Abstract
The mushroom Cordyceps militaris has been used for a long time in eastern Asia as a nutraceutical and in traditional Chinese medicine as a treatment for cancer patients. In the present study, a cytotoxic antifungal protease was purified from the dried fruiting bodies of C. militaris using anion-exchange chromatography on a DEAE-Sepharose column. Electrophoretic analyses indicated that this protein, designated C. militaris protein (CMP), has a molecular mass of 12 kDa and a pI of 5.1. The optimum conditions for protease activity were a temperature of 37°C and pH of 7.0~9.0. The enzyme activity was specifically inhibited by the serine protease inhibitor phenylmethylsulfonyl fluoride. Amino acid composition of intact CMP and amino acid sequences of three major peptides from a tryptic digest of CMP were determined. CMP exerted strong antifungal effect against the growth of the fungus Fusarium oxysporum, and exhibited cytotoxicity against human breast and bladder cancer cells. These results indicate that C. militaris represents a source of a novel protein that might be applied in diverse biological and medicinal applications.
Go to : 
REFERENCES
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 72:248–254. 1976.

Cunningham KG., Manson W., Spring FS., Hutchinson SA. Cordycepin, a metabolic product isolated from cultures of Cordyceps militaris (Linn.) Link. Nature. 166:949. 1950.
Hattori M., Isomura S., Yokoyama E., Ujita M., Hara A. Extracellular trypsin-like proteases produced by. Cordyceps militaris. J Biosci Bioeng. 100:631–636. 2005.
Hong SY., Oh JE., Lee KH. In vitro antifungal activity and cytotoxicity of a novel membrane-active peptide. Antimicrob Agents Chemother. 43:1704–1707. 1999.

Joshi BN., Sainani MN., Bastawade KB., Gupta VS., Ranjekar PK. Cysteine protease inhibitor from pearl millet: a new class of antifungal protein. Biochem Biophys Res Commun. 246:382–387. 1998.

Jung EC., Kim KD., Bae CH., Kim JC., Kim DK., Kim HH. A mushroom lectin from ascomycete. Cordyceps militaris. Biochim Biophys Acta-General Subjects. 1770:833–838. 2007.
Kim BS., Oh KT., Cho DH., Kim YJ., Koo WM., Kong KH., Kim HH. A sialic acid-binding lectin from the legume Maackia fauriei: comparison with lectins from M. amurensis. Plant Science. 167:1315–1321. 2004.
Kim JS., Sapkota K., Park SE., Choi BS., Kim S., Nguyen TH., Kim CS., Choi HS., Kim MK., Chun HS., Park Y., Kim SJ. A fibrinolytic enzyme from the medicinal mushroom. Cordyceps militaris. J Microbiol. 44:622–631. 2006.
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227:680–685. 1970.

Li H., Matsunaga S., Fusetani N. Halicylindramides A-C, antifungal and cytotoxic depsipeptides from the marine sponge. Halichondria cylindrata. J Med Chem. 38:338–343. 1995.
Lindequist U., Niedermeyer TH., Julich WD. The pharmacological potential of mushrooms. Evid Based Complement Alternat Med. 2:285–299. 2005.

Moreno AB., del Pozo AM., Borja M., Segundo BS. Activity of the antifungal protein from Aspergillus giganteus against Botrytis cinerea. Phytopathology. 93:1344–1353. 2003.
Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 65:55–63. 1983.

Nagai PHK., Ng TB. Lentin, a novel and potent antifungal protein from shitake mushroom with inhibitory effects on activity of human immunodeficiency virus-1 reverse transcriptase and proliferation of leukemia cells. Life Sciences. 73:3363–3374. 2003.

Nagai PHK., Zhao Z., Ng TB. Agrocybin, an antifungal peptide from the edible mushroom. Agrocybe cylindracea. Peptides. 26:191–196. 2005.
St Leger RJ., Charnley AK., Cooper RM. Characterization of cuticle-degrading proteases produced by the entomopathogen. Metarhizium anisopliae. Arch Biochem Biophys. 253:221–232. 1987.
Vernekar JV., Ghatge MS., Deshpande VV. Alkaline protease inhibitor: a novel class of antifungal proteins against phytopathogenic fungi. Biochem Biophys Res Commun. 262:702–727. 1999.

Watanabe N., Hattori M., Yokoyama E., Isomura S., Ujita M., Hara A. Entomogenous fungi that produce 2,6-pyridine dicarboxylic acid (dipicolinic acid). Biosci Bioeng. 102:365–368. 2006.

Yen YH., Li PL., Wang CL., Wang SL. An antifungal protease produced by Pseudomonas aeruginosa M-1001 with shrimp and crab shell powder as a carbon source. Enzyme Microb Technol. 39:311–317. 2006.
Yu R., Song L., Zhao Y., Bin W., Wang L., Zhang H., Wu Y., Ye W., Yao X. Isolation and biological properties of polysaccharide CPS-1 from cultured. Cordyceps militaris. Fitoterapia. 75:465–472. 2004.
Go to : 
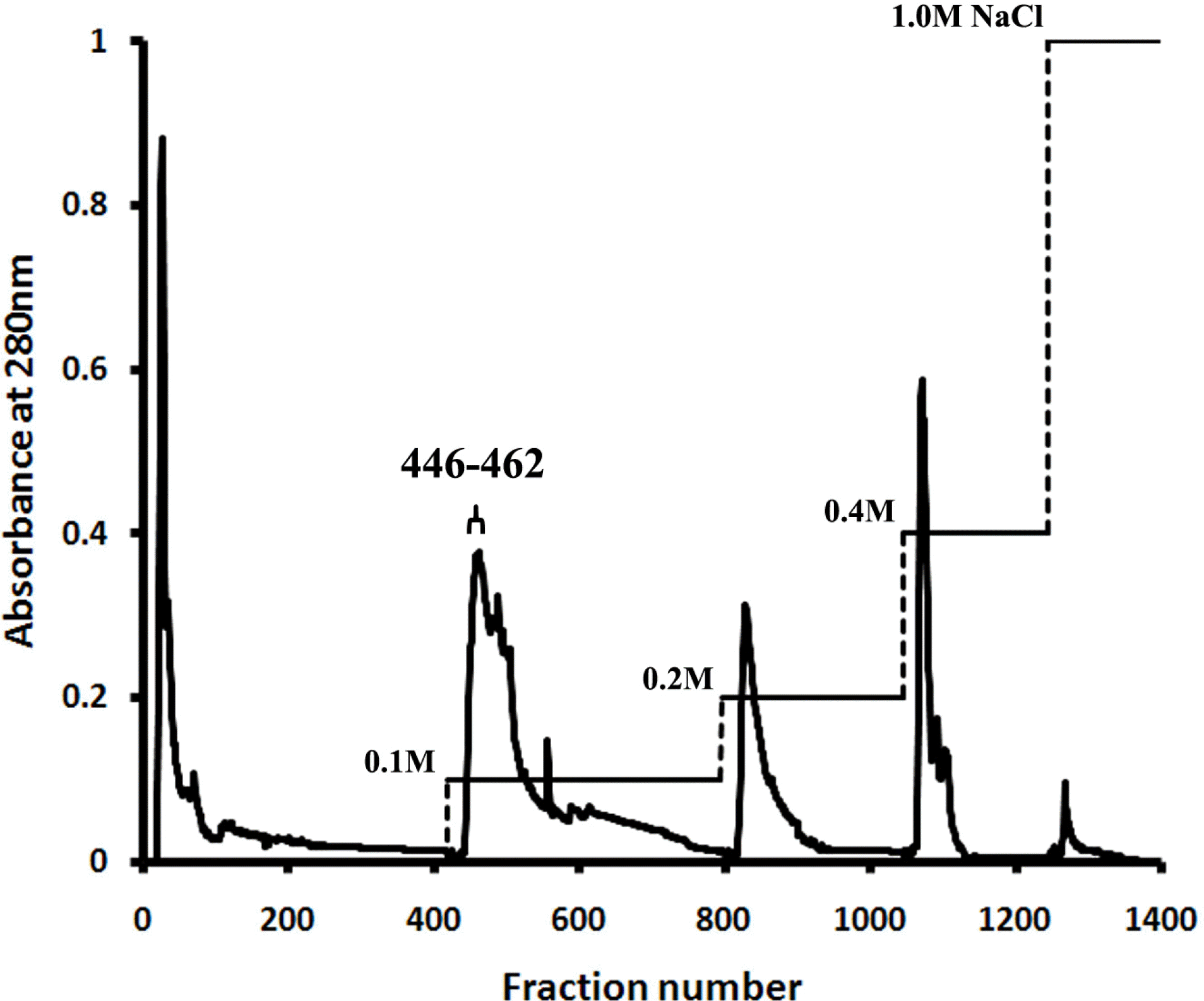 | Fig. 1.Elution profile of C. militaris protein extract applied to DEAE-Sepharose anion-exchange column chromatography, showing absorbance at 280 nm and NaCl concentration gradient. |
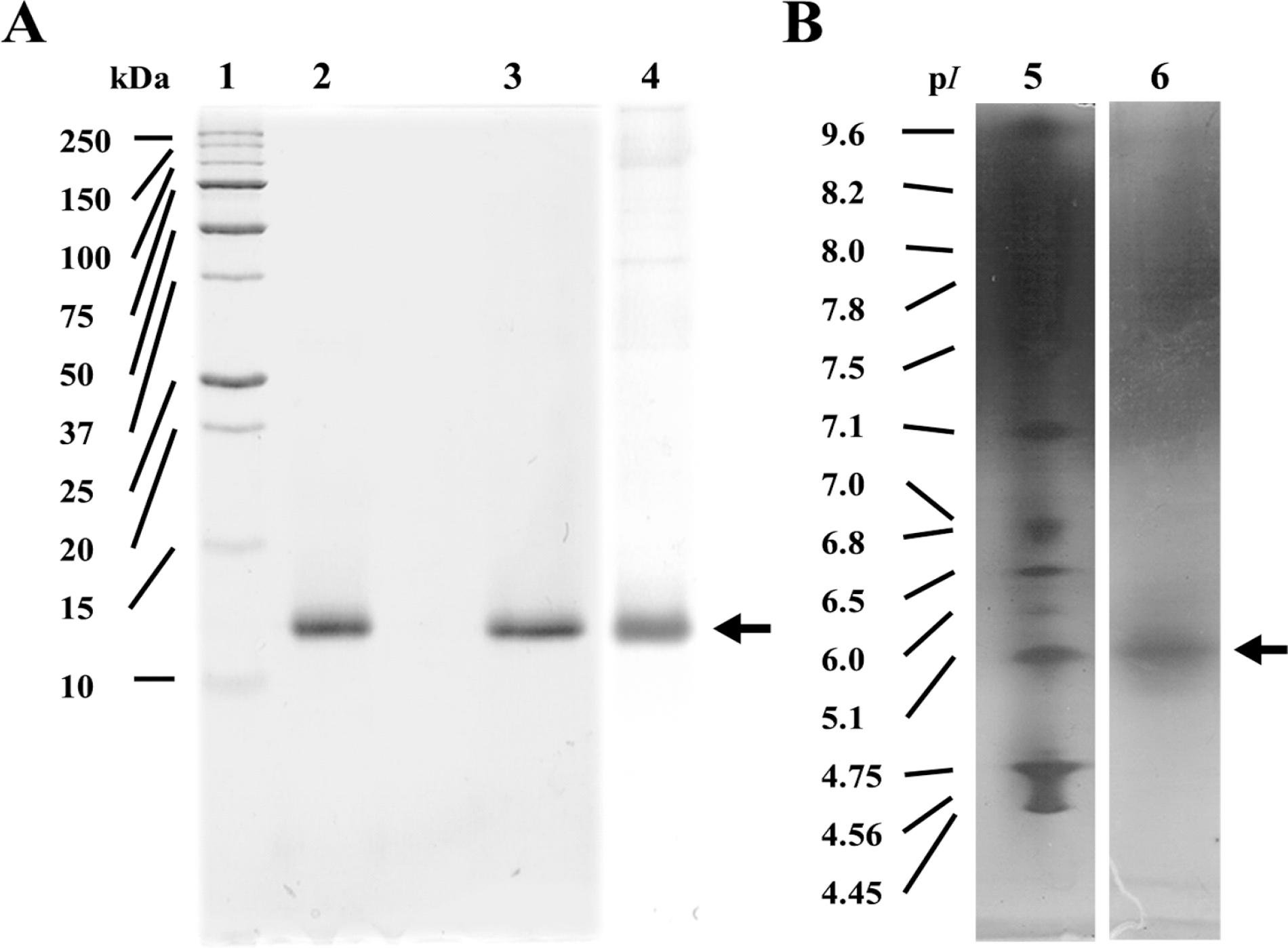 | Fig. 2.(A) SDS-PAGE and (B) IEF gel of purified CMP. Lane 1, standard protein markers; lane 2, CMP without 2-mercaptoethanol; lane 3, CMP with 2-mercaptoethanol; lane 4, CMP stained with silver nitrate; lane 5, standard pI markers; lane 6, CMP. |
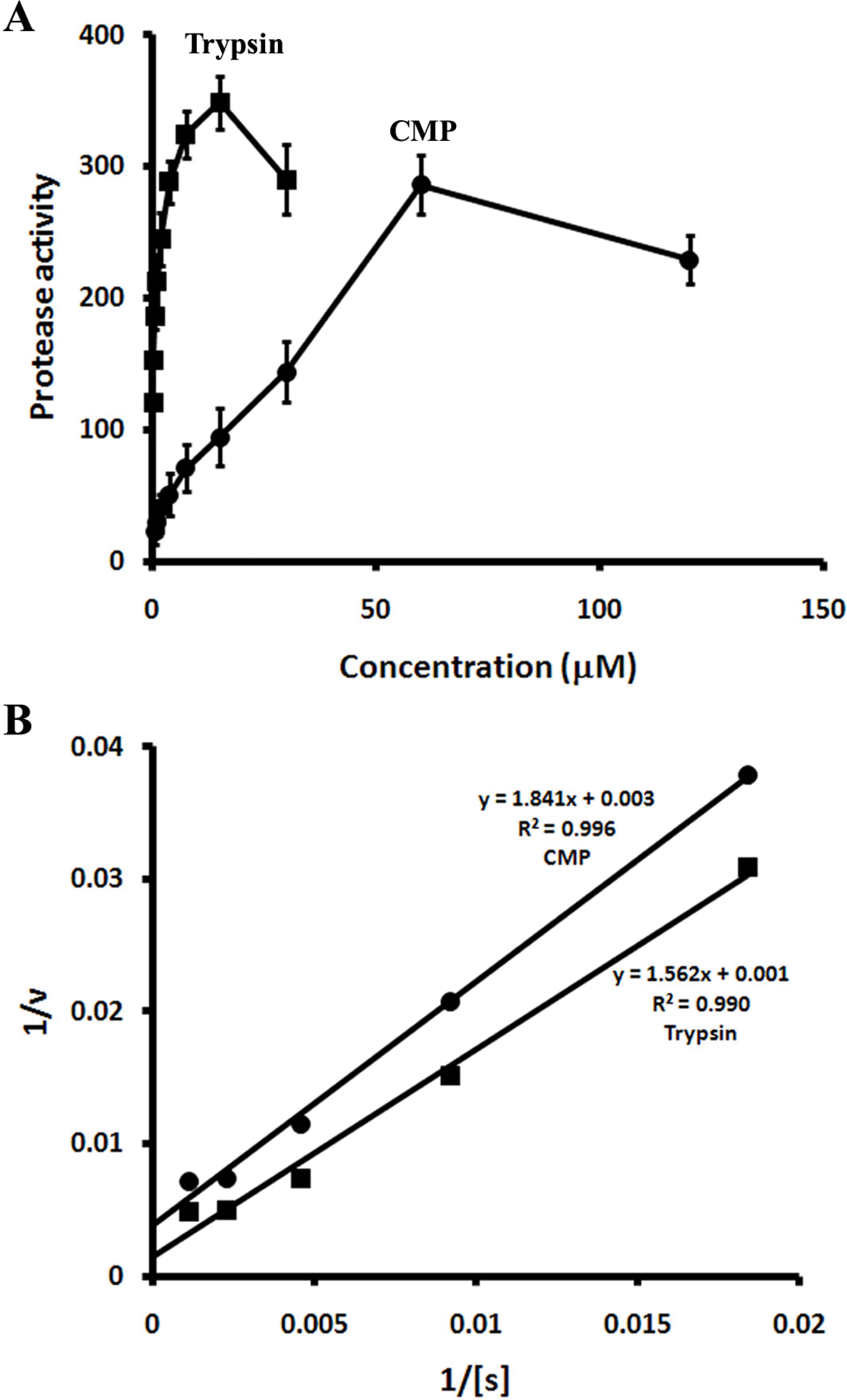 | Fig. 3.(A) Comparison of the protease activities of CMP (•) and trypsin (▪) using casein as a substrate. Protease activity is expressed as the fluorescence change per minute. (B) Lineweaver-Burk plots of casein hydrolysis by CMP (•) and trypsin (▪). |
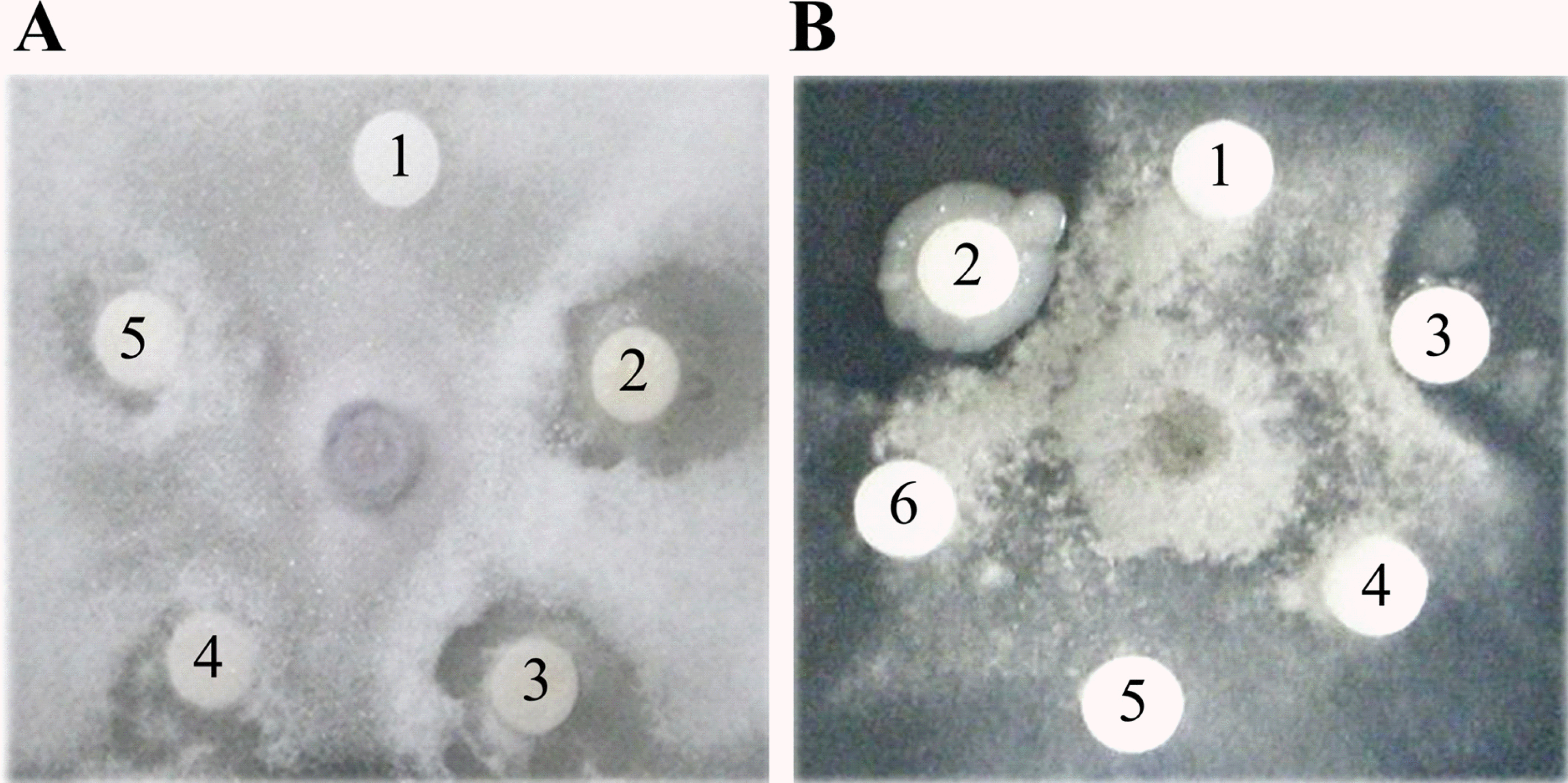 | Fig. 4.Antifungal activity of CMP against the growth of (A) F. oxysporum and (B) B. cinerea. Discs contained either buffer alone (10 mM Tris-HCl with 0.1 M NaCl, pH 7.0) or following concentrations of CMP in this buffer: disc 1, 0 μM; disc 2, 200 μM; disc 3, 40 μM; disc 4, 8 μM; disc 5, 1.6 μM; and disc 6, 0.32 μM. |
Table 1.
Summary of CMP purification from C. militaris
| Fraction | Protein (mg/ml) | Volume (ml) | Protease activitya | Specific activityb | Relative purification | Yield (%) |
|---|---|---|---|---|---|---|
| Crude protein extract | 12.6 | 7 | 471.1 | 37.4 | 1 | 100.0 |
| CMP | 0.2 | 15 | 130.4 | 652.0 | 17.4 | 3.4 |
Table 2.
Effects of pH, temperature, and inhibitors on the protease activity of CMP
Table 3.
Amino acid composition of CMP
| Amino acid | Molar ratio (%) |
|---|---|
| Asp/Asn | 9.63 |
| Glu/Gln | 11.01 |
| Ser | 6.22 |
| Thr | 9.43 |
| Gly | 10.74 |
| Val | 6.34 |
| Leu | 5.54 |
| His | 3.61 |
| Arg | 4.46 |
| Met | 1.97 |
| Tyr | 1.41 |
| Trp | 6.27 |
| Phe | 4.49 |
| Cys | 1.93 |
| Total | 100 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download