Abstract
Purpose
Though there are many reports regarding the structure-function relationship in glaucoma, they are too complicated to apply to the routine clinical setting. The aim of this study was to investigate the direct relationship between peripapillary retinal nerve fiber layer (RNFL) thickness measured by optical coherence tomography (OCT) and visual field (VF) severity indices computed by standard automated perimetry.
Methods
This cross-sectional comparative study included 104 glaucomatous patients and 59 healthy subjects. Peripapillary RNFL thickness was measured by spectral domain (SD) and time domain (TD) OCTs. Four glaucoma VF severity indices, including mean deviation (MD), pattern standard deviation (PSD), Collaborative Initial Glaucoma Treatment Study (CIGTS) VF score, and Advanced Glaucoma Intervention Study (AGIS) VF score, were calculated using standard automated perimetry. The Pearson's correlation coefficients (r) between the average and quadrants of peripapillary RNFL thicknesses and the four VF severity indices were calculated.
Results
In glaucomatous eyes, the r value between the average RNFL thickness measured by SD OCT and each VF severity index were 0.562, -0.514, -0.577, and -0.567 for the MD, PSD, CIGTS VF score, and AGIS VF score, respectively (all p < 0.001). Among each quadrant, the inferior RNFL thickness showed the largest r value; 0.587, -0.552, -0.613, and -0.598 for the MD, PSD, CIGTS VF score, and AGIS VF score, respectively (all p < 0.001). Measurements by TD OCT showed similar strengths of association with SD OCT.
Conclusions
Moderate correlation was identified between peripapillary RNFL thicknesses measured by SD/TD OCT and glaucoma VF severity indices. Among each quadrant, the inferior RNFL thickness showed the greatest association with glaucoma VF severity indices. There was no significant difference according to the type of VF severity index or the type of OCTs.
Selective loss of retinal ganglion cells (RGCs) and thinning of their axonal layer, known as the retinal nerve fiber layer (RNFL), are characteristic structural changes in glaucomatous eyes. Functional visual field (VF) deficits are generally detected at the corresponding area of these structural alterations [1234]. In the last decade, various new technologies have been developed to evaluate the structural changes secondary to RGC damage [5], one of which is optical coherence tomography (OCT) [67]. This newly developed spectral domain (SD) OCT acquires real-time depth scans and ultrahigh-resolution tomographic intraretinal images. Therefore, it allows for more precise measurements of RNFL thickness than conventional time domain (TD) OCT [89]. Even though there are several studies comparing the use of these two technologies for peripapillary RNFL thickness measurements [10111213], the potential difference in their association with glaucomatous VF losses has not yet been well studied [13].
In the present study, the correlation between peripapillary RNFL thickness measured by SD/TD OCTs and VF severity indices computed using standard automated perimetry (SAP) was determined. Regarding the VF severity indices, not only the global indices of SAP but also two scoring systems used in major glaucoma trials (Collaborative Initial Glaucoma Treatment Study, CIGTS; Advanced Glaucoma Intervention Study, AGIS) were assessed [1415].
After obtaining approval from our institutional review board of Gangnam Severance Hospital, Yonsei University College of Medicine, 188 subjects (age, 20 to 70 years old) who either had open angle glaucoma or who were healthy were enrolled in this study. The study protocol adhered to the tenets of the Declaration of Helsinki, and all participants gave informed consent.
Each participant underwent a comprehensive ophthalmologic examination, including a review of medical history, best-corrected visual acuity (BCVA), intraocular pressure (IOP) measurement with Goldmann applanation tonometry, central corneal thickness (CCT) measurement by ultrasonography, slit-lamp biomicroscopy, gonioscopy, stereoscopic optic disc examination with a 90-diopter lens, indirect fundus examination after pupil dilation, red-free RNFL photography (Heidelberg Retina Angiograph 1; Heidelberg Engineering, Dossenheim, Germany) [516], and SAP using the 30-2 Swedish interactive threshold algorithm standard strategy (Humphrey Field Analyzer II; Carl Ziess Meditec, Dublin, CA, USA). Peripapillary RNFL scans were performed using SD Cirrus HD OCT (Carl Zeiss Meditec) and TD Stratus OCT (Carl Zeiss Meditec). All tests were completed within one month.
To be included in the study, subjects had to have a spherical refractive error within ±4.00 diopters, a cylinder refractive error within ±3.00 diopters, and open anterior chamber angles on gonioscopic examination. Subjects who had any history of ocular trauma, intraocular surgery, or laser treatment were excluded; however, the use of ocular hypotensive medications was permitted in glaucoma patients. All participants with diabetes or any other disease or medication affecting the VF or RNFL were also excluded. One eye from each subject was randomly selected for data analysis if they both satisfied the entry criteria. The diagnosis of glaucoma was based on the presence of optic nerve head or RNFL changes characteristic of glaucoma and the presence of glaucomatous VF loss on two consecutive tests [5]. Healthy controls had no VF loss and no optic nerve head or RNFL changes suggestive of glaucoma, and also had a maximal IOP less than 21 mmHg without the use of any ocular hypotensive medication.
For each eye, the peripapillary RNFL thickness was measured by an Optic Disc Cube 200 × 200 Scan using the Cirrus HD OCT (model 4000, software ver. 3.0) and by a Fast RNFL Scan using the Stratus OCT (model 3000, software ver. 4.0). The Fast RNFL Scan of Stratus OCT consists of 256 axial scans along a circle with a diameter of 3.46 mm. The center of the circular scan was manually positioned at the optic disc center. Meanwhile, the Optic Disc Cube 200 × 200 Scan of Cirrus HD OCT obtains 200 × 200 axial scans in a 6 × 6 mm2 optic disc area and extracts 256 axial scans along the path of the comparable calculation circle with a 3.46 mm diameter. The center of the calculation circle was automatically determined but it was rectified if there was a significant misalignment. Measurements were taken without pupil dilation and were performed as previously described elsewhere [17]. Scans with blinks or with a low signal strength (≤6) were excluded from the analysis. Corrected centering and RNFL segmentation were checked for each OCT image. All tests were performed by the same operator.
The VF was considered unreliable when fixation losses were greater than 20%, or false positive/negative errors were greater than 15%. The mean deviation (MD) and pattern standard deviation (PSD) were recorded as VF global indices of the SAP result. The CIGTS VF score was manually calculated based on the probabilities in the total deviation probability plot over the region of the field covered by the Humphrey 24-2 VF test (52 points) [15]. The AGIS VF score was also manually calculated [14]. Both the CIGTS and AGIS VF scores ranged from 0 (no defect) to 20 (all test sites deeply depressed).
Statistics including a Student t-test, Pearson's correlation coefficient (r) and regression analysis were performed using the SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA). Steiger's Z-test was computed using the FZT Computator (http://psych.unl.edu/psycrs/statpage/regression.html) and the corrected Akaike's information criteria (AIC) was calculated using QuickCalcs (http://www.graphpad.com/quickcalcs/AIC1.cfm). A p-values less than 0.05 was considered statistically significant.
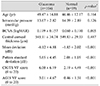
One hundred and sixty-three eyes of 163 subjects were analyzed; 104 eyes belonged to the glaucoma group and 59 eyes belonged to the healthy group. The overall demographics and four VF severity indices (MD, PSD, CIGTS VF score, and AGIS VF score) are shown in Table 1. Though the BCVA of the glaucoma group was poorer than that of the healthy group, other characteristics including age, IOP, and CCT were similar in the two study groups. All four glaucoma VF severity indices were worse in the glaucoma group compared to the healthy group (all p < 0.001).
In both groups, the correlations between VF severity indices were calculated (Table 2). For the glaucoma group, the MD and CIGTS VF score showed the best association with each other (r = -0.992, p < 0.001). Overall, the PSD showed the worst association with the other three VF indices.
The overall average and average by quadrant of the peripapillary RNFL thicknesses, measured by the two OCTs, are shown in Table 3. For both study groups, the average RNFL thickness measured by SD OCT was thinner than that measured by TD OCT.
The strengths of association between the RNFL thicknesses and the VF severity indices were computed. In the glaucoma group (Table 4), for both types of OCT, the VF indices showed moderate correlations with the average/quadrant average RNFL thicknesses. The r values between the average RNFL thickness measured by SD OCT and the VF severity index were 0.562, -0.514, -0.577, and -0.567 for MD, PSD, CIGTS VF score, and AGIS VF score, respectively (all p < 0.001). Among the quadrants, the inferior RNFL thickness showed the largest r value; 0.587, -0.552, -0.613, and -0.598 for MD, PSD, CIGTS VF score, and AGIS VF score, respectively (all p < 0.001). Measurements by TD OCT showed similar strengths of association as those by SD OCT. Using Steiger's Z-test, the association strength between the VF severity indices and the RNFL thicknesses was not significantly different according to the type of OCT. In the healthy group, for both types of OCT, none of the VF severity indices were significantly associated with the RNFL thickness of any of the scanned sectors.
In the glaucoma group, the coefficients of determination (R2) of RNFL thicknesses and VF severity indices were calculated for linear, second order polynomial, and third order polynomial regressions (Table 5). For all indices, the R2 value was below 0.400.
Overall, a moderate correlation was identified between the peripapillary RNFL thicknesses measured by SD/TD OCT and the glaucoma VF severity indices in this study. The absolute values of the correlation coefficients for the average RNFL thickness were just greater than 0.5, even in the glaucoma group. This may have been due to not only the severity of glaucomatous damage but also many other factors that influence the absolute values of peripapillary RNFL thickness and their measurements [18192021]. Age, ethnicity, axial length, and optic disc size affect the RNFL thickness. Type of imaging device, pupil size, presence of cataracts, and refractive errors affect the measurement of RNFL thickness.
In the normal group, for any scanned sector, the RNFL thickness showed no association with any of the VF indices. This implies that the interference from factors other than VF status may have been more remarkable in the normal group. In the glaucoma group, there was a significant relationship between glaucomatous VF loss and RNFL thinning. According to the AIC, the average RNFL thickness and all four VF severity indices better fit a linear model rather than a curvilinear regression model. These results are somewhat different from previous studies [22]. This might be due to the fact that we performed the regression analyses only in glaucoma patients.
With the exception of the PSD, the other three VF severity indices showed good associations with one another (the absolute value of r was approximately 0.9 for the glaucoma group and approximately 0.8 for the normal group). The PSD had a much weaker association with the other three indices (the absolute value of r was approximately 0.7 for the glaucoma group and 0.4 for the normal group). In addition, although not statistically significant, the PSD was more likely to have a weaker strength of association with the average RNFL thickness than the other indices. This is presumed because there are innate characteristics of each VF severity index. The MD shows how much, on average, the whole field departs from normal and is a weighted average of the decibel deviations shown in the total deviation plot. The CIGTS and AGIS VF scores also indicate the extent and degree of decreased light sensitivity of the entire field. In contrast, the PSD reflects irregularities in the field, such as those caused by localized VF defects. Due to this unique characteristic, the PSD showed a different pattern of association with the RNFL thickness compared to the other indices.
In addition, in this study, the RNFL thickness measurements made by SD Cirrus HD OCT did not completely agree with those made by TD Stratus OCT. Since there is no definitive histological data to explain this difference, we are still uncertain as to its cause. However, this difference has been recently reported [10111213].
According to our study, although the RNFL thicknesses measured by two different OCT devices were not equal, their relationship with various VF parameters was very similar. Our data are consistent with other reports by Leung et al. [23], which evaluated the association between average RNFL thickness and MD expressed on a decibel scale in normal and glaucomatous eyes. They showed that the correlation between structure and function was similar according to either TD OCT or SD OCT. This is considered to be because the conventional achromatic VF test has a relatively low sensitivity compared to the OCTs; the difference in measured RNFL thickness between SD OCT and TD OCT is not large enough to affect the results of VF.
In conclusion, we identified a moderate correlation between peripapillary RNFL thicknesses measured by SD/TD OCT and the severity indices of glaucomatous VF deficits. Among the various quadrants, the inferior RNFL thickness showed the greatest association with glaucoma VF severity indices. There was no significant difference according to the type of VF severity index or the type of OCT.
Figures and Tables
Table 3
Peripapillary retinal nerve fiber layer thickness as measured by spectral domain Cirrus HD OCT and time domain Stratus OCT in glaucoma and healthy eyes

Table 4
Correlation between the peripapillary retinal nerve fiber layer thicknesses measured by OCTs and the VF severity scores in glaucomatous eyes

OCT = optical coherence tomography; VF = visual field; CIGTS = Collaborative Initial Glaucoma Treatment Study; AGIS = Advanced Glaucoma Intervention Study; r = Pearson's correlation coefficient; Z-score = critical value of Steiger's Z-test; Avg = average; C = Cirrus HD OCT; S = Stratus OCT; Sup = superior quadrant; Nas = nasal quadrant; Inf = inferior quadrant; Tem = temporal quadrant.
*p < 0.05 for Z-score.
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (no. 2010-0008721 and 2011-0013288), and a grant from the Korea Health Technology R&D Project, Ministry of Health and Welfare (no. A101727).
Notes
References
1. Sommer A, Katz J, Quigley HA, et al. Clinically detectable nerve fiber atrophy precedes the onset of glaucomatous field loss. Arch Ophthalmol. 1991; 109:77–83.
2. Johnson CA, Cioffi GA, Liebmann JR, et al. The relationship between structural and functional alterations in glaucoma: a review. Semin Ophthalmol. 2000; 15:221–233.
3. Harwerth RS, Vilupuru AS, Rangaswamy NV, Smith EL 3rd. The relationship between nerve fiber layer and perimetry measurements. Invest Ophthalmol Vis Sci. 2007; 48:763–773.
4. Hood DC, Kardon RH. A framework for comparing structural and functional measures of glaucomatous damage. Prog Retin Eye Res. 2007; 26:688–710.
5. Hong S, Ahn H, Ha SJ, et al. Early glaucoma detection using the Humphrey Matrix Perimeter, GDx VCC, Stratus OCT, and retinal nerve fiber layer photography. Ophthalmology. 2007; 114:210–215.
6. Huang D, Swanson EA, Lin CP, et al. Optical coherence tomography. Science. 1991; 254:1178–1181.
7. Hee MR, Izatt JA, Swanson EA, et al. Optical coherence tomography of the human retina. Arch Ophthalmol. 1995; 113:325–332.
8. Menke MN, Knecht P, Sturm V, et al. Reproducibility of nerve fiber layer thickness measurements using 3D fourier-domain OCT. Invest Ophthalmol Vis Sci. 2008; 49:5386–5391.
9. Pagliara MM, Lepore D, Balestrazzi E. The role of OCT in glaucoma management. Prog Brain Res. 2008; 173:139–148.
10. Vizzeri G, Weinreb RN, Gonzalez-Garcia AO, et al. Agreement between spectral-domain and time-domain OCT for measuring RNFL thickness. Br J Ophthalmol. 2009; 93:775–781.
11. Knight OJ, Chang RT, Feuer WJ, Budenz DL. Comparison of retinal nerve fiber layer measurements using time domain and spectral domain optical coherent tomography. Ophthalmology. 2009; 116:1271–1277.
12. Kim JS, Ishikawa H, Gabriele ML, et al. Retinal nerve fiber layer thickness measurement comparability between time domain optical coherence tomography (OCT) and spectral domain OCT. Invest Ophthalmol Vis Sci. 2010; 51:896–902.
13. Lee JR, Jeoung JW, Choi J, et al. Structure-function relationships in normal and glaucomatous eyes determined by time- and spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2010; 51:6424–6430.
14. Advanced Glaucoma Intervention Study. 2. Visual field test scoring and reliability. Ophthalmology. 1994; 101:1445–1455.
15. Gillespie BW, Musch DC, Guire KE, et al. The collaborative initial glaucoma treatment study: baseline visual field and test-retest variability. Invest Ophthalmol Vis Sci. 2003; 44:2613–2620.
16. Hong S, Moon JW, Ha SJ, et al. Evaluation of a new scoring system for retinal nerve fiber layer photography using HRA1 in 964 eyes. Korean J Ophthalmol. 2007; 21:216–221.
17. Budenz DL, Fredette MJ, Feuer WJ, Anderson DR. Reproducibility of peripapillary retinal nerve fiber thickness measurements with stratus OCT in glaucomatous eyes. Ophthalmology. 2008; 115:661–666.e4.
18. Budenz DL, Anderson DR, Varma R, et al. Determinants of normal retinal nerve fiber layer thickness measured by Stratus OCT. Ophthalmology. 2007; 114:1046–1052.
19. Parikh RS, Parikh SR, Sekhar GC, et al. Normal age-related decay of retinal nerve fiber layer thickness. Ophthalmology. 2007; 114:921–926.
20. Nagai-Kusuhara A, Nakamura M, Fujioka M, et al. Association of retinal nerve fibre layer thickness measured by confocal scanning laser ophthalmoscopy and optical coherence tomography with disc size and axial length. Br J Ophthalmol. 2008; 92:186–190.
21. Savini G, Zanini M, Barboni P. Influence of pupil size and cataract on retinal nerve fiber layer thickness measurements by Stratus OCT. J Glaucoma. 2006; 15:336–340.
22. Leung CK, Chan WM, Chong KK, et al. Comparative study of retinal nerve fiber layer measurement by StratusOCT and GDx VCC, I: correlation analysis in glaucoma. Invest Ophthalmol Vis Sci. 2005; 46:3214–3220.
23. Leung CK, Cheung CY, Weinreb RN, et al. Retinal nerve fiber layer imaging with spectral-domain optical coherence tomography: a variability and diagnostic performance study. Ophthalmology. 2009; 116:1257–1263.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download