Abstract
Purpose
To evaluate the efficacy of intravitreal aflibercept monotherapy in submacular hemorrhage (SMH) secondary to wet age-related macular degeneration (AMD).
Methods
This study included 25 eyes in 25 patients with SMH involving the fovea secondary to wet-AMD. All patients were treated with three consecutive monthly intravitreal aflibercept (2.0 mg/0.05 mL) injections, followed by as-needed reinjection. They were followed for at least 6 months. Best-corrected visual acuity (BCVA), central foveal thickness (CFT), and area of SMH were measured at diagnosis, as well as at 3 and 6 months after treatment initiation.
Results
The BCVA significantly improved from 0.79 ± 0.41 logarithm of the minimum angle of resolution (logMAR) at baseline to 0.54 ± 0.41 logMAR at 6 months (p < 0.001). BCVA ≥3 lines and stable vision were observed in 96% of the eyes. The CFT significantly decreased from 560.8 ± 215.3 µm at baseline to 299.8 ± 160.2 µm at 6 months (p < 0.001). The area of SMH significantly decreased from 10.5 ± 7.1 mm2 at baseline to 1.8 ± 6.5 mm2 at 6 months (p < 0.001). The BCVA, CFT, and area of SMH at baseline, as well as duration of symptoms, all correlated with BCVA at the 6-month follow-up.
The wet age-related macular degeneration (AMD) that is accompanied by submacular hemorrhage (SMH) may cause sudden visual loss. The natural history of the condition is characterized by poor visual prognosis, with an expected visual acuity loss of three lines within 3 years and a final visual acuity <20 / 200 in 53% of cases [12]. SMH can seriously damage the retinal tissue through several potential mechanisms [3].
A variety of approaches, including surgical submacular clot evacuation, photodynamic therapy, and pneumatic displacement with or without tissue plasminogen activator, have been evaluated with regard to efficacy in promoting visual recovery [45]. However, these strategies have generally been limited or produced serious complications.
Intravitreal anti-vascular endothelial growth factor (VEGF) therapy is currently the most commonly adopted treatment for wet-AMD. Large-scale clinical trials have demonstrated the efficacy of ranibizumab in preserving and improving the final visual acuity in patients with wet-AMD [6]. However, choroidal neovascularization (CNV) with hemorrhage involving an area >50% of the entire CNV lesion was specifically excluded in the above trials. Some previous studies have reported improvements in the visual acuity of eyes with SMH after bevacizumab or ranibizumab monotherapy [789].
Aflibercept (Eylea; Bayer HealthCare, Berlin, Germany) is an anti-VEGF drug with a high affinity for VEGF, a long half-life, and the ability to inhibit placental growth factor [10]. The efficacy of aflibercept in the treatment of wet-AMD has been demonstrated [1112]. However, to the best of our knowledge, the efficacy of intravitreal aflibercept (IVA) injection in SMH secondary to wet-AMD has not been evaluated.
The purpose of this study was to evaluate the 6-month efficacy of IVA in SMH secondary to wet-AMD. We also analyzed predictive factors in visual acuity outcomes.
This retrospective, observational case series was performed at a single center according to the tenets of the Declaration of Helsinki. This study was approved by the institutional review board of Kim's Eye Hospital, Konyang University College of Medicine.
Medical records were reviewed for all patients presenting to our hospital between June 2014 and December 2014, who were diagnosed with wet-AMD and SMH comprising greater than 1 disc diameter of the fovea and >50% of the lesion, and who received IVA therapy.
The inclusion criteria were: (1) age >50 years, (2) newly diagnosed wet-AMD and SMH, (3) duration of symptoms <3 months, (4) >6 months of follow-up, and (5) treatment-naïve eyes. The diagnosis of active CNV was confirmed by fluorescein angiography (FAG) and indocyanine green angiography (ICGA). Patients who had previously received treatment or who had evidence of end-stage AMD were excluded from this study. Furthermore, patients with ocular diseases that could potentially influence visual acuity—such as glaucoma, macular hole, diabetic retinopathy, retinal vascular occlusion, rhegmatogenous retinal detachment, or history of intraocular surgery (except cataract surgery)—were also excluded.
All subjects had undergone comprehensive ophthalmologic examinations, including measurements of best-corrected visual acuity (BCVA), 90-diopter lens slit-lamp biomicroscopy, FAG, ICGA (HRA-2; Heidelberg Engineering, Heidelberg, Germany), and spectral domain OCT (Spectralis, Heidelberg Engineering). The results of the FAG and the ICGA were analyzed by two independent examiners (KHS a nd JHK). Cases of wet-AMD were classified a s typical exudative AMD or polypoidal choroidal vasculopathy (PCV) on the basis of FAG and ICGA findings. Cases exhibiting branching vascular networks or terminating polypoidal lesions were diagnosed with PCV. The other cases were classified as typical exudative AMD. In cases of thick SMH interfering with identification of underlying pathologic features, the diagnosis was based on the results of the follow-up examinations, which included ICGA.
The main outcome measures were BCVA, duration of symptoms, area of SMH, and central foveal thickness (CFT). At each follow-up visit, measurement of BCVA, binocular fundus examination, and optical coherence tomography (OCT) were performed. Snellen BCVA values were converted to the logarithm of the minimum angle of resolution (logMAR) values for analysis. The area of SMH was manually outlined with a computer mouse using the option provided via the HRA-2 built-in software. The results were expressed in square millimeters. CFT was defined as the distance between the internal limiting membrane and Bruch's membrane at the fovea. It was manually measured using the calipers provided by an OCT software program. In this study, we considered the possibility that SMH exceeding 30 mm2 in area and CFT exceeding 1,500 µm could be inaccurate. Thus, 30 mm2 of hemorrhage area and 1,500 µm of CFT were considered threshold values. Lesions exceeding these thresholds were considered 30 mm2 in area or 1,500 µm in depth. The area of the SMH and the CFT were estimated by a single examiner (KHS).
All patients received three consecutive monthly intravitreal injections of aflibercept (2.0 mg/0.05 mL) through the pars plana via a 30-gauge needle. During the maintenance phase, aflibercept was administered to all patients according to the same treatment and retreatment protocols if any of the following were observed: visual deterioration of more than 0.2 Snellen BCVA, evidence of persistent fluid or hemorrhage involving the macula on OCT at least 1 month after the previous injection, newly developed hemorrhage or recurrence of fluid involving the macula, and evidence of an active CNV lesion found on FAG, ICGA, or OCT. The minimum interval between two intravitreal injections during the maintenance period was 8 weeks.
Statistical analyses were performed with a commercially available software package (SPSS ver. 12.0; SPSS Inc., Chicago, IL, USA). Changes in visual acuity, CFT, and area of the SMH compared to baseline values were assessed by paired Student's t-test. Pearson correlation analysis was performed to verify any associations between variables. For the non-parametric data, the Kruskal-Wallis test was used. A p-value less than 0.05 was considered significant.
Of the 35 patient records examined, following the inclusion criteria outlined above, 25 eyes belonging to 25 patients were finally enrolled. Ten eyes were excluded for the following reasons: less than 6 months of follow-up (five eyes), fewer than three IVA injections (three eyes), and vitrectomy during the follow-up period due to the development of severe vitreous hemorrhage (two eyes).
Ultimately, 25 eyes belonging to 25 patients were included in the statistical analysis. Among them, 14 eyes (56%) and seven eyes (28%) were ultimately diagnosed with typical exudative AMD and PCV, respectively. A definite diagnosis was not possible in the remaining four cases (16%) because thick SMH precluded reliable ICGA imaging of the lesion. The BCVA, CFT, and area of the SMH were 0.79 ± 0.41 logMAR, 560.8 ± 215.3 µm, and 10.5 ± 7.1 mm2, respectively. The patients were treated with 3.6 ± 0.8 (range, 3 to 5) IVA injections during the 6-month follow-up period (Table 1).
The BCVA at baseline and at 3 and 6 months after treatment initiation was 0.79 ± 0.41, 0.61 ± 0.46, and 0.54 ± 0.41 logMAR, respectively. The BCVA at diagnosis was significantly different from that measured at 3 or 6 months after treatment initiation (p = 0.007, p < 0.001, respectively). The BCVA at 3 months after treatment initiation was significantly different from that measured at 6 months (p = 0.046) (Fig. 1A). Eleven of 25 eyes (44%) demonstrated improvements of 0.3 or more in logMAR visual acuity. One eye (4%) had a decrease of 0.3 or more in logMAR visual acuity, and 13 eyes (52%) had change less than 0.3 in the logMAR visual acuity at 6 months after treatment initiation (Table 2). Baseline BCVA was significantly associated with change in BCVA at the 6-month follow-up (p = 0.022). There was no association between change in BCVA at the 6-month follow-up and age, duration of symptoms, area of the SMH, CFT at baseline, or number of injections (Table 3). However, BCVA, duration of symptoms, area of the SMH, and CFT at baseline all correlated with BCVA at the 6-month follow-up. The Pearson correlation coefficients were 0.671 (p < 0.001), 0.512 (p = 0.044), 0.552 (p = 0.004), and 0.562 (p = 0.003), respectively. The number of IVA injections was not significantly associated with BCVA at 6 months (p = 0.931).
The mean CFT at baseline and at 3 and 6 months after treatment initiation was 560.8 ± 215.3, 313.1 ± 189.3, and 299.8 ± 160.2 µm, respectively. The CFT at diagnosis was significantly different from that measured at 3 or 6 months after treatment initiation (p < 0.001 and p < 0.001, respectively). However, the CFT at 3 months after treatment initiation was not significantly different from that measured at 6 months (p = 0.583) (Fig. 1B).
The area of the SMH at baseline and at 3 and 6 months after treatment initiation was 10.5 ± 7.1, 3.9 ± 8.6, and 1.8 ± 6.5 mm2, respectively. The area of the SMH at diagnosis was significantly different from that measured at 3 or 6 months after treatment initiation (p < 0.001 and p < 0.001, respectively). The area of the SMH at 3 months after treatment initiation was significantly different from that measured at 6 months (p = 0.028) (Fig. 1C). Complete resolution of SMH within 3 and 6 months was noted in 13 (52%) and 22 (88%) of these patients, respectively (Fig. 2A-2I).
Three of the 25 eyes (12%) showed a decrease of 0.1 or more in logMAR visual acuity. Two eyes developed new SMH at 3 months after the initial IVA injection (at 1 month after the three loading injections). One of the 25 eyes (4%) (a typical exudative AMD type) developed an RPE tear at 3 months after treatment initiation. No significant adverse systemic effects were registered over the course of follow-up.
Retrospective studies of the natural history indicate that the final visual outcome in SMH secondary to wet-AMD is frequently poor [1]. In our study, visual acuity improved significantly after IVA monotherapy in eyes with SMH secondary to wet-AMD. The mean visual acuity improved from 0.79 logMAR at baseline to 0.54 logMAR at the 6-month follow-up. As a result, an improvement in BCVA ≥3 lines or stable vision was observed in 44% and 52% of eyes, respectively. To our knowledge, this is the first case series to evaluate the efficacy of IVA in the treatment of wet-AMD accompanied by SMH.
Previous studies have suggested that ranibizumab or bevacizumab is beneficial for treatment of SMH secondary to wet-AMD. Shienbaum et al. [8] reported a retrospective analysis in patients receiving bevacizumab or ranibizumab. This study reported improvement in logMAR visual acuity of 0.3 or more in nine of 15 eyes (60.0%) and of less than 0.3 in six of 15 eyes (40.0%). Kim et al. [13] reported improvement of 0.3 or more in logMAR visual acuity in 54 of 91 eyes (59.3%) and of less than 0.3 in 29 (31.9%) eyes. Altaweel et al. [9] most recently reported a prospective analysis in patients receiving bevacizumab or ranibizumab. This study reported improvement of 0.3 or more in logMAR visual acuity (15 or more letters) in 25 of 78 eyes (32.1%) and of less than 0.3 (15 or more letters) in 47 eyes (60.3%). These studies showed that the combination of BCVA ≥3 lines and stable vision was observed in 100%, 91.2%, and 92.3% of cases, respectively (Table 4).
Our study showed improvement of 0.3 or more in logMAR visual acuity in 11 of 25 eyes (44.0%) and of less than 0.3 in 13 eyes (52.0%). BCVA ≥3 lines and stable vision were observed in 96% of cases. Therefore, IVA therapy can produce the same results as intravitreal ranibizumab or bevacizumab with regard to improving visual acuity in the treatment of SMH secondary to wet-AMD.
Meanwhile, some studies have suggested that aflibercept is beneficial for the treatment of PCV. Inoue et al. [14] reported a prospective analysis in patients receiving aflibercept for PCV. That study reported improvements of 0.3 or more in logMAR visual acuity and of less than 0.3 in the logMAR visual acuity at 6 months after treatment in 3 (18.8%) and 13 (81.2%) of 16 eyes, respectively. Yamamoto et al. [15] reported a retrospective analysis in patients receiving aflibercept for PCV. Their study reported improvements of 0.3 or more in logMAR visual acuity and of less than 0.3 at 12 months after an initial injection in 22 (24.4%) and 64 (71.1%) of 90 eyes, respectively. Therefore, IVA appears to have a similar efficacy in improving visual acuity in both PCV and SMH secondary to wet-AMD.
However, the obvious differences in baseline features between the present study and the previous studies limit direct comparison of these results.
A previous study showed that predictive factors of final visual acuity include duration of symptoms, baseline BCVA, CFT, area of the SMH, and time to resolution of bleeding [1316]. Our study also confirmed that predictive factors include baseline BCVA, duration of symptoms, area of the SMH, and baseline CFT. Therefore, it has been shown that visual prognosis is generally poor in cases in which the treatment is delayed and when there is a large amount of hemorrhage. However, in this study, change in BCVA during the 6-month follow-up period was associated only with baseline BCVA. Presumably, any amount of hemorrhage on the fovea limited BCVA may not only be estimated by the CFT or the area of the SMH; that is, density of hemorrhage can be associated with amount of hemorrhage. This may lead to a significant difference in studies regarding the predictive factors associated with final visual acuity, as well as changes in visual acuity. Improving this is beyond the scope of the present study.
Shienbaum et al. [8] reported that central lesion thickness decreased from 755 µm at baseline to 349 µm at the 6-month follow-up, and the degree of change was approximately 406 µm. Altaweel et al. [9] reported that the mean total foveal thickness decreased by 199 µm during the first year. Kim et al. [13] reported that the mean CFT at baseline and at 6 months was 596.8 and 282.9 µm, respectively, and the degree of change was approximately 313.9 µm. Our study reported that the mean CFT at baseline and at 6 months was 560.8 ± 215.3 and 299.8 ± 160.2 µm, respectively, and the degree of change was approximately 261 µm. The degree of change in CFT was similar to that reported in previous studies.
Kim et al. [13] reported complete resolution of SMH in 33 of 73 eyes (45.2%) and 49 of 73 eyes (67.1%) at 3 and 6 months, respectively. Iacono et al. [16] reported complete resolution of SMH in 18 of 23 eyes (78.2%) at 6 months. In our study, the area of the SMH was completely resolved in 13 of 25 eyes (52%) and 22 of 25 eyes (83%) at 3 months and 6 months, respectively. Despite the absence of a control group, there is evidence of rapid regression of SMH after IVA therapy, even without use of gas for hemorrhage displacement.
Scupola et al. [2] reported bleeding recurrence in 4% of cases, and vitreous hemorrhage in 6.6% of cases over a natural course of wet-AMD. Kim et al. [13] reported vitreous hemorrhage in 12.0% of cases. Cho et al. [17] reported bleeding recurrence in 11.1% of cases and vitreous hemorrhage in 11.1% of cases of PCV treated with bevacizumab or ranibizumab. Our study reported similar results for bleeding recurrence and vitreous hemorrhage (7% for both). Thus, although the difference was not large, the frequencies of bleeding recurrence and vitreous hemorrhage were similar compared to those of other drugs.
The main limitations of this study were its small sample size, the lack of a control group without IVA, and the relatively short follow-up period. Further investigations are needed to assess the long-term safety and efficacy of IVA administration. Furthermore, a randomized clinical trial comparing intravitreal bevacizumab, ranibizumab, and IVA for SMH should be planned to identify the optimal treatment. Subtypes of AMD, including PCV and retinal angiomatous proliferation, are not differentiated in the analysis for main outcome measures.
In conclusion, although evaluated after only a short follow-up period, IVA is well-tolerated as treatment in maintaining or improving vision in patients with SMH secondary to wet-AMD. There are associations between BCVA at 6 months and baseline BCVA, duration of symptoms, area of the SMH, and baseline CFT. Further long-term studies are warranted in order to establish the best treatment algorithm for managing SMH secondary to wet-AMD.
Figures and Tables
 | Fig. 1(A) Changes in best-corrected visual acuity (BCVA) during follow-up after intravitreal aflibercept injection. BCVA improved at 3 months from baseline. The mean BCVA improved from 0.79 ± 0.41 to 0.61 ± 0.46 logarithm of the minimum angle of resolution (logMAR) (p = 0.007), and this overall improvement continued throughout the 3-month follow-up. (B) Changes in central foveal thickness (CFT) with optical coherence tomography during follow-up after intravitreal aflibercept injection. The CFT improved at 3 months from baseline. The mean CFT decreased from 560.8 ± 215.3 to 313.1 ± 189.3 µm (p < 0.001), and this overall decrease continued throughout the 3-month follow-up. (C) Changes in area of the submacular hemorrhage during follow-up after intravitreal aflibercept injection. Area of the submacular hemorrhage decreased at 3 months from baseline. The mean area decreased from 10.5 to 3.9 mm2 (p < 0.001), and this overall decrease continued throughout the 3-month follow-up. |
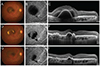 | Fig. 2At the time of diagnosis, Best-corrected visual acuity (BCVA) and central foveal thickness (CFT) were measured as 1.0 logarithm of the minimum angle of resolution (logMAR) and 493 µm, respectively (A-C). After 3 monthly aflibercept injections, the submacular hemorrhage partially resolved, and BCVA and CFT were 0.52 logMAR and 148 µm, respectively (D-F). At 6 months after the initial aflibercept injection, the submacular hemorrhage completely resolved, and BCVA and CFT improved to 0.42 logMAR and 134 µm, respectively (G-I). |
Table 1
Characteristics of patients with wet AMD with submacular hemorrhage as an initial presentation

Table 3
Frequency distribution of changes in visual acuity at 6 months

Values are presented as mean ± standard deviation.
BCVA = best-corrected visual acuity; SMH = submacular hemorrhage; logMAR = logarithm of the minimum angle of resolution; CFT = central foveal thickness.
*n = 11 (44%); †n = 13 (52%); ‡n = 1 (4%); §Statistical analysis was performed using the Kruskal-Wallis test.
References
1. Avery RL, Fekrat S, Hawkins BS, Bressler NM. Natural history of subfoveal subretinal hemorrhage in age-related macular degeneration. Retina. 1996; 16:183–189.
2. Scupola A, Coscas G, Soubrane G, Balestrazzi E. Natural history of macular subretinal hemorrhage in age-related macular degeneration. Ophthalmologica. 1999; 213:97–102.
3. Toth CA, Morse LS, Hjelmeland LM, Landers MB 3rd. Fibrin directs early retinal damage after experimental subretinal hemorrhage. Arch Ophthalmol. 1991; 109:723–729.
4. Bakri SJ, Nickel J, Yoganathan P, Beer PM. Photodynamic therapy for choroidal neovascularization associated with submacular hemorrhage in age-related macular degeneration. Ophthalmic Surg Lasers Imaging. 2006; 37:278–283.
5. Hassan AS, Johnson MW, Schneiderman TE, et al. Management of submacular hemorrhage with intravitreous tissue plasminogen activator injection and pneumatic displacement. Ophthalmology. 1999; 106:1900–1906.
6. Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006; 355:1419–1431.
7. Stifter E, Michels S, Prager F, et al. Intravitreal bevacizumab therapy for neovascular age-related macular degeneration with large submacular hemorrhage. Am J Ophthalmol. 2007; 144:886–892.
8. Shienbaum G, Garcia Filho CA, Flynn HW Jr, et al. Management of submacular hemorrhage secondary to neovascular age-related macular degeneration with anti-vascular endothelial growth factor monotherapy. Am J Ophthalmol. 2013; 155:1009–1013.
9. Altaweel MM, Daniel E, Martin DF, et al. Outcomes of eyes with lesions composed of >50% blood in the comparison of Age-related Macular Degeneration Treatments Trials (CATT). Ophthalmology. 2015; 122:391–398.e5.
10. Stewart MW, Grippon S, Kirkpatrick P. Aflibercept. Nat Rev Drug Discov. 2012; 11:269–270.
11. Heier JS, Brown DM, Chong V, et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology. 2012; 119:2537–2548.
12. Cho H, Shah CP, Weber M, Heier JS. Aflibercept for exudative AMD with persistent fluid on ranibizumab and/or bevacizumab. Br J Ophthalmol. 2013; 97:1032–1035.
13. Kim JH, Chang YS, Kim JW, et al. Intravitreal anti-vascular endothelial growth factor for submacular hemorrhage from choroidal neovascularization. Ophthalmology. 2014; 121:926–935.
14. Inoue M, Arakawa A, Yamane S, Kadonosono K. Short-term efficacy of intravitreal aflibercept in treatment-naive patients with polypoidal choroidal vasculopathy. Retina. 2014; 34:2178–2184.
15. Yamamoto A, Okada AA, Kano M, et al. One-year results of intravitreal aflibercept for polypoidal choroidal vasculopathy. Ophthalmology. 2015; 122:1866–1872.
16. Iacono P, Parodi MB, Introini U, et al. Intravitreal ranibizumab for choroidal neovascularization with large submacular hemorrhage in age-related macular degeneration. Retina. 2014; 34:281–287.
17. Cho HJ, Koh KM, Kim HS, et al. Anti-vascular endothelial growth factor monotherapy in the treatment of submacular hemorrhage secondary to polypoidal choroidal vasculopathy. Am J Ophthalmol. 2013; 156:524–531.e1.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download