Abstract
Purpose
To evaluate the 3-month natural course of recurrent macular edema secondary to branch retinal vein occlusion (BRVO) treated with intravitreal bevacizumab.
Methods
This retrospective, observational study included 36 eyes with macular edema secondary to BRVO. All patients were initially treated with intravitreal bevacizumab for macular edema. Recurrence of macular edema was either not treated (untreated group) or treated with a single intravitreal bevacizumab injection (treated group). Central foveal thickness (CFT) and best-corrected visual acuity (BCVA) were compared at the time of recurrence and 3 months later.
Results
At the time of recurrence, the mean CFT and logarithm of the minimum angle of resolution BCVA were 484.9 ± 124.1 µm and 0.58 ± 0.26 in the untreated group (n = 19) and 456.3 ± 126.8 µm and 0.51 ± 0.21 in the treated group (n = 17), respectively. Three months later, the mean CFT and BCVA had changed to 493.7 ± 123.9 µm and 0.62 ± 0.29 in the untreated group and 294.7 ± 104.4 µm and 0.40 ± 0.24 in the treated group, respectively. The differences in CFT and BCVA between the two time points were not significant in the untreated group (p = 0.106 and p = 0.687, respectively), whereas statistically significant differences were noted in the treated group (p = 0.002 and p < 0.001, respectively).
It has been reported that macular edema (ME) secondary to branch retinal vein occlusion (BRVO) has a relatively favorable natural course. ME generally resolves and visual acuity generally improves without intervention [123]. Previous optical coherence tomography (OCT) studies have also shown a marked decrease in ME with no treatment during the first several months following BRVO [4567]. Some studies have failed to demonstrate a significant beneficial effect of either intravitreal triamcinolone [47] or intravitreal bevacizumab [67] on visual outcomes in acute BRVO because of this favorable natural course.
Intravitreal anti-vascular endothelial growth factor (VEGF) has been advocated as an effective therapy for ME secondary to BRVO [891011121314], but recurrent ME has been frequently noted following anti-VEGF therapy [891115161718]. Therefore, treating recurrent ME has been raised as an important issue for BRVO patients [1920]. However, the natural course of recurrent ME following intravitreal anti-VEGF has not yet been examined.
The purpose of the present study was to evaluate the short-term natural course of recurrent ME following intravitreal bevacizumab injection. The natural course was compared with that following a single intravitreal bevacizumab injection.
This retrospective, observational case series was performed at a single center, and study conduct adhered to the tenets of the Declaration of Helsinki. The study was approved by the institutional review board of Kim's Eye Hospital.
We conducted a retrospective review of the medical records of patients who were newly diagnosed with ME secondary to BRVO between January 2010 and January 2013. The inclusion criteria were as follows: (1) previously received an intravitreal bevacizumab injection to treat ME, (2) resolution of ME (less than 250 µm of central foveal thickness [CFT] confirmed on OCT after a bevacizumab injection, (3) recurrence of ME noted during a follow-up visit, and (4) 3-month or longer follow-up after recurrence of ME, regardless of the number of recurrences. The cost, efficacy, and possible complications of available treatment modalities for recurrent ME were discussed with each patient and was untreated when patients denied receiving additional treatment.
Exclusion criteria were as follows: (1) recurrence of ME accompanied by definite progression of retinal vein occlusion (e.g., increase in retinal hemorrhage size or severity), as noted on fundus examination, (2) evidence of vitreomacular traction, (3) severe media opacity that may have interfered with OCT imaging, (4) previous history of any treatment for ME other than intravitreal bevacizumab, (5) history of retinal photocoagulation, (6) previous vitreoretinal surgery, (7) intraocular inflammation, and (8) any other disorder that may have influenced macular function, including exudative age-related macular degeneration, proliferative diabetic retinopathy and presence of an epiretinal membrane or macular hole.
All subjects underwent a comprehensive ophthalmologic examination, including best-corrected visual acuity (BCVA) measurement, 90-diopter lens slit-lamp biomicroscopy, fundus photography, fluorescein angiography, and spectral domain OCT (Spectral OCT/SLO; OTI Ophthalmic Technologies, Miami, FL, USA). The regular follow-up examinations were performed within 1 to 4 months at the discretion of the treating physician. Because evaluation of macular volume was not routinely performed, CFT measurements were used in the analyses. The vertical distance between the internal limiting membrane and the retinal pigment epithelium at the foveal center was measured in both horizontal and vertical OCT images centered on the fovea. The mean CFT value of these two images was used in the analyses. Visual acuity data were converted to the logarithm of the minimum angle of resolution (logMAR) for the analyses. Foveal capillary nonperfusion was considered to be present when fluorescein angiography revealed a broken perifoveal capillary ring at the border of the foveal avascular zone that was associated with a distinct area of capillary nonperfusion within one disc diameter of the foveal center [21].
To compare the natural course of recurrent ME with outcomes of bevacizumab-treated recurrent ME, eyes with and without treatment were included in the study. The untreated group received no therapy for recurrent ME for at least 3 months. Eyes that had ME recurrence between 9 and 13 months after the initial diagnosis and were treated with a single intravitreal bevacizumab injection were included in the treated group. All other inclusion and exclusion criteria of both study groups were the same.
In each treated or untreated group, the BCVA and CFT 3 months after ME recurrence were compared to measurements when the ME recurrence was initially noted. Patient age, sex, proportion of eyes with foveal capillary nonperfusion, mean number of bevacizumab injections before ME recurrence, time between symptom onset and ME recurrence, and time between the last bevacizumab injection and ME recurrence were compared between the treated and the untreated groups. The BCVA and CFT before ME recurrence and 3 months after ME recurrence were also compared between the two groups.
Statistical analyses were performed with the commercially available software package SPSS ver. 12.0 for Windows (SPSS Inc., Chicago, IL, USA). Comparisons of values between different time points were performed using the paired t-test. Comparisons between the treated group and the untreated group were performed using independent sample t-tests with or without a Bonferroni's correction or Fisher's exact test. A p-value < 0.05 was considered significant.
Nineteen eyes from 19 patients (5 males and 14 females) were included in the untreated group (Table 1). Mean patient age was 67.7 ± 8.5 years (range, 53 to 84 years) and no eye had a history of retinal photocoagulation. Foveal capillary nonperfusion was noted at diagnosis in nine eyes (47.4%). Eyes previously treated with intravitreal bevacizumab underwent an average of 2.3 ± 1.1 injections (range, 1 to 5 injections) before the ME recurrence that was left untreated. Time between symptom onset and recurrence averaged 10.4 ± 4.8 months (range, 5 to 20 months) and time between the last bevacizumab injection and ME recurrence averaged 5.1 ± 1.9 months (range, 3 to 9 months).
Seventeen eyes from 17 patients (10 males and 7 females) were included in the treated group (Table 1). Mean patient age was 63.8 ± 5.6 years (range, 55 to 73 years). Foveal capillary nonperfusion was noted in seven eyes (41.2%) at the time of diagnosis. Eyes were previously treated with intravitreal bevacizumab and underwent an average of 2.6 ± 0.9 injections (range, 1 to 5 injections) before ME recurrence. The time between symptom onset and ME recurrence averaged 11.1 ± 1.5 months (range, 9 to 13 months) and the time between the last bevacizumab injection and ME recurrence averaged 4.9 ± 1.6 months (range, 2 to 8 months).
The BCVA values at the time of BRVO diagnosis, following initial treatment but before ME recurrence, at the time of recurrence and 3 months after recurrence were 0.59 ± 0.21 (Snellen equivalent, 20 / 77), 0.29 ± 0.12 (20 / 38), 0.58 ± 0.26 (20 / 76), and 0.62 ± 0.29 (20 / 83), respectively (Fig. 1A); the CFT measurements at these same time points were 514.9 ± 135.6, 214.9 ± 23.9, 484.9 ± 124.1, and 493.7 ± 123.9 µm, respectively (Fig. 1B). Both BCVA and CFT were measured, on average, 2.2 ± 0.9 months (range, 1 to 4 months) before ME recurrence. Neither BCVA nor CFT changed from their ME recurrence values over the 3-month follow-up period (p = 0.106 and p = 0.687, respectively; paired t-test).
Only one eye without foveal capillary nonperfusion exhibited a decrease in CFT of more than 100 µm (Fig. 2A-2D). An improvement in BCVA of one line (0.1 logMAR) was also noted in this eye. In the remaining 18 eyes, CFT was either stable or increased (Fig. 2E-2H). None of the examined eyes showed a CFT decrease to within normal limits (≤250 µm) during the 3 months following ME recurrence. Additionally, BCVA deteriorated by one or more lines (≥0.1 logMAR) in four eyes and remained unchanged in the remaining 14 eyes.
In the treated group, the BCVA values at the time of BRVO diagnosis, before ME recurrence, at the time of ME recurrence and 3 months after ME recurrence were 0.51 ± 0.23 (20 / 64), 0.24 ± 0.12 (20 / 34), 0.51 ± 0.21 (20 / 64), and 0.40 ± 0.24 (20 / 50), respectively (Fig. 1A); the CFT measurements at these same time points were 499.5 ± 124.8, 207.9 ± 31.6, 456.3 ± 126.8, and 294.7 ± 104.4 µm, respectively (Fig. 1B). The BCVA and CFT were measured an average of 2.4 ± 0.9 months (range, 1 to 4 months) before ME recurrence. Eleven eyes showed a decrease in CFT by ≥100 µm, with eight of these eyes having a CFT within normal limits (≤250 µm) at 3 months. By 3 months after ME recurrence, BCVA values had significantly improved (p = 0.002, paired t-test) and CFT measurements had significantly decreased (p < 0.001, paired t-test) compared to values at the time of ME recurrence. A BCVA improvement by one or more lines (≥0.1 logMAR) was noted in 10 eyes and a BCVA deterioration of one or more lines was noted in only one eye. The remaining six eyes had stable BCVA values.
The age (p = 0.078, independent samples t-test), sex (p = 0.196, Fisher's exact test), proportion of eyes with foveal capillary nonperfusion (p = 0.709), mean number of bevacizumab injections before ME recurrence (p = 0.422), time between symptom onset and ME recurrence (p = 0.524), and time between the last bevacizumab injection and ME recurrence (p = 0.859) were not significantly different between the treated and untreated groups (Table 1). At the time of ME recurrence, BCVA and CFT values were not significantly different between the treated and untreated groups (p = 0.772 and p = 0.996, respectively; independent samples t-test with a Bonferroni's correction) (Fig. 1). However, BCVA and CFT values 3 months after ME recurrence were significantly different between the two groups (p = 0.034 and p < 0.001, respectively; independent samples t-test with a Bonferroni's correction) (Fig. 1).
In this study, recurrent ME secondary to BRVO did not spontaneously resolve during the 3-month follow-up period. Additionally, CFT spontaneously decreased by more than 100 µm and visual acuity spontaneously improved in only one of 19 untreated eyes. The BCVA and CFT results were significantly better in eyes treated with a single intravitreal bevacizumab injection than in eyes left to follow the natural course of recurrent ME. These results indicate an unfavorable natural course of recurrent ME, which is distinct from the initial episode of ME following BRVO. Therefore, our results suggest the need for prompt treatment of recurrent ME secondary to BRVO.
Previous OCT studies have demonstrated that ME secondary to BRVO spontaneously decreases in many cases without any treatment [456]. This phenomenon was particularly and markedly noted during the first several months following initial BRVO diagnosis [456]. Shroff et al. [5] showed a decrease in ME and a subsequent improvement in visual acuity in eyes without foveal capillary nonperfusion. In the present study, there was no BCVA improvement in the majority of eyes in the untreated group, regardless of the presence of foveal capillary nonperfusion.
There exist several possible explanations for an unfavorable short-term natural course of recurrent ME, which are discussed below. First, an up-regulation of VEGF receptors [16] can occur following intravitreal anti-VEGF therapy. Matsumoto et al. [16] found that rebound ME after an-ti-VEGF intravitreal injection was more pronounced than the initial ME before treatment. They hypothesized that inhibiting the VEGF pathway up-regulates VEGF receptors within the retina, inducing a more severe ME when recurrence occurs. Although this hypothesis is plausible, we do not believe this explains all of our observations. The mean time between the last bevacizumab injection and ME recurrence was longer than 5 months in untreated patients. Knowing that intraocular bevacizumab is hardly detectable 50 days after injection [22], the influence of an increased number of VEGF receptors likely did not play a significant role in our patients with delayed ME recurrence. Second, it is possible that intravitreal anti-VEGF negatively influences vascular perfusion. Although there is some controversy, previous studies have showed that anti-VEGF therapy promotes retinal nonperfusion in eyes with retinal vein occlusion [2324]. In eyes with BRVO, retinal perfusion is already compromised and further deterioration from previous intravitreal bevacizumab therapy may impede spontaneous resolution of recurrent ME. Last, it is possible that selection bias led to the enrollment of more susceptible eyes. Recurrent ME after intravitreal bevacizumab therapy only occurs in a limited percentage of eyes with BRVO [1525]. In the present study, only eyes with recurrent ME were included, some of which had experienced multiple episodes of ME recurrence. It is possible that our study cohort had a more compromised ocular perfusion than eyes without ME recurrence following BRVO. These eyes may have also had higher levels of ME-inducing agents, including VEGF [26], interlukin-6 [26], and interlukin-8 [27].
In terms of anatomical outcome, the natural course of recurrent ME is definitively worse than that of eyes treated with intravitreal bevacizumab injection. Three months after a single intravitreal bevacizumab injection, a decrease in CFT of more than 100 µm was observed in 11 of 17 eyes, with eight eyes reaching a normal retinal thickness. In contrast, only one of 19 untreated eyes showed a decrease in CFT of more than 100 µm 3 months following ME recurrence. Because our follow-up period was only 3 months, our data applies only to short-term outcomes, and long-term prognosis cannot be estimated from our results. However, it is likely that sustained ME in untreated eyes results in cumulative retinal damage, which would negatively influence long-term visual function. Therefore, our study strongly suggests that a spontaneous decrease in ME recurrence after intravitreal bevacizumab therapy is unusual and treatment should be promptly initiated to avoid further deterioration in retinal function. This finding may be useful when discussing prognosis and determining treatment plans with patients.
The main strength of the present study is that we first examined the natural course of untreated recurrent ME following BRVO. However, there are study limitations as well. This study was retrospective, had a small sample size, and had a relatively short follow-up period after ME recurrence. In this non-controlled retrospective study, the criteria to decide whether or not to retreat are not clear. Additionally, this study was not performed on a consecutive series of patients suggesting some influence of selection bias. Most patients who experienced ME recurrence were retreated, with only a small proportion of patients followed-up without treatment. The OCT evaluation was not routinely performed during every follow-up visit. Thus, it is possible that mild recurrence of ME, not detectable with ophthalmoscopy, may have been missed. There was also a considerable amount of variation in the time between the symptom onset and ME recurrence. Last, the time between the last clinical examination and ME recurrence was approximately 2 months. It is possible that visual function deteriorated during this period, which may have influenced the study results.
In conclusion, recurrent ME secondary to BRVO that was previously treated with intravitreal bevacizumab did not spontaneously resolve after 3 months. Functional and anatomical outcomes at this time were significantly worse in eyes that did not undergo further anti-VEGF treatment compared to eyes that received a single intravitreal bevacizumab injection. These results may suggest the potential benefit of prompt treatment for recurrent ME. Further controlled studies in a consecutive group of patients with longer follow-up periods are needed to more accurately reveal the natural course of recurrent ME following BRVO, as well as the influence of prompt treatment on long-term visual prognosis.
References
1. Rogers SL, McIntosh RL, Lim L, et al. Natural history of branch retinal vein occlusion: an evidence-based systematic review. Ophthalmology. 2010; 117:1094–1101.e5. PMID: 20430447.

2. Argon laser photocoagulation for macular edema in branch vein occlusion: the Branch Vein Occlusion Study Group. Am J Ophthalmol. 1984; 98:271–282. PMID: 6383055.
3. Hayreh SS. Ocular vascular occlusive disorders: natural history of visual outcome. Prog Retin Eye Res. 2014; 41:1–25. PMID: 24769221.

4. Ramezani A, Entezari M, Moradian S, et al. Intravitreal triamcinolone for acute branch retinal vein occlusion: a randomized clinical trial. J Ophthalmic Vis Res. 2011; 6:101–108. PMID: 22454718.
5. Shroff D, Mehta DK, Arora R, et al. Natural history of macular status in recent-onset branch retinal vein occlusion: an optical coherence tomography study. Int Ophthalmol. 2008; 28:261–268. PMID: 17668149.

6. Moradian S, Faghihi H, Sadeghi B, et al. Intravitreal bevacizumab vs. sham treatment in acute branch retinal vein occlusion with macular edema: results at 3 months (report 1). Graefes Arch Clin Exp Ophthalmol. 2011; 249:193–200. PMID: 21337043.
7. Kwon SI, Kim YW, Bang YW, et al. Comparison of natural course, intravitreal triamcinolone, and intravitreal bevacizumab for treatment of macular edema secondary to branch retinal vein occlusion. J Ocul Pharmacol Ther. 2013; 29:5–9. PMID: 23237542.

8. Tan MH, McAllister IL, Gillies ME, et al. Randomized controlled trial of intravitreal ranibizumab versus standard grid laser for macular edema following branch retinal vein occlusion. Am J Ophthalmol. 2014; 157:237–247. PMID: 24112635.

9. Brown DM, Campochiaro PA, Bhisitkul RB, et al. Sustained benefits from ranibizumab for macular edema following branch retinal vein occlusion: 12-month outcomes of a phase III study. Ophthalmology. 2011; 118:1594–1602. PMID: 21684606.

10. Gunduz K, Bakri SJ. Intravitreal bevacizumab for macular oedema secondary to branch retinal vein occlusion. Eye (Lond). 2008; 22:1168–1171. PMID: 18344969.
11. Hanada N, Iijima H, Sakurada Y, Imasawa M. Recurrence of macular edema associated with branch retinal vein occlusion after intravitreal bevacizumab. Jpn J Ophthalmol. 2012; 56:165–174. PMID: 22183139.

12. Zhu D, Jin ZY, Tao Y, Jonas JB. Meta-analysis of the effect of intravitreal bevacizumab in branch retinal vein occlusion. J Ocul Pharmacol Ther. 2013; 29:523–529. PMID: 23537150.

13. Kim JY, Park SP. Comparison between intravitreal bevacizumab and triamcinolone for macular edema secondary to branch retinal vein occlusion. Korean J Ophthalmol. 2009; 23:259–265. PMID: 20046685.

14. Lee K, Jung H, Sohn J. Comparison of injection of intravitreal drugs with standard care in macular edema secondary to branch retinal vein occlusion. Korean J Ophthalmol. 2014; 28:19–25. PMID: 24505197.

15. Yasuda S, Kondo M, Kachi S, et al. Rebound of macular edema after intravitreal bevacizumab therapy in eyes with macular edema secondary to branch retinal vein occlusion. Retina. 2011; 31:1075–1082. PMID: 21478810.

16. Matsumoto Y, Freund KB, Peiretti E, et al. Rebound macular edema following bevacizumab (Avastin) therapy for retinal venous occlusive disease. Retina. 2007; 27:426–431. PMID: 17420693.

17. Jaissle GB, Szurman P, Feltgen N, et al. Predictive factors for functional improvement after intravitreal bevacizumab therapy for macular edema due to branch retinal vein occlusion. Graefes Arch Clin Exp Ophthalmol. 2011; 249:183–192. PMID: 21337042.

18. Yunoki T, Miyakoshi A, Nakamura T, et al. Treatment of macular edema due to branch retinal vein occlusion with single or multiple intravitreal injections of bevacizumab. Jpn J Ophthalmol. 2012; 56:159–164. PMID: 22246387.

19. Yunoki T, Mitarai K, Yanagisawa S, et al. Effects of vitrectomy on recurrent macular edema due to branch retinal vein occlusion after intravitreal injection of bevacizumab. J Ophthalmol. 2013; 2013:415974. PMID: 23533707.

20. Ogino K, Tsujikawa A, Murakami T, et al. Grid photocoagulation combined with intravitreal bevacizumab for recurrent macular edema associated with retinal vein occlusion. Clin Ophthalmol. 2011; 5:1031–1036. PMID: 21845030.
21. Chen SD, Sundaram V, Lochhead J, Patel CK. Intravitreal triamcinolone for the treatment of ischemic macular edema associated with branch retinal vein occlusion. Am J Ophthalmol. 2006; 141:876–883. PMID: 16527226.

22. Krohne TU, Eter N, Holz FG, Meyer CH. Intraocular pharmacokinetics of bevacizumab after a single intravitreal injection in humans. Am J Ophthalmol. 2008; 146:508–512. PMID: 18635152.

23. Campochiaro PA, Bhisitkul RB, Shapiro H, Rubio RG. Vascular endothelial growth factor promotes progressive retinal nonperfusion in patients with retinal vein occlusion. Ophthalmology. 2013; 120:795–802. PMID: 23260261.

24. Terui T, Kondo M, Sugita T, et al. Changes in areas of capillary nonperfusion after intravitreal injection of bevacizumab in eyes with branch retinal vein occlusion. Retina. 2011; 31:1068–1074. PMID: 21451440.

25. Karagiannis DA, Karampelas MD, Soumplis VM, et al. Recurrence of macular edema in retinal vein occlusions after treatment with intravitreal ranibizumab (Lucentis). Can J Ophthalmol. 2011; 46:486–490. PMID: 22153634.

26. Noma H, Funatsu H, Yamasaki M, et al. Pathogenesis of macular edema with branch retinal vein occlusion and intraocular levels of vascular endothelial growth factor and interleukin-6. Am J Ophthalmol. 2005; 140:256–261. PMID: 16086947.

27. Fonollosa A, Garcia-Arumi J, Santos E, et al. Vitreous levels of interleukine-8 and monocyte chemoattractant protein-1 in macular oedema with branch retinal vein occlusion. Eye (Lond). 2010; 24:1284–1290. PMID: 20111061.

Fig. 1
Changes in the logarithm of the minimum angle of resolution (logMAR), best-corrected visual acuity (BCVA, A) and central foveal thickness (CFT, B) in eyes with recurrent macular edema (ME) following branch retinal vein occlusion (BRVO). The solid line (closed circles) indicates data from eyes without treatment (untreated group, 19 eyes) and the dashed line (closed squares) indicates data from eyes treated with a single intravitreal bevacizumab (IVB) injection (treated group, 17 eyes). Differences in BCVA and CFT between the two groups were not statistically significant at the time of ME recurrence (p = 0.772 and p = 0.996, respectively). However, 3 months after ME recurrence, the treated group had significantly improved results compared to those of the untreated group for both BCVA (p = 0.034) and CFT (p < 0.001). “At diagnosis” indicates the time when BRVO with ME was first diagnosed. “After IVB” indicates the period before ME recurrence. “Recurrence” indicates the time at which ME recurrence was noted. “3 mon” indicates 3 months after ME recurrence. Statistical analyses were performed using repeated measures analysis of variances with a Bonferroni's correction and independent sample t-test. An asterisk indicates a statistically significant difference between the two groups.
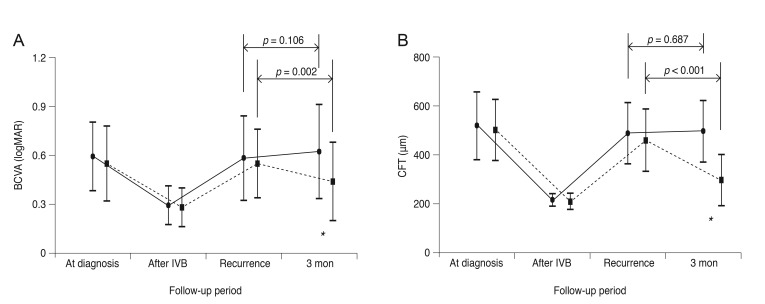
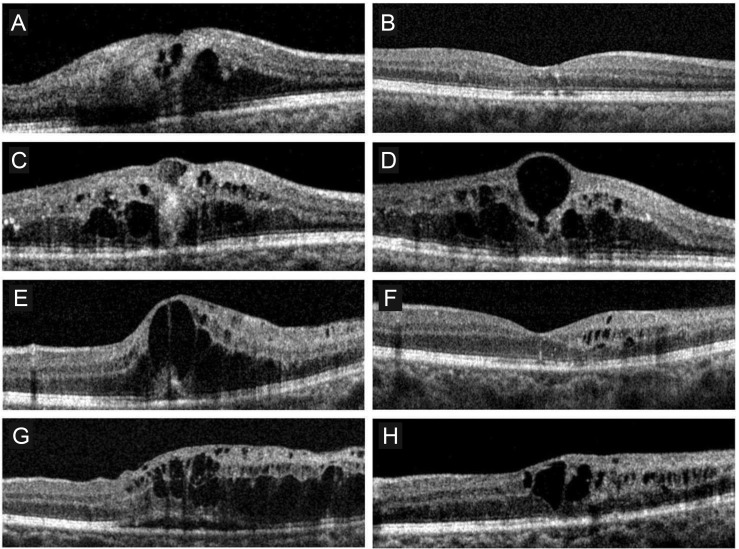
Fig. 2
Optical coherence tomography findings in two eyes diagnosed with macular edema (ME) secondary to branch retinal vein occlusion. Both eyes received no therapy for recurred ME for 3 months. Case 1 (A-D): at diagnosis (A), ME resolved after 2 monthly intravitreal bevacizumab injection (B), ME recurred 9 months after the last treatment (C). Retinal thickness remained elevated and relatively unchanged during the 3-month follow-up period (D). Case 2 (E-H): at diagnosis (E), ME resolved after 2 monthly intravitreal bevacizumab injections (F), the ME recurred 7 months after the last treatment (G). Although ME slightly decreased during the 3-month follow-up period, macular thickness did not return to normal (H).




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download