Abstract
Purpose
Chronic use of topical hypotensive agents induces several side effects caused by preservatives. The purpose of this study was to evaluate the effects of prostaglandin analogs with varying concentrations of benzalkonium chloride (BAC), preservative-free (PF), and alternative preservatives on mouse corneal tissue.
Methods
Thirty-five, 8- to 10-week-old female C57BL/6 mice (five mice for each group) were used for this study. To the control group, we applied normal saline, and to each drug-treated group we applied 0.02% BAC, bimatoprost 0.01% (with BAC 0.02%), latanoprost 0.005% (with BAC 0.02%), travoprost 0.004% (with 0.001% polyquad) or tafluprost 0.0015% with/without 0.001% BAC, once a day (9 p.m.) for 4 weeks. Corneal fluorescein staining was evaluated in all groups. After harvest, the corneal tissues were embedded in paraffin and then Hematoxylin-Eosin stain was performed for histopathological examination. Immunofluorescence staining was done against TNF-α, IL-6, HLA DR, pJNK, and pAkt.
Results
In corneal fluorescein staining, severe punctate epithelial keratitis was seen in the groups of 0.02% BAC, 0.02% BAC containing bimatoprost 0.01% and latanoprost 0.005%. The surface desquamation, irregular surface, loss of cell borders, anisocytosis and stromal shrinkage were observed in the groups of BAC-containing eye drops. Moreover, the groups treated with BAC-containing eye drops have high inflammatory markers, significantly decreased cell viability-related signal, pAkt, and higher apoptosis-inducing signal, pJNK, than the control group. On the other hand, travoprost 0.004% and PF tafluprost 0.0015% have less cellular morphologic changes, lower inflammation, and higher cellular viability than BAC-containing formulations.
Glaucoma, as a progressive optic neuropathy, needs chronic medical therapy as long as intraocular pressure (IOP) is controlled. Topical medical treatment is usually the first choice in therapy. Moreover, prostaglandin analogs (PGs) are the first-line therapy for lowering IOP in patients with glaucoma as a monotherapy and a combination therapy with other hypotensive agents. PGs are superior to beta-adenoreceptor antagonists in terms of lowering IOP, and they have no severe systemic side effects during long-term clinical use [123]. Currently, five different PGs, isopropyl unoprostone, latanoprost, travoprost, bimatoprost, and tafluprost are used for the treatment of glaucoma. Chronic use of topical hypotensive agents induces side effects, such as ocular inflammation, allergy, dry eye syndrome and failure of filtration surgery [456], which can be caused by components such as preservatives. With increasing attention on the ocular surface effect of preservatives, new preservative-free (PF) PGs and other types of preservatives-containing PGs have been developed. They have consistently demonstrated good tolerance in vitro and in patients, who disclosed significantly fewer ocular symptoms and signs of irritation, such as pain, discomfort, or dry eye sensation [78].
Benzalkonium chloride (BAC) is the most commonly used preservative in eye-drops. For glaucoma treatment, many patients need two or more different eye-drops. Instillation of several BAC-containing ophthalmic medications has potential safety concerns. Several studies showed that BAC is associated with decreased tear film stability, worsening of dry eyes, inflammatory cell infiltration, superficial punctate keratitis, blepharitis, and eyelid eczema [9101112].
The purpose of this study was to evaluate the effects of PGs with BAC, alternative preservatives, and no preservatives in a validated mouse model.
Thirty-five, 8- to 10 week-old female C57BL/6 mice (18 to 20 g, purchased from Hyochang Science, Daegu, Korea) were used for this study. These mice were kept in the facility with a standard environment throughout the study as follows: room temperature 25 ± 1℃, relative humidity 60 ± 10%, and alternating 12-hour light/dark cycles (8 a.m. to 8 p.m.). All experimental procedures were performed in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. Based on the clinical evaluations (described below), the mice were then randomly divided into seven groups (5 mice for each group): normal saline-treated control group, BAC-treated group and five anti-glaucomatic agent-treated groups.
Anti-TNF-α, anti-IL-6 and anti-HLA DR were purchased from Abcam (Cambridge, MA, USA). Anti-phosphorylated c-Jun NH(2)-terminal kinase (pJNK) and anti-phosphorylated-Akt (pAkt) were purchased from Cell Signaling Technology (Allschwil, Switzerland). 4',6-diamidino-2-phenylindole (DAPI) and Prolong Gold were purchased from Invitrogen (Carlsbad, CA, USA).
After grouping, the control group was administered 5 µL of normal saline topically and the BAC-treated group was processed with 5 µL of 0.02% BAC topically in each right eye. Others were t reated with 5 µL of one t he five different anti-glaucomatic agents: bimatoprost 0.01% (Lumigan; Allergan, Irvine, CA, USA), latanoprost 0.005% (Xalatan; Pfizer, New York, NY, USA), travoprost 0.004% (Travatan; Alcon, Fort Worth, TX, USA) and tafluprost 0.0015% with/without 0.001% BAC (Taflotan/Taflotan-s; Santen, Osaka, Japan). Each eye drop was applied once a day (9 p.m.) for 4 weeks. We did not treat the left eyes, but used them for control samples. During the treatment, the clinical evaluations were performed by a single masked ophthalmologist. On day 30, corneal fluorescein staining was evaluated and then all mice were euthanized and the eye tissues were carefully harvested for histological analysis following the methods described below.
One microliter of 0.5 % fluorescein solution was applied to the lower conjunctival sac of each right eye of mice with a micropipette. The stained status was evaluated by slit-lamp examination.
Corneal tissues were isolated from harvested eye tissues and fixed in 4% paraformaldehyde in phosphate buffered saline (PBS) for 24 hours at 4℃, and then washed twice in PBS. The samples were then dehydrated through an ethanol series, cleared by soaking in xylene, embedded in paraffin, and sectioned into 5 µm slices using a microtome RM 2125RT (Leica, Wetzlar, Germany). Slides containing paraffin sections were deparaffinized in xylene and rehydrated through an ethanol series, and then H&E staining was performed for histopathological examination. The specimens were mounted with Permount (Fisher, Fair Lawn, NJ, USA), and images were captured by microscope (Axio Vision 4; Carl Zeiss, Jena, Germany).
For immunofluorescence staining, the corneal tissue sections were deparaffinized in a 65℃ incubator for 1 hour in xylene and rehydrated through an ethanol series. The specimens were permeabilized with 0.3% Triton-X for 10 minutes, and blocked overnight with 5% normal goat serum and bovine serum albumin in Tris-buffered saline at 4℃. The specimens were then incubated overnight with monoclonal or polyclonal antibodies against TNF-α, IL-6, HLA DR, pJNK, and pAkt (diluted 1:50-100) in 5% bovine serum albumin, and washed three times in Tris-buffered saline. The specimens were further incubated with the appropriate Alexa 488-or 555-conjugated secondary antibodies at room temperature for 1 hour, and nuclei were counterstained with DAPI (1 µg/mL). The slides were washed once with PBS and mounted, and images were captured using a Zeiss fluorescence microscope (Axio Vision 4, Carl Zeiss). Density of green-or red-fluorescence was measured in color histogram by using ImageJ software program.
Severe punctate epithelial keratitis was seen in the groups of 0.02% BAC, 0.02% BAC-containing bimatoprost 0.01% and latanoprost 0.005% compared with control group (Fig. 1).
We investigated whether treatment with different formulations affected normal mouse cornea. Morphological changes were examined in sectioned corneal tissue treated with normal saline, 0.02% BAC, bimatoprost 0.01% (with BAC 0.02%), latanoprost 0.005% (with BAC 0.02%), travoprost 0.004% (with 0.001% polyquad), tafluprost 0.0015% (with 0.001% BAC) and PF tafluprost 0.0015% by H&E stain (Fig. 2). The surface desquamation, irregular surface, loss of cell borders, anisocytosis and stromal shrinkage were observed in the groups of 0.02% BAC and BAC-containing eye drops, while the travoprost 0.004% (with 0.001% polyquad) and PF tafluprost 0.0015% groups had morphology similar to the control group.
We examined these markers in corneal tissue by immunofluorescence staining to identify the inflammatory effect of several eye drop formulations. Immunostaining of TNF-α-positive inflammatory cells in the cornea is shown and the density of fluorescence is presented in Fig. 3. The IL-6-positive and HLA DR-positive cells and the density of fluorescence are also presented in Figs. 4 and 5. As seen under histological examination, immunofluorescence-stained inflammatory markers are highly expressed in BAC-containing eye drop-treated groups. In contrast, travoprost 0.004% (with 0.001% polyquad) and PF tafluprost 0.0015% groups had only a few inflammatory marker-positive cells present.
The pJNK expression was evaluated in this study by immunofluorescence staining (Fig. 6). 0.02% BAC-containing formulations show significantly higher expression of pJNK than the control group (p < 0.03), while 0.001% BAC-containing tafluprost 0.0015%, PF tafluprost 0.0015%, 0.001% polyquad-containing travoprost 0.004% had significantly lower expression of pJNK-positive cells than 0.02% BAC (p < 0.05).
We investigated the pAkt expression in corneal tissue to evaluate the cell viability influenced by preservatives (Fig. 7). Immunofluorescence staining of pAkt showed significantly lower expression in 0.02% BAC and latanoprost 0.005% (BAC 0.02%) groups than the control group (p < 0.05). The PF tafluprost 0.0015% group had significantly higher expression than the control group (p < 0.05). Moreover, the PF tafluprost 0.0015% group had significantly higher expression than the 0.02% BAC and preserved tafluprost 0.0015% group (p < 0.05).
Eye drops need preservatives for antimicrobial action and drug absorption; BAC is the most commonly used preservative for antimicrobial action. BAC, a quaternary ammonium compound, is a highly effective antimicrobial agent that acts by denaturing proteins and disrupting cytoplasmic membranes [13]. In the eyes, BAC turnover is very slow and is retained in ocular tissues up to 48 hours after administering a single drop [14]. Several studies revealed ocular surface toxicity resulting from BAC present in ophthalmic agents [4568151617181920212223]. Our previous in vitro study [24] reported about the toxicity of BAC-preserved, polyquad-preserved and PF PGs on primary cultured human conjunctival fibroblast cells. The morphological changes were observed in cells treated with BAC in a dose-dependent manner by phase-contrast microscopy. Severe cell shrinkage was observed in the bimatoprost 0.01% (with BAC 0.02%) and latanoprost 0.005% (with BAC 0.02%) groups. In the tafluprost 0.0015% group (with BAC 0.001%), mild cell shrinkage was visible. In contrast, the travoprost 0.004% (with 0.001% polyquad) and PF tafluprost 0.0015% groups showed relatively similar cell morphology to that observed in the control group. Also, in our in vivo study, severe punctate epithelial keratitis was seen on corneal fluorescein staining examination, and increased cellular shrinkage, surface desquamation, increased inflammation and decreased cell viability was observed on histological examination in BAC-containing agents compared to BAC-free agents. These results correspond with previous studies about ocular surface toxicity by BAC. Liang et al. [18] determined that rabbit corneoconjunctival surfaces presented a better tolerance when treated with PF tafluprost compared with commercially available BAC-containing latanoprost or BAC solution. Pauly et al. [25] reported that rabbit corneoconjunctival surfaces presented better tolerance when treated with PF latanoprost compared with the standard BAC-containing latanoprost preparation or the BAC solution.
In our study, immunofluorescence staining revealed that inflammatory markers, TNF-α, IL-6, and HLA DR were more highly expressed in groups of preservative-containing eye drops than the control group. However, polyquad-preserved and PF groups had lower inflammatory markers-positive cells than BAC-preserved groups. Other in vitro and in vivo studies have also reported on the inflammatory effects of BAC. One study reported that PF latanoprost-treated rabbit eyes showed no or slight infiltration of CD 45+ inflammatory cells in the cornea and conjunctiva. BAC 0.02% and BAC-containing latanoprost induced numerous CD 45+ inflammatory cells in the cornea and conjunctiva [1825]. In other studies, travoprost/timolol with polyquad was not significantly different from PBS for immunopositive cell counts of CD 45. However, travoprost/timolol with BAC and latanoprost/timolol with BAC induced significantly more CD 45+ cells than PBS and travoprost/timolol with polyquad. Travoprost/timolol with BAC also induced significantly fewer CD 45+ cells than latanoprost/timolol with BAC [2326]. The JNK pathway has been shown to play a major role in apoptosis in many cell death paradigms [2728]. In our study, immunofluorescent staining of pJNK was higher in the groups of 0.02% BAC-containing eye drops than the control group; polyquad-preserved, low-BAC-preserved and PF groups had significantly lower pJNK-positive cells than the 0.02% BAC group. These results correspond with other studies about lower apoptosis with PF and alternatively-preserved eye drops than BAC-containing eye drops. Baudouin et al. [15] evaluated the apoptotic marker annexinV/7-AAD; no increase in apoptosis and necrosis was observed in cells treated with PBS or with travoprost Z without BAC compared with cells treated with travoprost with BAC, latanoprost with BAC or BAC in their in vitro study. Other in vivo studies evaluated apoptosis by TUNEL assay. Few TUNEL+ apoptotic cells were observed after PBS or PF tafluprost instillations. For latanoprost, more TUNEL+ apoptotic cells in the limbus and in the conjunctiva were seen. Similar counts were found for BAC instillation compared with both PBS and PF tafluprost groups [1825].
We investigated the pAkt expression in corneal tissue to evaluate the cell viability influenced by preservatives. Immunofluorescence staining of pAkt showed lower expression in BAC groups than the control group. On the other hand, 0.02% BAC-containing bimatoprost and latanoprost groups had slightly higher expression of pAKt than the 0.02% BAC-alone group. As in other studies, these results suggest a possible protective effect of PGs against BAC-induced toxicity to the ocular surface [1629]. Further study will be needed to investigate the protective effect of PGs.
Although our mouse-model study does not closely mimic clinical practice, it is valuable for comparing toxicity of various drugs. If a drug tested in this model is toxic, then it is likely that it will be toxic in a clinical setting. This is particularly true for eye drops administered chronically. However, no in vivo or in vitro models can replace clinical assessment or reflect the real life of patients because of the vast variety of clinical situations, such as duration of treatment, number of drugs administered chronically and individual sensitivity to eye drops.
As discussed above, BAC-containing PGs induced more ocular surface damage, increased inflammatory and apoptotic markers and decreased cell viability than PF or alternatively preserved PGs in this in vivo study.
Chronic use of topical medications is needed in several eye diseases, including dry eye syndrome or glaucoma; they usually require two or more eye drops for treatment. Using many eye drops to control the disease increases the ocular surface BAC accumulation and induces the risk of side-effect occurrence. Therefore, PF or alternatively preserved topical medications seem to be a reasonable and viable alternative to those preserved with BAC. More studies are needed to apply these results to human eyes.
Figures and Tables
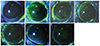 | Fig. 1Corneal fluorescein staining of seven C57BL/6 mice groups. (A) Control, (B) 0.02% benzalkonium chloride (BAC), (C) bimatoprost 0.01% (BAC 0.02%), (D) latanoprost 0.005% (BAC 0.02%), (E) travoprost 0.004% (with 0.001% polyquad), (F) tafluprost 0.0015%(BAC 0.001%), and (G) tafluprost 0.0015% (preservative-free). Severe punctuate epithelial keratitis was seen in the groups of 0.02% BAC (B), 0.02% BAC contained bimatoprost 0.01% (C) and latanoprost 0.005% (D). |
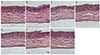 | Fig. 2Histopathologic examination by H&E stain for each group. (A) Control, (B) 0.02% benzalkonium chloride (BAC), (C) bimatoprost 0.01% (BAC 0.02%), (D) latanoprost 0.005% (BAC 0.02%), (E) travoprost 0.004% (with 0.001% polyquad), (F) tafluprost 0.0015% (BAC 0.001%), and (G) tafluprost 0.0015% (preservative-free). The surface desquamation, irregular surface, loss of cell borders, anisocytosis and stromal shrinkage were shown in the groups of BAC contained eye drops (B,C,D,F). However, travoprost 0.004% (with 0.001% polyquad) (E) and preservative-free tafluprost 0.0015% (G) groups were similar morphology with control group (A). |
 | Fig. 3Immunofluorescence staining of TNF-α positive cells in the corneal section (A-G) and the density of TNF-α fluorescence (H). (A) Control, (B) 0.02% benzalkonium chloride (BAC), (C) bimatoprost 0.01% (BAC 0.02%), (D) latanoprost 0.005% (BAC 0.02%), (E) travoprost 0.004% (with 0.001% polyquad), (F) tafluprost 0.0015% (BAC 0.001%), and (G) tafluprost 0.0015% (preservative-free, PF). Immunofluorescence staining of TNF-α was expressed more in preservative contained eye drops groups than control group. But, travoprost 0.004% (with 0.001% polyquad) and preservative-free tafluprost 0.0015% groups had significantly lower TNF-α positive cells than 0.02% BAC group. And BAC contained tafluprost 0.0015% has more TNF-α positive cells than preservative-free one. *,**p < 0.05, #p < 0.02. |
 | Fig. 4Immunofluorescence staining of IL-6 positive cells in the corneal section (A-G) and the density of IL-6 fluorescence (H). (A) Control, (B) 0.02% benzalkonium chloride (BAC), (C) bimatoprost 0.01% (BAC 0.02%), (D) latanoprost 0.005% (BAC 0.02%), (E) travoprost 0.004% (with 0.001% polyquad), (F) tafluprost 0.0015% (BAC 0.001%), and (G) tafluprost 0.0015% (preservative-free, PF). Immunofluorescence staining of IL-6 was highly expressed in 0.02% BAC contained eye drops groups than control group. But, travoprost 0.004% (with 0.001% polyquad), 0.001% BAC contained tafluprost 0.0015% and preservative-free tafluprost 0.0015% groups had significantly lower IL-6 positive cells than 0.02% BAC group. And BAC contained tafluprost 0.0015% has more IL-6 positive cells than preservative-free one. *p < 0.006, **,#p < 0.005. |
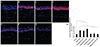 | Fig. 5Immunofluorescence staining of HLA DR positive cells in the corneal section (A-G) and the density of HLA DR fluorescence (H). (A) Control, (B) 0.02% benzalkonium chloride (BAC), (C) bimatoprost 0.01% (BAC 0.02%), (D) latanoprost 0.005% (BAC 0.02%), (E) travoprost 0.004% (with 0.001% polyquad), (F) tafluprost 0.0015% (BAC 0.001%), and (G) tafluprost 0.0015% (preservative-free, PF). Immunofluorescence staining of HLA DR was highly expressed in 0.02% BAC contained eye drops groups than control group. But, travoprost 0.004% (with 0.001% polyquad), 0.001% BAC contained tafluprost 0.0015% and preservative-free tafluprost 0.0015% groups had significantly lower HLA DR positive cells than 0.02% BAC group. *p < 0.02, **p < 0.01. |
 | Fig. 6Immunofluorescence staining of pJNK positive cells in the corneal section (A-G) and the density of pJNK fluorescence (H). (A) Control, (B) 0.02% benzalkonium chloride (BAC), (C) bimatoprost 0.01% (BAC 0.02%), (D) latanoprost 0.005% (BAC 0.02%), (E) travoprost 0.004% (with 0.001% polyquad), (F) tafluprost 0.0015% (BAC 0.001%), and (G) tafluprost 0.0015% (preservative-free, PF). Immunofluorescence staining of pJNK was highly expressed in 0.02% BAC contained eye drops groups than control group. But, travoprost 0.004% (with 0.001% polyquad), 0.001% BAC contained tafluprost 0.0015% and preservative-free tafluprost 0.0015% groups had significantly lower pJNK positive cells than 0.02% BAC group. *p < 0.03, **p < 0.05. |
 | Fig. 7Immunofluorescence staining of pAkt positive cells in the corneal section (A-G) and the density of pAkt fluorescence (H). (A) Control, (B) 0.02% benzalkonium chloride (BAC), (C) bimatoprost 0.01% (BAC 0.02%), (D) latanoprost 0.005% (BAC 0.02%), (E) travoprost 0.004% (with 0.001% polyquad), (F) tafluprost 0.0015% (BAC 0.001%), and (G) tafluprost 0.0015% (preservative-free, PF). Immunofluorescence staining of pAkt shows significantly lower expression in 0.02 % BAC and latanoprost 0.005 % (BAC 0.02 %) than control group. PF tafluprost 0.0015 % group had significantly higher expression than control group. Moreover, PF tafluprost 0.0015 % group had significantly higher expression than 0.02 % BAC and preserved tafluprost 0.0015 % group. *,**p < 0.05, #p < 0.01. |
References
1. Goldberg I, Cunha-Vaz J, Jakobsen JE, et al. Comparison of topical travoprost eye drops given once daily and timolol 0.5% given twice daily in patients with open-angle glaucoma or ocular hypertension. J Glaucoma. 2001; 10:414–422.
2. Alm A, Stjernschantz J. Effects on intraocular pressure and side effects of 0.005% latanoprost applied once daily, evening or morning: a comparison with timolol. Scandinavian Latanoprost Study Group. Ophthalmology. 1995; 102:1743–1752.
3. Sherwood M, Brandt J. Bimatoprost Study Groups 1 and 2. Six-month comparison of bimatoprost once-daily and twice-daily with timolol twice-daily in patients with elevated intraocular pressure. Surv Ophthalmol. 2001; 45:Suppl 4. S361–S368.
4. Broadway DC, Grierson I, O'Brien C, Hitchings RA. Adverse effects of topical antiglaucoma medication.The outcome of filtration surgery. Arch Ophthalmol. 1994; 112:1446–1454.
5. Baudouin C. Allergic reaction to topical eyedrops. Curr Opin Allergy Clin Immunol. 2005; 5:459–463.
6. Noecker RJ, Herrygers LA, Anwaruddin R. Corneal and conjunctival changes caused by commonly used glaucoma medications. Cornea. 2004; 23:490–496.
7. Jaenen N, Baudouin C, Pouliquen P, et al. Ocular symptoms and signs with preserved and preservative-free glaucoma medications. Eur J Ophthalmol. 2007; 17:341–349.
8. Brasnu E, Brignole-Baudouin F, Riancho L, et al. In vitro effects of preservative-free tafluprost and preserved latanoprost, travoprost, and bimatoprost in a conjunctival epithelial cell line. Curr Eye Res. 2008; 33:303–312.
9. Baudouin C. The ocular surface in glaucoma. Cornea. 2009; 28:S14–S19.
10. Baudouin C, Labbe A, Liang H, et al. Preservatives in eyedrops: the good, the bad and the ugly. Prog Retin Eye Res. 2010; 29:312–334.
11. Baudouin C, Pisella PJ, Fillacier K, et al. Ocular surface inf lammatory changes induced by topical antiglaucoma drugs: human and animal studies. Ophthalmology. 1999; 106:556–563.
12. Pisella PJ, Pouliquen P, Baudouin C. Prevalence of ocular symptoms and signs with preserved and preservative free glaucoma medication. Br J Ophthalmol. 2002; 86:418–423.
13. Noecker R. Effects of common ophthalmic preservatives on ocular health. Adv Ther. 2001; 18:205–215.
14. De Saint Jean M, Brignole F, Bringuier AF, et al. Effects of benzalkonium chloride on growth and survival of Chang conjunctival cells. Invest Ophthalmol Vis Sci. 1999; 40:619–630.
15. Baudouin C, Riancho L, Warnet JM, Brignole F. In vitro studies of antiglaucomatous prostaglandin analogues: travoprost with and without benzalkonium chloride and preserved latanoprost. Invest Ophthalmol Vis Sci. 2007; 48:4123–4128.
16. Guenoun JM, Baudouin C, Rat P, et al. In vitro study of inflammatory potential and toxicity profile of latanoprost, travoprost, and bimatoprost in conjunctiva-derived epithelial cells. Invest Ophthalmol Vis Sci. 2005; 46:2444–2450.
17. Lee JK, Ryu YH. The effect of antiglaucoma medication on cultured human conjunctival epithelial cells. J Korean Ophthalmol Soc. 2006; 47:1811–1818.
18. Liang H, Baudouin C, Pauly A, Brignole-Baudouin F. Conjunctival and corneal reactions in rabbits following short- and repeated exposure to preservative-free tafluprost, commercially available latanoprost and 0.02% benzalkonium chloride. Br J Ophthalmol. 2008; 92:1275–1282.
19. Tripathi BJ, Tripathi RC, Kolli SP. Cytotoxicity of ophthalmic preservatives on human corneal epithelium. Lens Eye Toxic Res. 1992; 9:361–375.
20. Asada H, Takaoka-Shichijo Y, Nakamura M, Kimura A. Optimization of benzalkonium chloride concentration in 0.0015% tafluprost ophthalmic solution from the points of ocular surface safety and preservative efficacy. Yakugaku Zasshi. 2010; 130:867–871.
21. Nakagawa S, Usui T, Yokoo S, et al. Toxicity evaluation of antiglaucoma drugs using stratified human cultivated corneal epithelial sheets. Invest Ophthalmol Vis Sci. 2012; 53:5154–5160.
22. Liang H, Baudouin C, Labbe A, et al. Conjunctiva-associated lymphoid tissue (CALT) reactions to antiglaucoma prostaglandins with or without BAK-preservative in rabbit acute toxicity study. PLoS One. 2012; 7:e33913.
23. Liang H, Brignole-Baudouin F, Riancho L, Baudouin C. Reduced in vivo ocular surface toxicity with polyquad-preserved travoprost versus benzalkonium-preserved travoprost or latanoprost ophthalmic solutions. Ophthalmic Res. 2012; 48:89–101.
24. Kim EJ, Kim YH, Kang SH, et al. In vitro effects of preservative-free and preserved prostaglandin analogs on primary cultured human conjunctival fibroblast cells. Korean J Ophthalmol. 2013; 27:446–453.
25. Pauly A, Roubeix C, Liang H, et al. In vitro and in vivo comparative toxicological study of a new preservative-free latanoprost formulation. Invest Ophthalmol Vis Sci. 2012; 53:8172–8180.
26. Liang H, Brignole-Baudouin F, Pauly A, et al. Polyquad-preserved travoprost/timolol, benzalkonium chloride (BAK)-preserved travoprost/timolol, and latanoprost/timolol in fixed combinations: a rabbit ocular surface study. Adv Ther. 2011; 28:311–325.
27. Lu L, Wang L, Shell B. UV-induced signaling pathways associated with corneal epithelial cell apoptosis. Invest Ophthalmol Vis Sci. 2003; 44:5102–5109.
28. Chen F. JNK-induced apoptosis, compensatory growth, and cancer stem cells. Cancer Res. 2012; 72:379–386.
29. Niwano Y, Iwasawa A, Ayaki M. Ocular surface cytotoxicity and safety evaluation of tafluprost, a recently developed anti-glaucoma prostaglandin analog. Ophthalmol Eye Dis. 2014; 6:5–12.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download