Abstract
Effects of quercetin, a kind of flavonoids, on the vasodilating actions were investigated. Among the mechanisms for quercetin-induced vasodilatation in rat aorta, the involvement with the Ca2+ activated K+ (KCa) channel was examined. Pretreatment with NE (5 μM) or KCl (60 mM) was carried out and then, the modulation by quercetin of the constriction was examined using rat aorta ring strips (3 mm) at 36.5°C. Quercetin (0.1 to 100 μM) relaxed the NE-induced vasoconstrictions in a concentration-dependent manner. NO synthesis (NOS) inhibitor, NG-monomethyl-L-arginine acetate (L-NMMA), at 100 μM reduced the quercetin (100 μM)-induced vasodilatation from 97.8±3.7% (n=10) to 78.0±11.6 % (n=5, p<0.05). Another NOS inhibitor, L-NG-nitro arginine methyl ester (L-NAME), at 100 μM also had the similar effect. In the presence of both 100 μM L-NMMA and 10 μM indomethacin, the quercetin-induced vasodilatation was further attenuated by 100 μM tetraethylammonium (TEA, a KCa channel inhibitor). Also TEA decreased the quercetin-induced vasodilatation in endothelium-denuded rat aorta. Used other KCa channel inhibitors, the quercetin-induced vasodilatation was attenuated by 0.3 μM apamin (a SK channel inhibitor), but not by 30 nM charybdotoxin (a BK and IK channel inhibitor). Quercetin caused a concentration-dependent vasodilatation, due to the endothelium-dependent and -independent actions. Also quercetin contributes to the vasodilatation selectively with SK channel on smooth muscle.
Go to : 
References
Ajay M., Gilani AH., Mustafa MR. Effects of flavonoids on vascular smooth muscle of the isolated rat thoracic aorta. Life Sci. 74:603–612. 2003.

Brayden JE., Nelson MT. Regulation of arterial tone by activation of calcium-dependent potassium channels. Science. 256:532–535. 1992.

Calderone V., Chericoni S., Martinelli C., Tetai L., Nardi A., Morelli I. Vasorelaxing effects of flavonoids: investigation on the possible involvement of potassium channels. Naunyn Schmiedebergs Arch Pharmacol. 370:290–298. 2004.

Campbell WB., Gebremedhin D., Pratt PF., Harder DR. Identification of epoxyeicosatrienoic acids as endothelium-derived hyperpolarizing factors. Circ Res. 78:415–423. 1996.

Carl A., Bayguinov O., Shuttleworth CW., Ward SM., Sanders KM. Role of Ca2+ activated K+ channels in electrical activity of longituidinal and circular muscle layers of canine colon. Am J Physiol. 268:619–627. 1995.
Catalin MF., Eugen B., Gabi H., Sebastian S., Ovidu B., Dimitrie DB. Multiple effects of tyrosine kinase inhibitors on vascular smooth muscle contraction. Eur J Pharmacol. 281:29–35. 1995.
Chen CK., Pace-Asciak CR. Vasorelaxing activity of reveratrol and quercetin in isolated rat aorta. Gen Pharmacol. 27:363–366. 1996.
Chen GF., Suzuki H. Calcium dependency of the endotheliumdependent hyperpolarization in smooth muscle cells of the rabbit carotid artery. J Physiol (Lond). 421:521–534. 1990.

Congolludo A., Frazziano G., Briones AM., Cobeno L., Moreno L., Lodi F. The dietary flavonoid quercetin activates BKCa currents in coronary arteries via production of H2O2. Role in vasodilatation. Cardiovasc Res. 73:424–431. 2007.
Duarte J., Perez-Vizcaino F., Zarzurelo J., Tamargo J. Vasodilator effects of quercetin in isolated rat vascular smooth muscle. Eur J Pharmacol. 239:1–7. 1993.

Duarte J., Perez-Vizcaino F., Utrilla P., Jiménez J., Tamargo J., Zarzuelo A. Vasodilatory effects of flavonoids in rat aortic smooth muscle. Structure-activity relation ship. Gen Pharmacol. 24:857–862. 1993.
Formica JV., Regelson W. Review of the biology of quercetin and related bioflavonoids. Food Chem Toxicol. 33:1061–1080. 1995.

Fukao M., Hattori Y., Kanno M., Sakuma I., Kitabatake A. Sources of Ca2+ in relation to generation of acetylcholine-induced endothelium-dependent hyperpolarization in rat mesenteric artery. Br J Pharmacol. 120:1328–1334. 1997.
Fusi F., Saponara S., Pessina F., Gorelli B., Sgaragli G. Effects of quercetin and rutin on vascular preparations. A comparison between mechanichal and electrophysiological phenomena. Eur J Nutr. 42:10–17. 2003.
Garcia-Pascual A., Labadia A., Jimenez E., Costa G. Endothelium-dependent relaxation to acetylcholine in bovine oviductal arteries: mediation by nitric oxide and changes in apamin-sensitive K+ conductance. Br J Pharmacol. 105:429–435. 1995.
Granado-Serrano AB., Angeles Martin M., Bravo L., Gaya L., Ramos S. Time-course regulation of quercetin on cell survival/proliferation pathways in human hepatoma cells. Mol Nutr Food Res. 52:457–464. 2008.

Hagiwara M., Inoue S., Tanaka T., Nunoki K., Ito M., Hidaka H. Differential effects of flavonoids as inhibitors of tyrosine protein kinases and serin/threonine protein kinases. Biochem Pharmacol. 37:2987–2992. 1988.
Hertog MG., Feskens EJ., Hollman PC., Katan MB., Kromhaut D. Dietary antioxidant flavonoids and risk of coronary heart disease: the zutphen elderly study. Lancet. 342:1007–1011. 1993b.

Hertog MG., Hollman PC., Katan MB., Kromhout D. Intake of potentially anticarcinogenic flavonoids and determinants in adults in the Netherlands. Nutr Cancer. 20:21–29. 1993a.
Kubota Y., Tanaka T., Umegaki K. Ginkgo biloba extract-induced relaxation of rat aorta is associated with increase in endothelial intracellular calcium level. Life Sci. 69:2327–2336. 2001.

Kwon SH., Nam JI., Kim SH., Kim JH., Yoon JH., Kim KS. Kaempferol and quercetin, essential ingredients in Ginkgo biloba extrat, inhibit interleukin-1β-induced MUC5AC gene expression in human airway epithelial cells. Phytother Res. 2009. (in press).
Lin CW., Hou WC., Shen SC., Juan SH., Ko CH., Wang LM., Chen YC. Quercetin inhibition of tumor invasion via suppressing PKCδ/ERK/AP-1-dependent matrix metalloproteinase-9 activation in breast carcinoma cells. Carcinogenesis. 29:1807–1815. 2008.
Martinez-Orgado J., Gonzalez R., Alonso MJ., Marin J. Nitric oxide-dependent and -independent mechanisms in the relaxation elicited by acetylcholine in fetal rat aorta. Life Sci. 64:269–277. 1999.
Mitamura M., Boussery K., Horie S., Murayama T., Voorde JV. Vasorelaxing effect of mesaconitine, an alkaloid from aconicum japonicum, on rat small gastric artery: possible involvement of endothelium-derived hyperpolarizing factor. Jpn J Pharmacol. 89:380–387. 2002.
Moussazadeh M., Haimovich B. Protein kinase C-delta activation and tyrosine phophorylation in platelets. FEBS Lett. 438:225–230. 1998.
Murota K., Terao J. Antioxidative flavonoid quercetin; implication of its intestinal absorption and metabolism. Arch Biochem Biophys. 417:12–17. 2003.

Murphy ME., Brayden JE. Apamin-sensitive K+ channels mediate an endothelium-dependent hyperporalization in rabbit mesenteric arteries. J Physiol (Lond). 489:723–724. 1995.
Murthy KS., Grider JR., Kuemmerle JF., Makhlouf GM. Sustained muscle contraction induced by agonists, growth factors, and Ca2+ mediated by distinct PKC isozymes. Am J Physiol Gastrointest Liver Physiol. 279:G201–G210. 2000.
Ndiaye M., Chataigneau T., Andriantsitohaina R., Stoclet JC., Schini-Kerth VB. Red wine polyphenols cause endothelium-dependent EDHF-mediated relaxations in porcine coronary arteries via a redox-sensitive mechanisms. Biochem Biophys Res Commun. 310:371–377. 2003.
Neylon CB., Lang RJ., Fu Y., Bobik A., Reinhart PH. Molecular cloning and characterization of the intermediate-conductance Ca2+-activated K+ channel in vascular smooth muscle: relationship between K (Ca) channel diversity and smooth muscle cell function. Circ Res. 85:33–43. 1999.

Nishida S., Satoh H. Comparative vasodilating actions among terpenoids and flavonoids contained in Ginkgo biloba extract. Clin Chim Acta. 339:129–133. 2004.

Perez-Vizcaino F., Ibarra M., Cogolludo AL., Duarte J., Zaragoza-Arnaez F., Moreno L. Endothelium-Independent Vasodilator effects of the flavonoid quercetin and its methylated metabolites in rat conductance and resistance arteries. J Pharmacol Exp Ther. 302:66–72. 2002.

Satoh H. Comparative electropharmacological actions of some constituents from Ginkgo biloba extract in guinea pig ventricular cardiomyocytes. Evid Based Complement Altern Med. 1:277–284. 2005.
Satoh H., Nishida S. Electropharmacological actions of Ginkgo biloba extract on vascular smooth and heart muscles. Clin Chim Acta. 342:13–22. 2004.

Satoh H., Sperelakis N. Calcium and potassium currents in cultured in rat aortic vascular smooth muscle cell lines. Sperelakis N, editor. ed,. Ion Channels of Vascular Smooth Muscle Cells and Endothelial Cells. Academic Press;New York: p. p. 55–63. 1991.
Satoh H., Sperelakis N. Modulation of L-typeCa2+ current by isoprenaline, carbachol and phorbol ester in cultured rat aortic vascular smooth muscle (A7r5) cells. Gen Pharmacol. 26:369–379. 1995.
Satoh H. Modulation of Ca2+-activated K+ current by isoprenaline, carbachol, and phorbol ester in cultured (and fresh) rat aortic vascular smooth muscle cells. Gen Pharmacol. 27:319–324. 1996.
Shin CM., Lin H., Liag YC., Lee WS., Bi WF., Juan SH. Concentration-dependent differential effects of quercetin on rat aortic smooth muscle cell. Eur J Pharmacol. 496:41–44. 2004.
Sticher O. Quality of Ginkgo preparations. Planta Med. 59:2–11. 1993.
Vogalis F., Zhang Y., Goyal RK. An intermediate conductance K+ channel in the cell membrane of mouse intestinal smooth musucle. Biochim Biophys Acta. 1371:309–316. 1998.
Go to : 
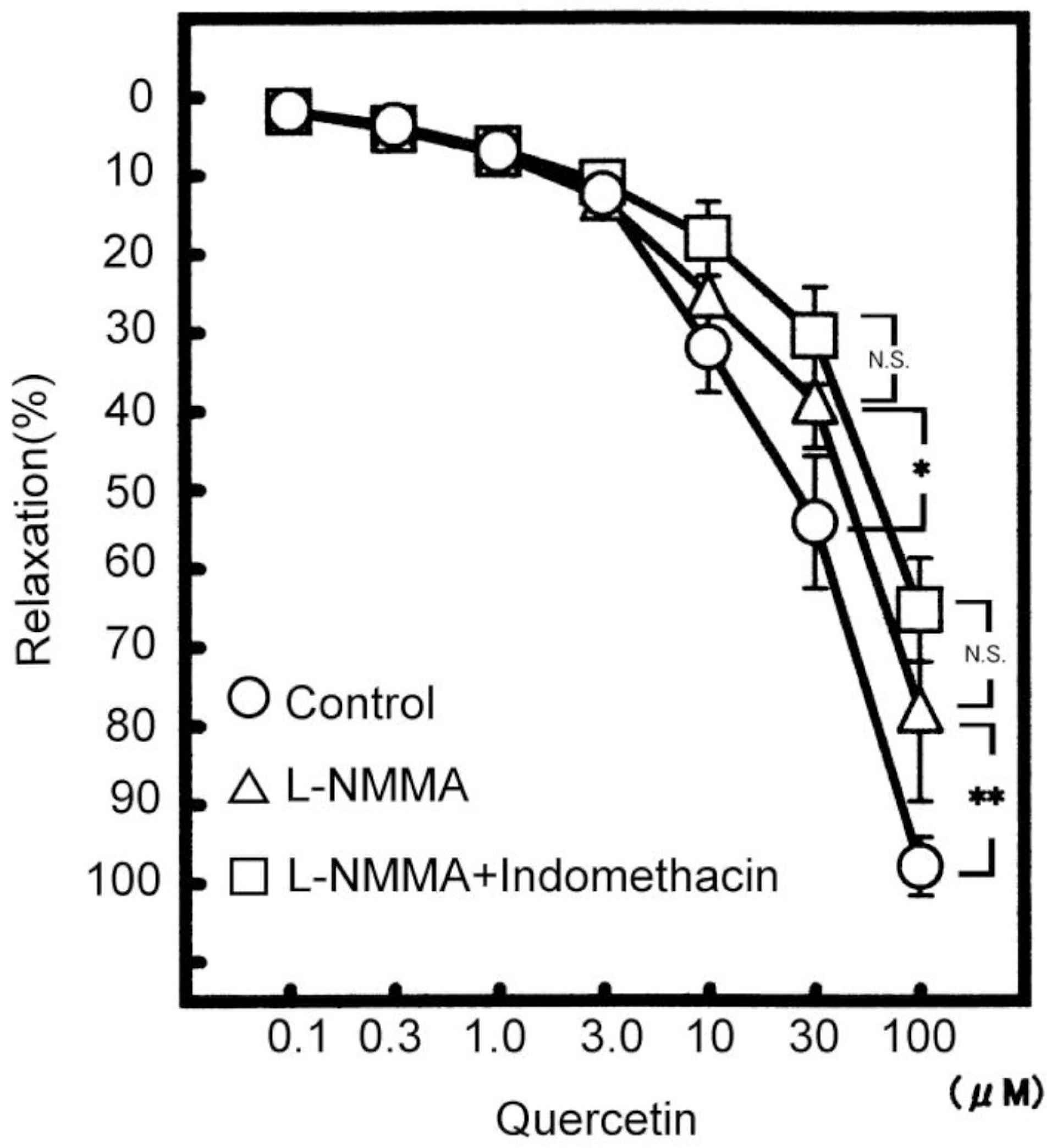 | Fig. 1.Concentration-dependent vasodilatation by cumulative administrations of quercetin. Symbols used are control (open circles, n=10), pretreatment with L-NMMA (triangles, n=5), L-NMMA and indomethacin (squares, n=5). Values (%) are represented as mean±S.E.M. ∗p<0.05, ∗∗p<0.01, with respect to control value. |
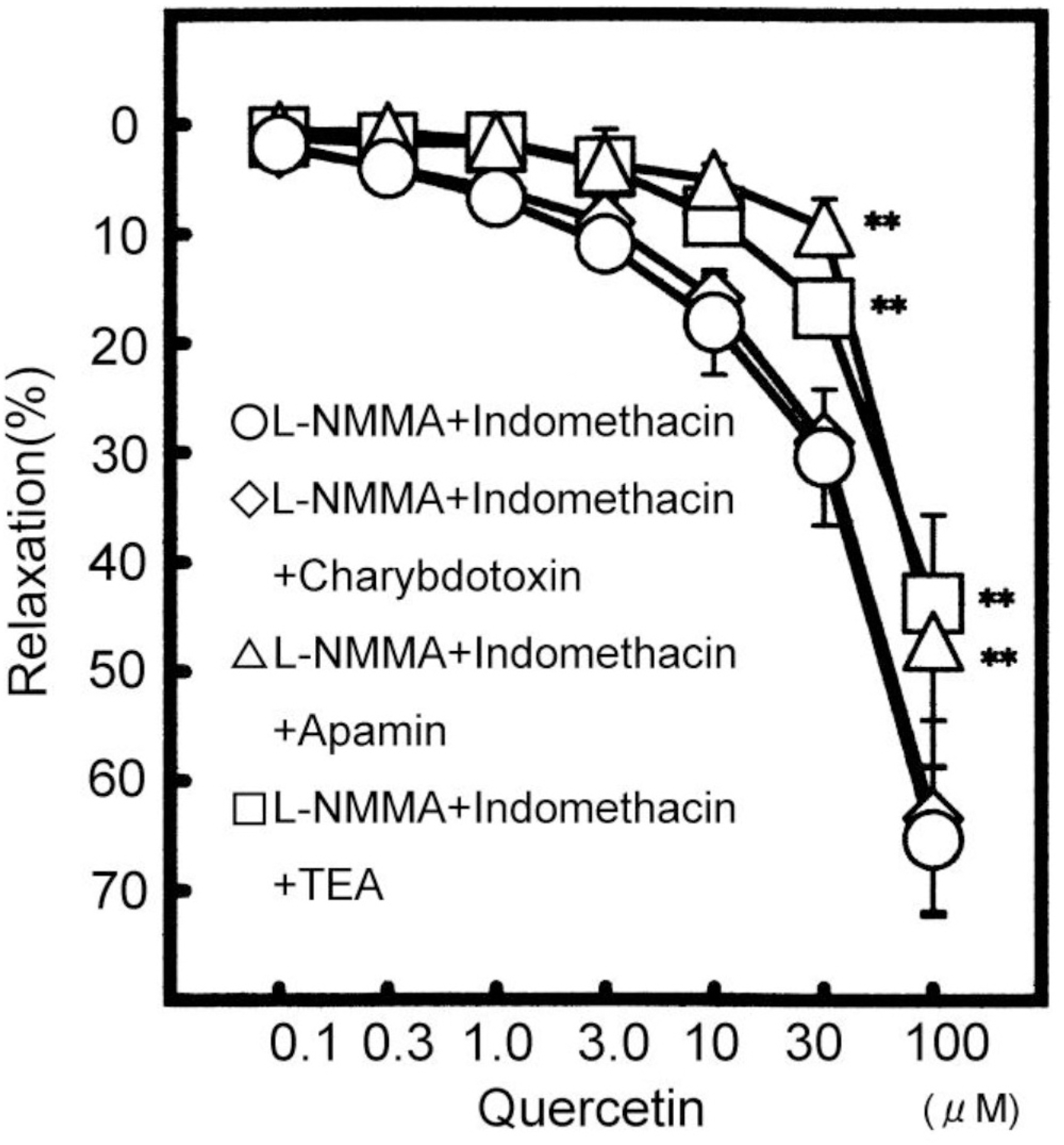 | Fig. 2.Modulation of L-NMMA and indomethacin resistant relaxation at 100 μM quercetin. The vasodilatation in the presence of L-NMMA and indomedhacin (n=5) means as control value in this graph. The vasodilatations by L-NMMA and indomethacin plus TEA (n=5), by L-NMMA and indomethacin plus apamin (n=5), and by L-NMMA and indomethacin plus charybdotoxin (n=5) were compared with that of L-NMMA and indomedhacin. Values (%) are represented as mean±S.E.M. ∗∗p<0.01, with respect to control value. |
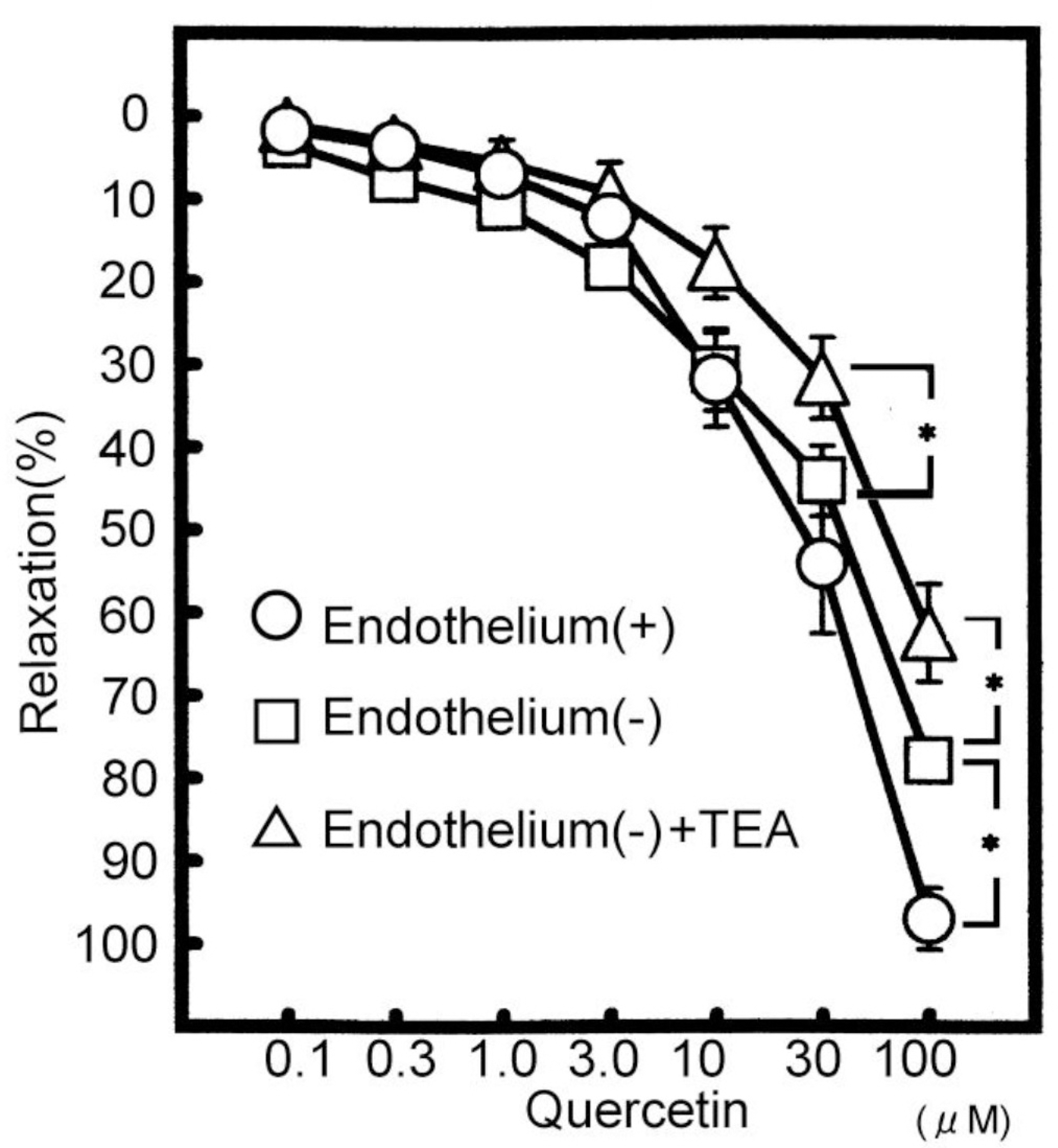 | Fig. 3.Modulation of quercetin (100 μM)-induced vasodilatation (n=10) in the removal of endothelium (n=5) and the removal of endothelium plus TEA (n=5). Values (%) are represented as mean± S.E.M. ∗p<0.05, with respect to control value. |
Table 1.
Modulation of the quercetin-induced vasodilatation
| Quercetin (μM) | ||||||||
|---|---|---|---|---|---|---|---|---|
| N | 0.1 | 0.3 | 1 | 3 | 10 | 30 | 100 | |
| Control | 10 | 1.7±0.53 | 3.6±0.90a | 6.9±0.91a | 12.4±1.1b | 32.0±5.7c | 54.1±4.4c | 97.8±3.7c |
| L-NMMA 100 μM | 5 | 1.9±0.55 | 4.1±1.3 | 7.3±1.8 | 13.0±2.4 | 25.4±4.8 | 38.7±6.0x | 78.0±11.6x |
| L-NAME 100 μM | 5 | 1.2±0.89 | 3.0±1.1 | 7.0±2.0 | 13.2±3.7 | 21.1±4.6x | 33.0±5.3y | 69.5±6.1y |
| Endothelium-denuded | 5 | 1.8±0.55 | 3.8±1.6 | 8.5±1.9 | 13.0±2.6 | 30.8±4.9 | 44.2±4.2x | 77.9±2.4x |
Values (%) represent mean±S.E.M. a and x: p<0.05, b and y: p<0.01, c: p<0.001. Symbols of a, b, and c mean significant difference in comparison between effect of quercetin itself at each concentration and the maximal contraction induced by NE. Symbols of x and y mean significant difference as compared with control (quercetin alone).




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download