Abstract
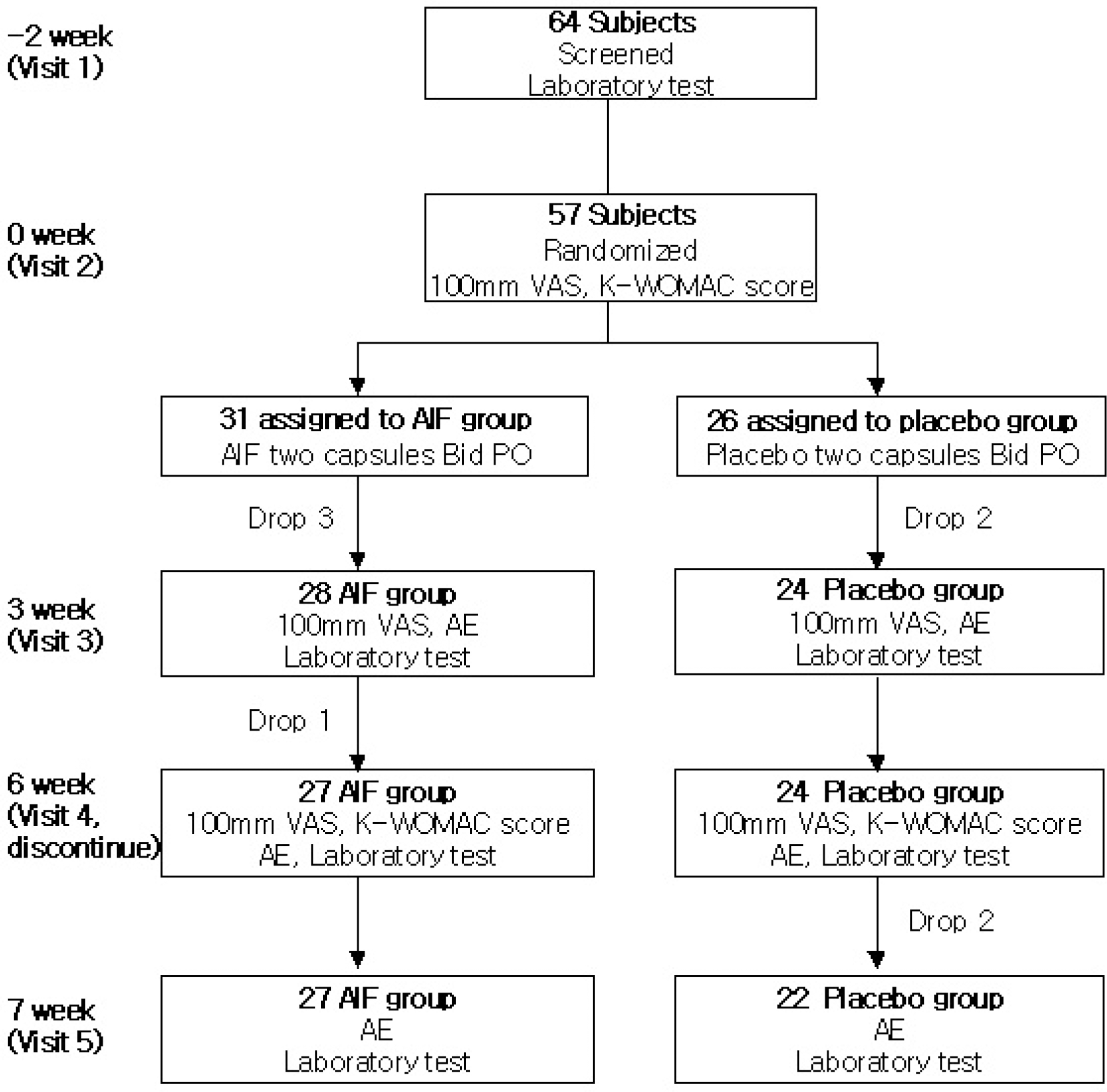
Anti-inflammatory factor (AIF) is a water soluble extract of three herbs, Panax notoginseng (Burk.) F. H. Chen, Rehmannia glutinosa Libosch and Eleutherococcus senticosus. The present study aimed to investigate the safety and efficacy of herb extracts, AIF, on Korean knee osteoarthritis patients for six weeks. Fifty seven patients with knee osteoarthritis, ranging from 43 to 73 years of age, who fulfilled the “American College of Rheumatology” (ACR) classification of idiopathic osteoarthritis of knee and radiographic criteria were randomly selected and enrolled for the study. After initial screening and resting period, two capsules each of AIF (Each capsule contains; 400 mg) and similar identical placebo were administered twice a day to both groups. Pain intensity at second, fourth, and sixth weeks of study as well as one week after discontinuation of drugs was assessed by using 100 mm visual analogue scale (VAS). Changes in the Korean version of the Western Ontario and McMaster Universities (K-WOMAC) index score were compared at the initiation and completion of the study. VAS assessed by patients were significantly reduced (at visit 2; 54.64±14.72, at visit 4, 37.32±16.58, p<0.001) after AIF administration. Results showed an improvement in the physical function of K-WOMAC scale which was significantly higher (p=0.013) in AIF than placebo group, and decreases of total K-WOMAC score were also significantly higher (p=0.030) in AIF groups than placebo group. No serious adverse effect was observed, and there was no difference in incidence of adverse effect between AIF and placebo groups. In this population of Korean patients with knee osteoarthritis, AIF was found to be safe, tolerable and effective for symptomatic improvement of pain and physical function.
Go to : 
REFERENCES
Altman R., Asch E., Bloch D., Bole G., Borenstein D., Brandt K., Christy W., Cooke TD., Greenwald R., Hochberg M. Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum. 29:1039–1049. 1986.
Bae SC., Lee HS., Yun HR., Kim TH., Yoo DH., Kim SY. Cross-cultural adaptation and validation of Korean Western Ontario and McMaster Universities (WOMAC) and Lequesne osteoarthritis indices for clinical research. Osteoarthritis Cartilage. 9:746–750. 2001.

Chang SH., Choi Y., Park JA., Jung DS., Shin J., Yang JH., Ko SY., Kim SW., Kim JK. Anti-inflammatory effects of BT-201, an n-butanol extract of Panax notoginseng, observed in vitro and in a collagen-induced arthritis model. Clin Nutr. 26:785–791. 2007.

Chang SH., Sung HC., Choi Y., Ko SY., Lee BE., Baek DH., Kim SW., Kim JK. Suppressive effect of AIF, a water extract from three herbs, on collagen-induced arthritis in mice. Int Immunopharmacol. 5:1365–1372. 2005.

Christensen R., Bartels EM., Astrup A., Bliddal H. Symptomatic efficacy of avocado-soybean unsaponifiables (ASU) in osteoarthritis (OA) patients: a meta-analysis of randomized controlled trials. Osteoarthritis Cartilage. 16:399–408. 2008.

Clegg DO., Reda DJ., Harris CL., Klein MA., O'Dell JR., Hooper MM., Bradley JD., Bingham CO., Weisman MH., Jackson CG., Lane NE., Cush JJ., Moreland LW., Schumacher HR Jr. Oddis CV, Wolfe F, Molitor JA, Yocum DE, Schnitzer TJ, Furst DE, Sawitzke AD, Shi H, Brandt KD, Moskowitz RW, Williams HJ. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 354:795–808. 2006.
Garstang SV., Stitik TP. Osteoarthritis: epidemiology, risk factors, and pathophysiology. Am J Phys Med Rehabil. 85:S11–12. 2006.
Kellgren JH., Lawrence JS. Radiological assessment of osteoarthrosis. Ann Rheum Dis. 16:494–502. 1957.

Kim HM., An CS., Jung KY., Choo YK., Park JK., Nam SY. Rehmannia glutinosa inhibits tumour necrosis factor-alpha and interleukin-1 secretion from mouse astrocytes. Pharmacol Res. 40:171–176. 1999.
Lawrence RC., Helmick CG., Arnett FC., Deyo RA., Felson DT., Giannini EH., Heyse SP., Hirsch R., Hochberg MC., Hunder GG., Liang MH., Pillemer SR., Steen VD., Wolfe F. Estimates of the prevalence of arthritis and selected musculoskeletal disorders in the United States. Arthritis Rheum. 41:778–799. 1998.

Ling S., Nheu L., Dai A., Guo Z., Komesaroff P. Effects of four medicinal herbs on human vascular endothelial cells in culture. Int J Cardiol. 128:350–358. 2008.

Martel-Pelletier J. Pathophysiology of osteoarthritis. Osteoarthritis Cartilage. 12(Suppl A):S31–33. 2004.

McAlindon TE., LaValley MP., Gulin JP., Felson DT. Glucosamine and chondroitin for treatment of osteoarthritis: a systematic quality assessment and meta-analysis. Jama. 283:1469–1475. 2000.
Ofman JJ., MacLean CH., Straus WL., Morton SC., Berger ML., Roth EA., Shekelle P. A metaanalysis of severe upper gastrointestinal complications of nonsteroidal antiinflammatory drugs. J Rheumatol. 29:804–812. 2002.
Peat G., McCarney R., Croft P. Knee pain and osteoarthritis in older adults: a review of community burden and current use of primary health care. Ann Rheum Dis. 60:91–97. 2001.

Pelletier JP., Yaron M., Haraoui B., Cohen P., Nahir MA., Choquette D., Wigler I., Rosner IA., Beaulieu AD. Efficacy and safety of diacerein in osteoarthritis of the knee: a double-blind, placebo-controlled trial. The Diacerein Study Group. Arthritis Rheum. 43:2339–2348. 2000.
Spiker B. Kraemer HC, Scott DT, Gross RT. Design issues in a randomized clinical trial of a behavioral intervention: Insights from the infant health and development program. J Dev Behav Pedatr. 12:386–393. 1992.
World Medical Association Declaration of Helsinki. Recommendations Guiding Medical Doctors in Biomedical Research Involving Human Subjects. Available at:. http://www.net.
Zhang W., Moskowitz RW., Nuki G., Abramson S., Altman RD., Arden N., Bierma-Zeinstra S., Brandt KD., Croft P., Doherty M., Dougados M., Hochberg M., Hunter DJ., Kwoh K., Lohmander LS., Tugwell P. OARSI Recommendations for the management of hip and knee osteoarthritis. part I: critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage. 15:981–1000. 2007.

Go to : 
Table 1.
Patient baseline characteristics
| Demographics | AIF N=31 | Placebo N=26 | p value | |
|---|---|---|---|---|
| Age, yr | 59.90 (7.54) | 58.92 (7.06) | >0.05∗ | |
| Gender, no (%) | ||||
| Female | 28 (90.32) | 24 (92.31) | >0.05† | |
| Male | 3 (9.68) | 2 (7.69) | ||
| Body weight (kg) | 58.54 (7.30) | 63.70 (9.17) | 0.022∗ | |
| Height (cm) | 155.68 (6.33) | 155.55 (5.42) | >0.05∗ | |
| Previous medical history | ||||
| Yes | 23 (74.19) | 17 (65.38) | >0.05‡ | |
| No | 8 (25.81) | 9 (34.62) | ||
| Radiographic exam, no (%) | ||||
| Right | I | 17 (54.84) | 16 (61.54) | >0.05‡ |
| II | 14 (45.16) | 10 (38.46) | ||
| III | 0 (0.00) | 0 (0.00) | ||
| IV | 0 (0.00) | 0 (0.00) | ||
| Left | I | 18 (58.06) | 14 (53.85) | >0.05‡ |
| II | 12 (38.71) | 12 (46.15) | ||
| III | 1 (3.23) | 0 (0.00) | ||
| IV | 0 (0.00) | 0 (0.00) | ||
Table 2.
Summary of adverse events (AEs)
Table 3.
Changes of VAS scores as assessed by patients
| Before administration | After administration | p value† | |||
|---|---|---|---|---|---|
| Week −2 | Week 0 | Week 3 | Week 6 | ||
| AIF | N=28 | N=28 | N=28 | N=28 | |
| 54.29 (14.32) | 54.64 (14.72) | 46.96 (16.46) | 37.32 (16.58) | <0.01 | |
| Placebo | N=24 | N=24 | N=24 | N=24 | |
| 52.92 (15.67) | 55.21 (15.00) | 51.04 (18.41) | 42.08 (18.88) | <0.01 | |
| p value∗ | 0.89 | 0.34 | |||
Table 4.
Changes of K-WOMAC scale scores
| Before administration | After administration | Δ | p value∗ | ||
|---|---|---|---|---|---|
| Week 0 | Week 6 | ||||
| N=27 | N=27 | ||||
| AIF | Pain | 6.22 (3.88) | 4.33 (3.03) | 1.89 (3.80) | <0.05 |
| Stiffness | 2.96 (3.01) | 2.22 (1.85) | 0.74 (1.72) | <0.05 | |
| Physical function | 24.04 (13.30) | 15.26 (13.18) | 8.78 (10.84) | <0.05 | |
| Total score | 33.22 (17.87) | 21.81 (17.33) | 11.41 (14.44) | <0.05 | |
| N=24 | N=24 | ||||
| Placebo | Pain | 5.22 (3.01) | 4.22 (3.52) | 1.00 (3.71) | 0.21 |
| Stiffness | 2.57 (1.81) | 2.26 (1.66) | 0.31 (1.69) | 0.40 | |
| Physical function | 20.17 (10.99) | 18.09 (12.14) | 2.09 (6.67) | 0.15 | |
| Total score | 27.96 (14.08) | 24.57 (16.67) | 3.39 (10.16) | 0.12 | |
| p value∗ | 0.41 | ||||
| 0.37 | |||||
| <0.05 | |||||
| <0.05 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download