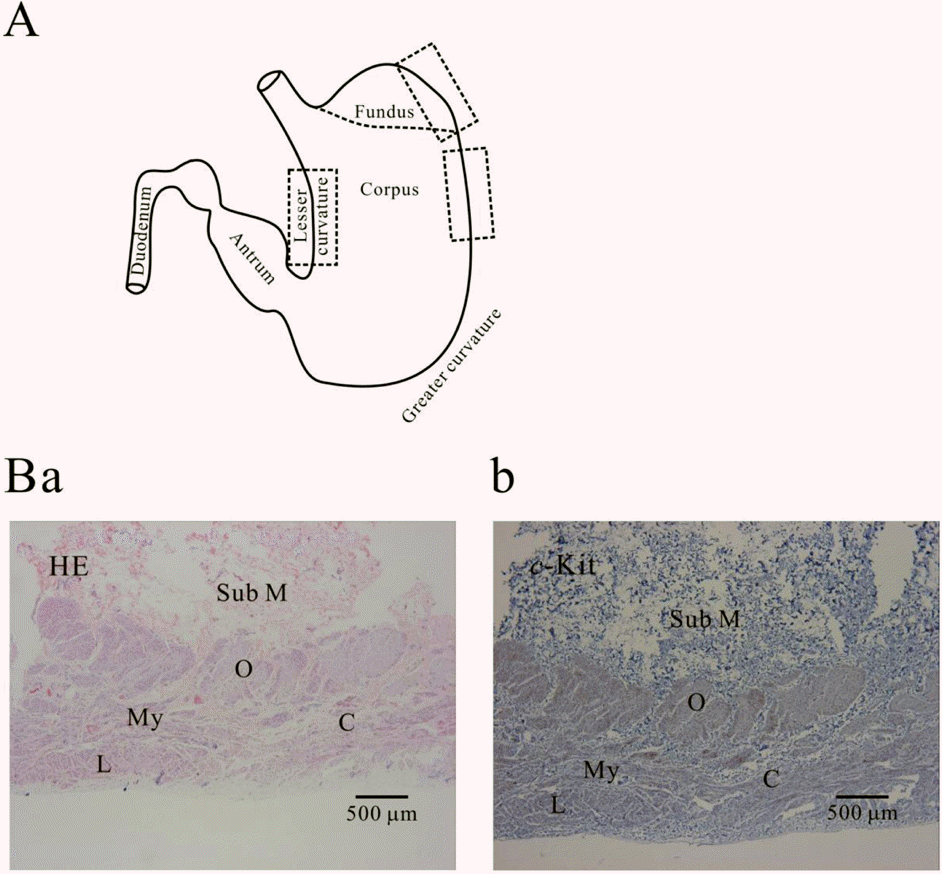 | Fig. 1.Histology of human gastric wall. As shown in panel A, we used fundus greater curvature, corpus greater curvature and corpus lesser curvature of human stomach. In HE staining of fundus greater curvature (Ba), we identified oblique, circular and longitudinal muscles. And then in c-Kit immunostaining (Bb), we observed c-Kit positive immnuhistochemical reactivity of ICC, which appeared as dark brown spots. Sub M, submucosa; O, oblique muscle; C, circular muscle; My, myenteric region; L, longitudinal muscle. |
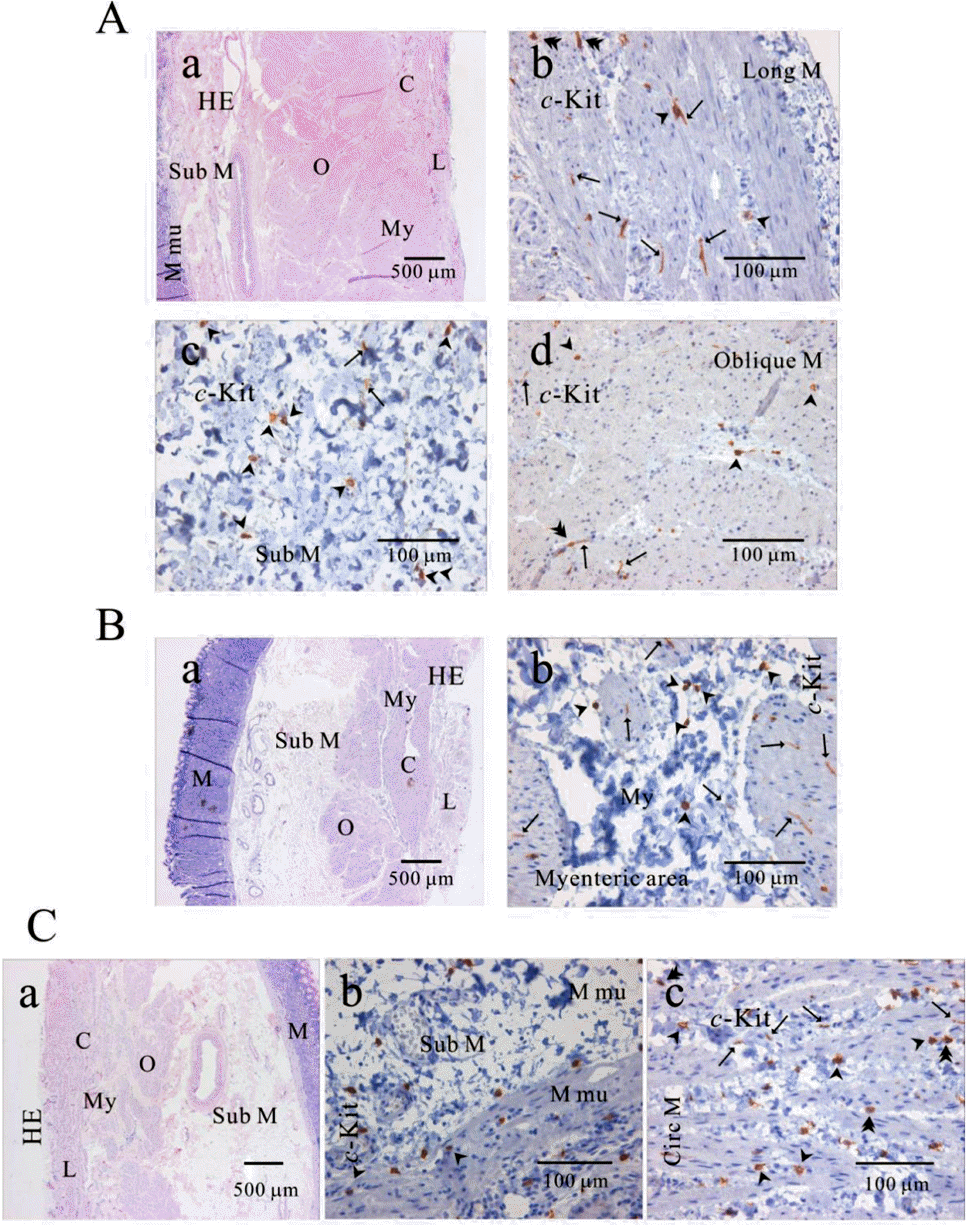 | Fig. 2.Distribution of ICC in human gastric fundus of greater curvature. In HE staining (Aa, Ba, Ca), the whole layers of human gastric fundus from greater curvature can be seen. Oblique muscle layer is prominent in human gastric fundus. In c-Kit immunostaining, we identified c-Kit positive ICC in every muscle layers: longitudinal muscle (Ab), submucosa (Ac), oblique muscle (Ad), myenteric area (Bb), muscularis mucosa (Cb) and circular muscle (Cc). Since we used cryosection for immunohistochemical study, ICC could not be seen as a whole structure. Instead, ICC is seen in long spindle-like (fusiform) shaped cell body and multiple processes from the central cell body, which is especially prominent in (Ab, Ad, Bb). ICC is identified in submucosa, muscularis mucosa and mucosa, too (Ac) and (Cb). Arrow: processes or ramification, arrow head: central cell body, double arrow head: ICC in septa (ICC-SEP). |
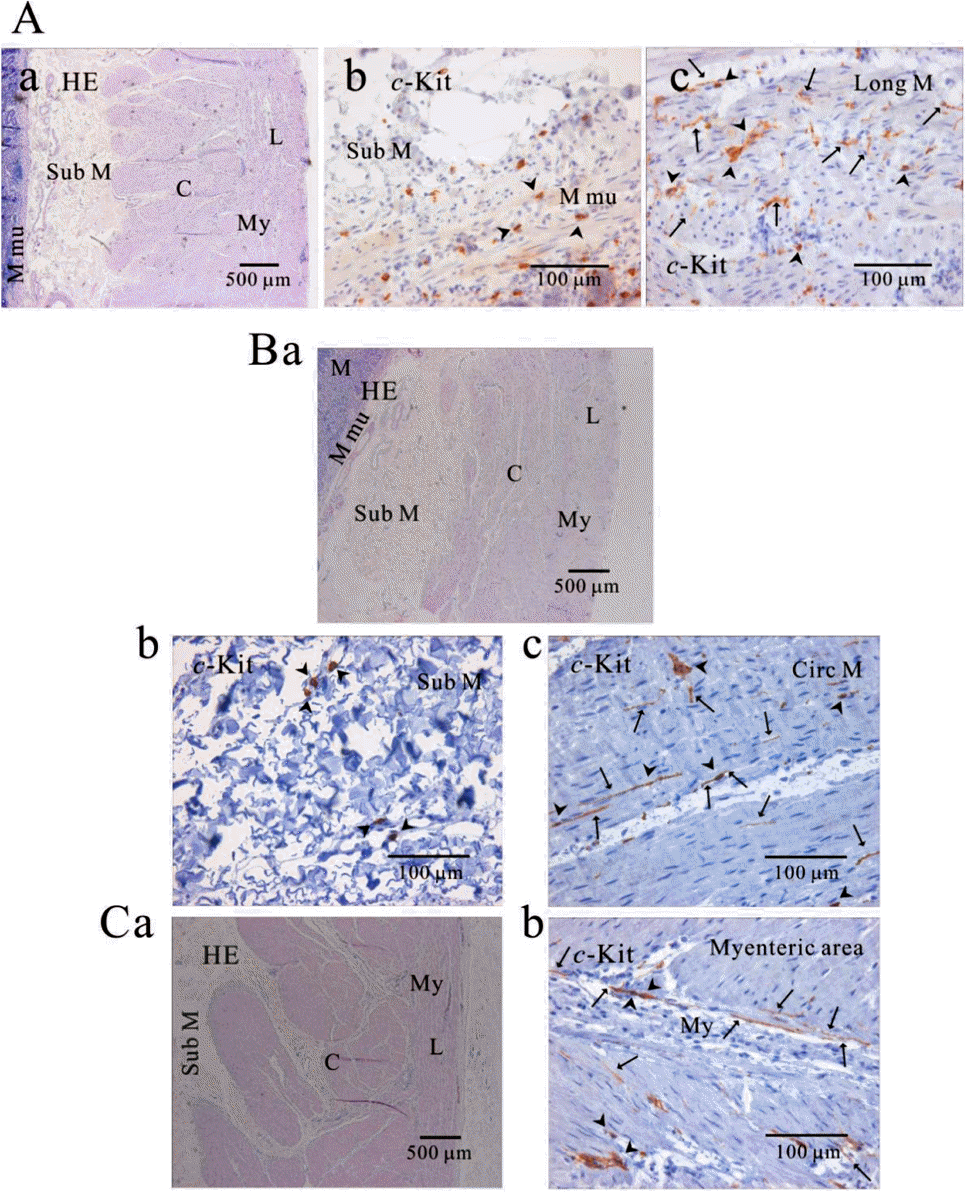 | Fig. 3.Distribution of ICC in human gastric corpus of greater curvature. In HE staining (Aa, Ba, Ca), the whole layers of human gastric corpus from greater curvature can be seen. In c-Kit immunostaining, c-Kit positive ICC is also observed in every layer; muscularis mucosa (Ab), longitudinal muscle (Ac), submucosa (Bb), circular muscle (Bc), and myenteric area (Cb). In human gastric corpus, oblique muscle is not as prominent as in fundus. ICC is seen in long spindle-like (fusiform) shaped cell body and multiple processes from the central cell body, and is also identified in mucosa (Ab). Arrow: processes or ramification, arrow head: central cell body. |
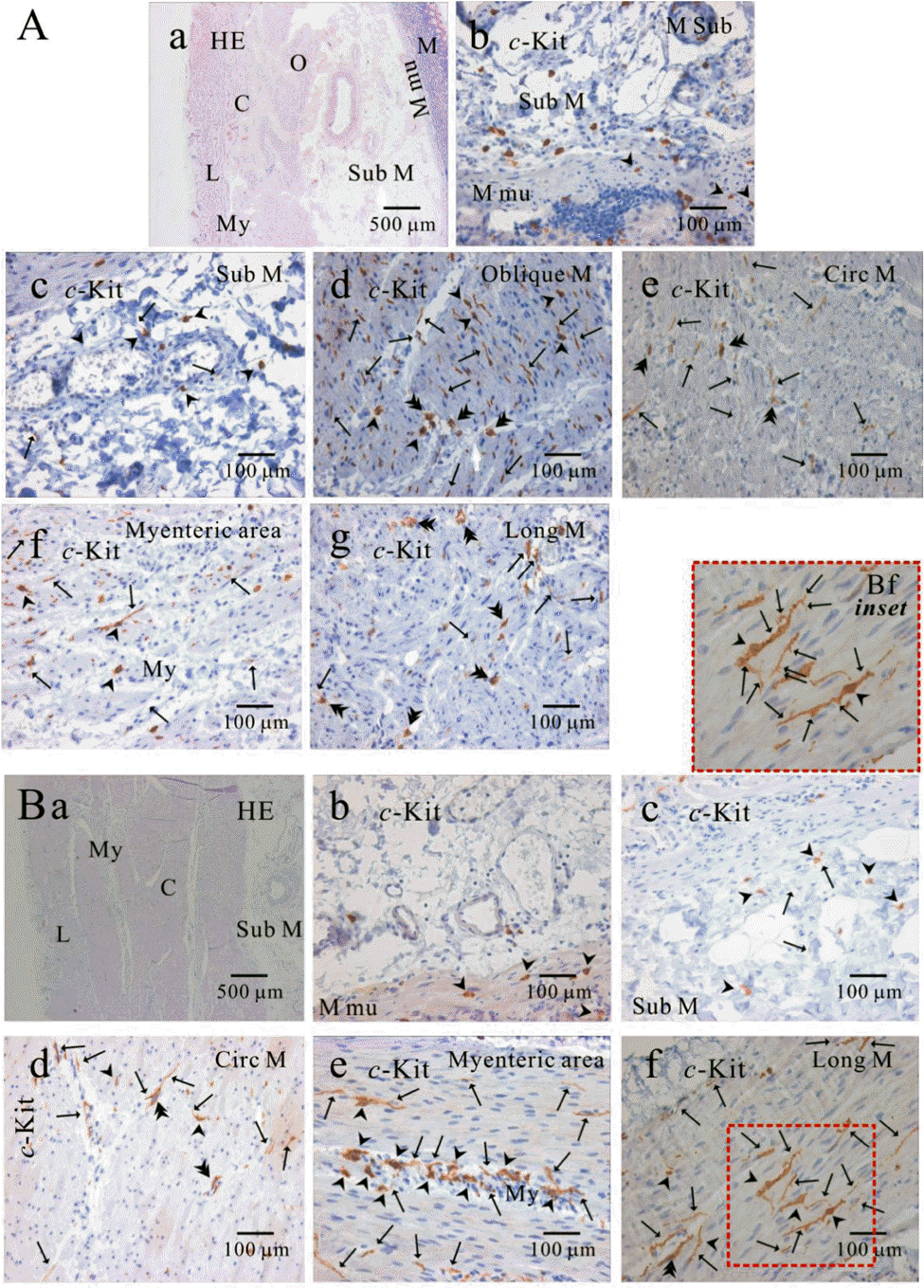 | Fig. 4.Comparison of histology between fundus and corpus in greater curvature of human stomach. In HE staining, the whole layer of fundus (Aa) and corpus (Ba) are shown. It is striking to find that fundus has prominent structure of oblique muscle, unlike corpus. c-Kit positive ICC is observed in every layer of human gastric fundus (Ab∼Ag) and corpus (Bb∼Bf) of greater curvature. Interestingly, almost all kinds of ICC are found in oblique muscle layer, however, ICCs of circular and longitudinal muscle are ICC-SEP. In panel (Bf and Bf inset), it is evident that ICC of longitudinal muscle has lots of processes and ramifications. Arrow: processes or ramification, arrow head: central cell body, double arrow head: ICC in septa (ICC-SEP). |
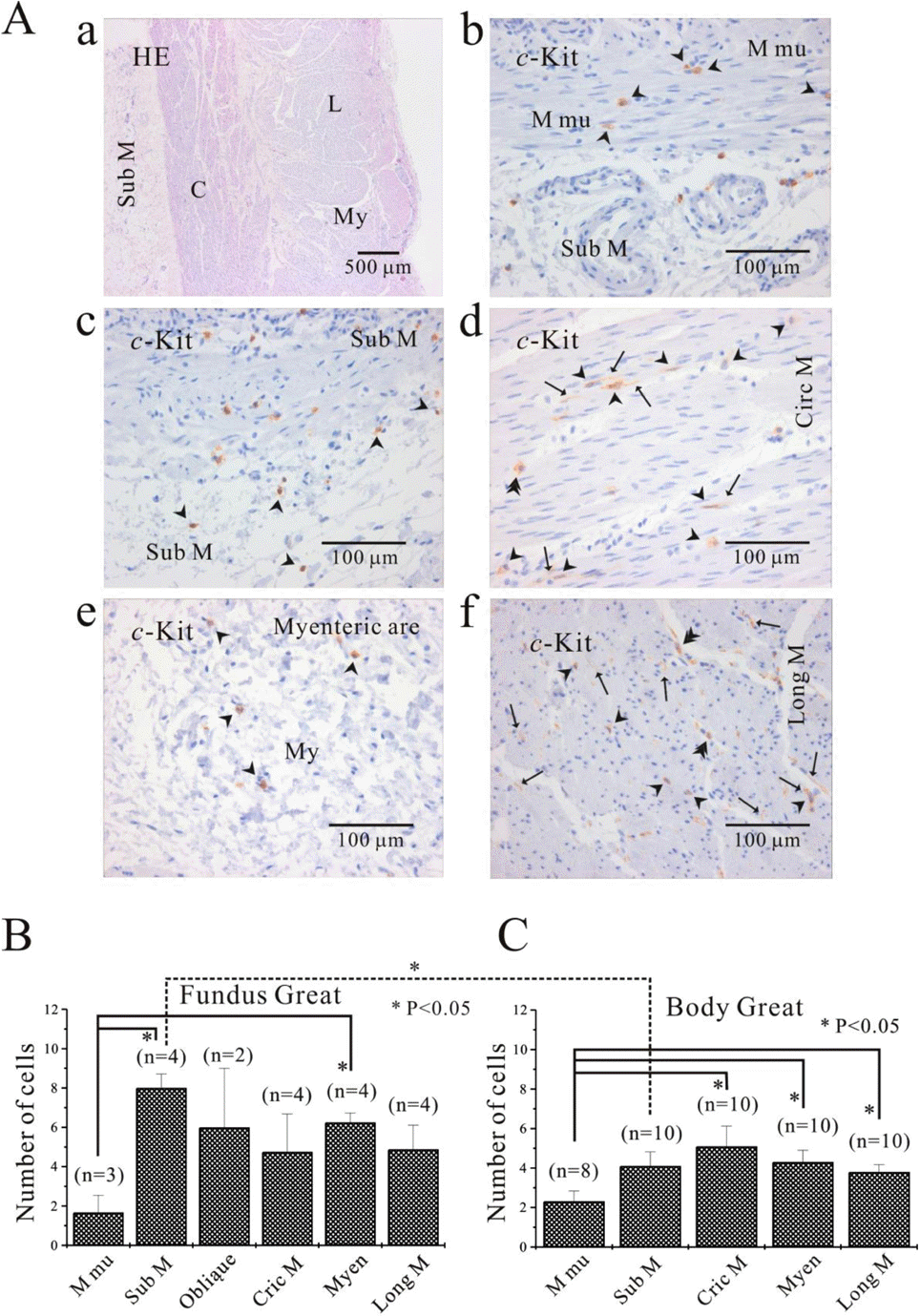 | Fig. 5.Comparison of ICC distribution between corpus and fundus and regional distribution of ICC in part of human stomach. In HE staining (Aa), the whole layers of human gastric corpus from lesser curvature are demonstrated. In corpus lesser curvature, c-Kit positive ICC is also found in all the layers (A). ICC numbers are compared between fundus greater curvature (B) and corpus greater curvature (C). There is no significant difference between two regions except for ICC from submucosa. The average number of ICC is 8±0.7 count/unit area in submucosa of fundus greater curvature, which is significantly higher than 4.1±0.7 count/unit area of body greater curvature (p<0.05). Meanwhile, the density of ICC in muscularis mucosa is significantly less than that of other layers of fundus (submucosa and myenteric area) and corpus (circular muscle, myenteric area and longitudinal muscle) (p<0.05; (5B) and (5C)). However, the density of ICC in muscularis mucosa is less than that of other layer with no significant difference (p> 0.05; (5B) and (5C)). Arrow: processes or ramification, arrow head: central cell body, double arrow head: ICC in septa (ICC-SEP). |
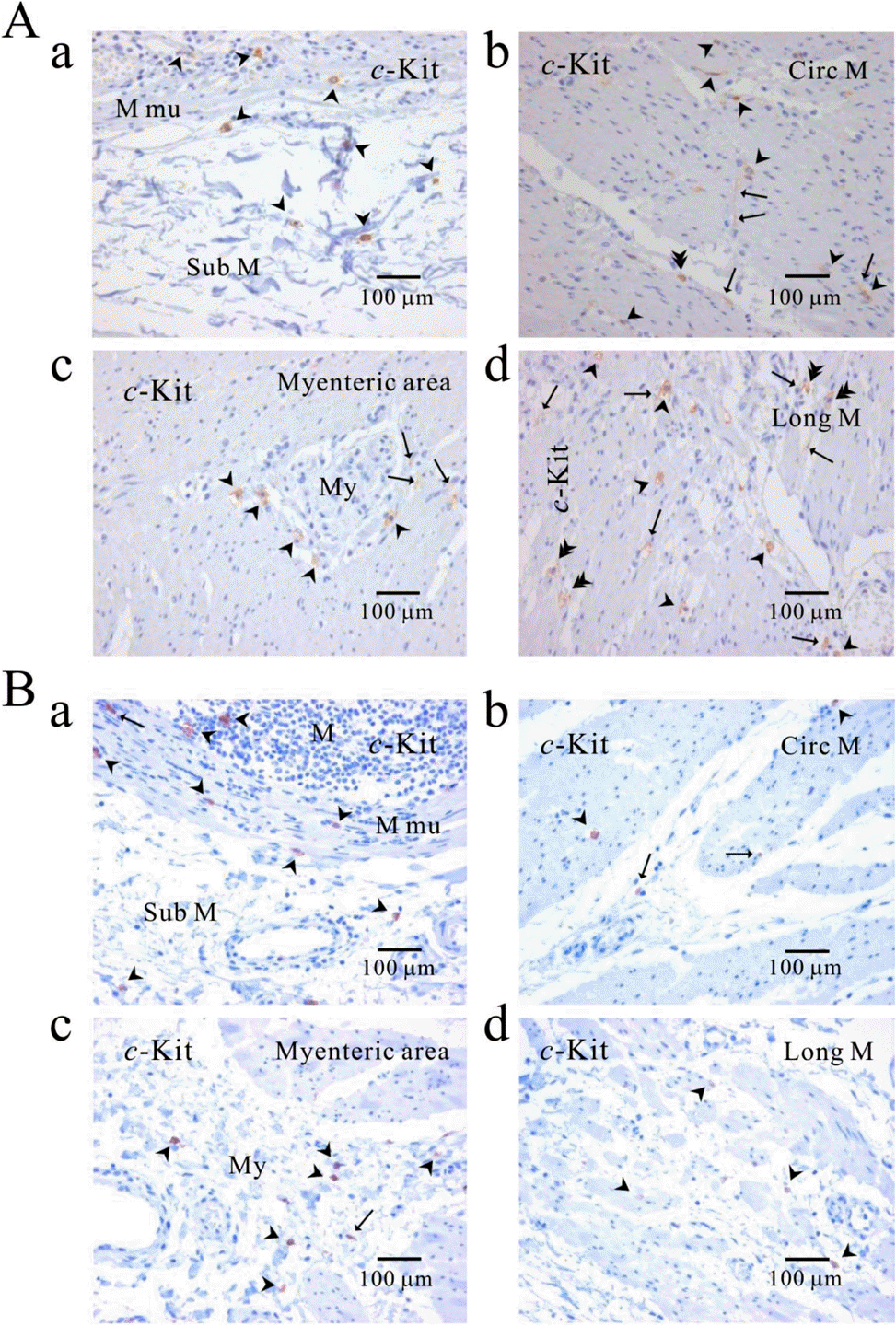 | Fig. 6.Comparison of ICC distribution between normal and cancerous tissue of human stomach. Distribution of ICC is compared between normal (A) and cancerous tissue (B) obtained from a patient who underwent pancreotomy and gastrectomy. c-Kit positive ICC is observed in every layer; submucosa (Aa, Ba), circular muscle (Ab, Bb), myenteric area (Ac, Bc), and longitudinal muscle (Ad, Bd). Note that c-Kit positive immunoreactivity is found also in gastric mucosa of cancer area (Ba) as well as normal area (Aa). Arrow: processes or ramification, arrow head: central cell body, double arrow head: ICC in septa (ICC-SEP). |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download