Abstract
Purpose
Blind individuals who have photoreceptor loss are known to perceive phosphenes with electrical stimulation of their remaining retinal ganglion cells. We proposed that implantable lateral geniculate body (LGB) stimulus electrode arrays could be used to generate phosphene vision. We attempted to refine the basic reference of the electrical evoked potentials (EEPs) elicited by microelectrical stimulations of the optic nerve, optic tract and LGB of a domestic pig, and then compared it to visual evoked potentials (VEPs) elicited by short-flash stimuli.
Methods
For visual function measurement, VEPs in response to short-flash stimuli on the left eye of the domestic pig were assessed over the visual cortex at position Oz with the reference electrode at Fz. After anesthesia, linearly configured platinum wire electrodes were inserted into the optic nerve, optic track and LGB. To determine the optimal stimulus current, EEPs were recorded repeatedly with controlling the pulse and power. The threshold of current and charge density to elicit EEPs at 0.3 ms pulse duration was about ±10 µA.
Results
Our experimental results showed that visual cortex activity can be effectively evoked by stimulation of the optic nerve, optic tract and LGB using penetrating electrodes. The latency of P1 was more shortened as the electrical stimulation was closer to LGB. The EEPs of two-channel in the visual cortex demonstrated a similar pattern with stimulation of different spots of the stimulating electrodes. We found that the LGB-stimulated EEP pattern was very similar to the simultaneously generated VEP on the control side, although implicit time deferred.
Conclusions
EEPs and VEPs derived from visual-system stimulation were compared. The LGB-stimulated EEP wave demonstrated a similar pattern to the VEP waveform except implicit time, indicating prosthetic-based electrical stimulation of the LGB could be utilized for the blind to perceive vision of phosphenes.
Irreversible blindness (caused by glaucoma, retinitis pigmentosa, age-related macular degeneration, etc.) is largely due to photoreceptor loss and a resultant reduced light sensitivity, while the rest of the visual system remains mainly undamaged [1]. In 1929, researchers discovered blind individuals could perceive a small point of light (phosphene) with electrical stimulation of the occipital lobe of the human cortex, suggesting vision analogue could be generated [2]. Several alternative target sites for electrical stimulation were suggested because retinotopy of the visual cortex was too coarse to generate high spatial resolution of a single perceived image [3]. In addition, visual percepts after optic nerve stimulation have been reported as similar to those seen after cortical stimulation [4]. While epiretinal and subretinal prostheses have been attempted to transmit electrical signals from the eye to the visual cortex through visual circuitry, the majority of blind individuals have a defect in visual circuitry and frequently few functional ganglion cells remain [5,6]. Therefore, the other visual prosthesis was trying to detour these systems and they considered direct intervention at the cortical level.
We are particularly interested in stimulation of the lateral geniculate body (LGB) positioned between the optic nerve and visual cortex because its receptive fields are well characterized and the functional areas of the magnocellular and parvocellular pathways are separate [7]. Although the LGB has structural and functional advantages, relatively little is known about its stimulation due to the intricacy of surgical access to it. Recently, however, the placement of deep brain stimulating electrodes in thalamic structures was successfully achieved for Parkinson's disease therapy [8]. Our experiments were performed in pigs because of their ocular morphology and physiology is very similar to humans [9].
Previously, Kara et al. [10] demonstrated the effectiveness of an extracellular method of measuring subthreshold afferent input to the visual cortex. Pezaris and Reid [11] established the feasibility of artificially created visual percepts through electrical stimulation of the lateral geniculate nucleus (LGN). Their experiments were performed in alert monkeys, using behavioral report to assess the effects of stimulation. They also reported that the placement of microwire electrodes would be available while clinical electrodes spacing as large as 600 µm in three dimensions would allow for over 250 phosphenes [12].
In this study, we attempted to see whether electrical evoked potentials (EEPs) elicited by microelectrical stimulations of the optic nerve, optic tract and LGB of an animal would treat electrical percepts in the same manner as the visual evoked potentials (VEPs) elicited by short flash stimuli on the eye.
For visual function measurement, a 10-month-old domestic male pig (Sus scrofa domesticus), weighing approximately 30 kg, was used. The animal was treated according to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. A standard anesthesia protocol was employed before electrical stimulation. The animal was sedated with an intramuscular injection (ketamine, 22 mg/kg; xylazine, 2 mg/kg; and glycopyrrolate, 0.01 mg/kg), masked with N2O, intubated, and maintained on N2O gas anesthesia. The pig was kept on intravenous maintenance fluids (lactated Ringer's solution 10 mL/kg/hr), and the heart rate and systemic arterial blood pressures were monitored throughout the procedures. After finishing the experiment, 1.2 g pentobarbital sodium and 150 mg phenytoin sodium were injected through a catheter in the central ear vein to euthanize the animals.
Visual stimuli were generated using a goggle type of flash stimulator (SLS-3100; Nihon Kohden, New York, NY, USA). at 5 to 20 Hz placed in front of the eyes of the animal. Flash stimuli effectiveness was defined by trials and error using different grades of filters. Visual acuity, in response to the flash stimulus, was assessed with VEPs. For electrical stimulation, the frontal lobe was removed and linearly configured platinum wire electrodes were implanted into the optic nerve, the optic tract and LGB of the right eye. Electric stimulations were delivered through an isolated biphasic stimulator (Grass 12; Astro-Med, West Warwick, RI, USA). The current stimulus consisted of a 0.3 ms negative square pulse was followed by 0.3 ms positive pulse and the amplitude of the current stimuli was varied between 0.5 and 5 mA. The VEP and EEP were recorded with a 2-channel data acquisition system using data collected on the scalp over the visual cortex at position Oz, referenced with position Fz.
We used a domestic pig as the experiment model, and the feasibility of visual prostheses based on penetrating electrical stimulations were investigated (Fig. 1). Firstly, VEP was performed on both eyes after sedation. The right side served as the experiment side and the left as the control. After anesthesia with endotracheal intubation, VEP was performed on the right eye. The globe was removed for exposure, a platinum wire electrode was implanted, and optic nerve EEP was done (Fig. 2). The frontal skull including right orbital wall was removed to visualize the right side intracranial optic nerve and optic chiasm. VEP was performed by implantation of the platinum wire electrode to the optic tract near the optic chiasm. Subsequently, the right frontal lobe and part of the temporal lobe were removed, and deep optic tract and LGB responses were recorded after direct electrical stimulation. Left eye VEP was done to compare with the right-side EEP.
In the domestic pig, the locations of the optic nerve, optic tract and LGB are analogous to those of human. Data were collected from the animal and consistent positive potentials were obtained for VEP (P75) and for EEPs of the intraorbital optic nerve (P70), optic tract (P37), and LGB (P36). For a control, we performed VEP in a normal left eye (Fig. 3A). An anesthesia effect was observed on VEPs after anesthesia in the right eye (Fig. 3B). The administration of anesthesia may modify the response to flash stimuli in comparison with a sedated animal. Characteristic peaks appeared in the early part of the waves N1 (40 msec) and P1 (70 msec) and with VEP on the optic nerve (Fig. 3). VEP of the right eye was observed as flat (Fig. 4A) after the optic nerve was severed. Overall peaks of EEP resembled those of VEP. However, when the optic nerve was stimulated with two electrodes (about 1.0 mm apart) the characteristic EEP patterns of each were different (Fig. 4B). EEPs on the optic tract and LGB were measured, and each showed short latency (P39 and P33, respectively) compared with P70 of VEP (Fig. 4C-4E). Finally, the skull of the animal was opened from the right frontal lobe to part of the temporal lobe, while VEP to flash stimuli on the left eye were recorded to compare with the preoperative VEP (Fig. 5).
The LGN is the first area to receive synaptic input from the axon of ganglion cells. Retinotopic organization makes it a potential target for artificial vision [13]. Studies in primate visual cortex indicate that the functional magnetic resonance imaging (fMRI) response is roughly correlated with local field potential activity when both are measured simultaneously [14]. From fMRI studies, a 3° retinal area of central vision is magnified anatomically in the LGN to occupy more than 60% of the LGN volume [15]. This enlarged LGN foveal representation could facilitate stimulation of the central visual area by an LGN-stimulated prosthetic device.
Even when there is substantial damage to the eye and possible atrophy of the optic nerve, the LGN remains largely intact. Therefore, stimulation of the LGN has the potential to be a viable treatment approach for many causes of blindness, including the most prevalent one as well as blindness caused by trauma to the eye or surgical enucleation. Approximately 10 mm away from the LGN are structures such as the subthalamic nucleus and substantia nigra that are accessed using surgical techniques from deep brain stimulation (DBS) for electrical stimulation treatment of movement disorders. This suggests that the same DBS techniques could be used to place stimulating electrodes in the LGN [12].
Many studies based on electrical stimulation of the human visual system promise to offer blind people the ability to see. Up to now, problems (in the stimulation of the visual cortex) exist in obtaining a useful correlation between a stimulus applied and the actual vision obtained. In stimulating the retina, other problems may occur because of the difficulty in pinpoint visual stimulation generating any useful degree of resolution. Unlike the non-homogeneous retinal visual field, the visual field of the LGB is homogeneous. Image-view mapping may be designed to take advantage of this as all of the currently known implants are based on a trial-and-error approach because little is known regarding exactly how the brain processes artificially-stimulated neurons. Experimental applications on humans proceed with extreme caution because of the potential risks involved. In our attempt to refine the basic reference ranges of EEPs on the optic nerve, optic tract and LGB, a model system was employed because of the ocular morphology and physiology is very similar to the human eye. It was difficult to define the magnocellular, parvocellular and koniocellular layers in the LGB of the pig, due to the varying exactness of and occasionally conflicting characterizations of the different functional cortex areas [16,17,18,19,20].
A limitation of this work is that as the number of subject in this study is small, and the result does not lead to a definite conclusion. In addition, further analysis as to whether wave-forms generated by LGN electrical stimulation can be processed in the brain and applied to movements is needed since actual stimuli responses such as walking and sitting were difficult to assess properly. Furthermore, no comprehensive functional mapping of the cortical areas has yet been defined in the animal. The results of the present study showed that a characteristic peak (P1) of EEP emerged earlier than that of the VEP and this may correlate with the different transmission between the electrical line and natural optical pathway including retina. EEP has similar peaks and amplitude to VEP, enabling us to conclude that electrical stimulation of LGB could mimic natural optical stimulation. Our findings support the idea that electrical microstimulation of the LGB creates visual percepts, or phosphenes, which could be interpreted as normal visual events.
Figures and Tables
 | Fig. 1Diagram of the procedure and taking the visual evoked potential (VEP) and electrical evoked potential (EEP) in this study. LGB = lateral geniculate body. |
 | Fig. 2The surgical approach to the retrobulbar optic nerve. Implantation of platinum wire electrode into the optic nerve. |
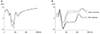 | Fig. 3Visual evoked potentials (VEPs) to the flash stimuli for the controlled object. (A) VEPs to the flash stimuli on a left eye. (B) VEPs to the flash stimuli on a right eye (before optic nerve was severed). |
 | Fig. 4Electrically-evoked potential (EEP) of optic nerve, optic track and lateral geniculate body (LGB). (A) Visual evoked potentials (VEPs) in response to short flash stimuli on a right eye of the pig after optic nerve was severed (control). (B) EEPs to electrical stimulations on optic nerve (right eye side). (C) EEPs to electrical stimulations on optic track before optic chiasm (right eye side). (D) EEPs to electrical stimulations on optic track after optic chiasm (right eye side). (E) EEPs to electrical stimulations on LGB (right eye side). |
References
1. MacLaren RE, Pearson RA, MacNeil A, et al. Retinal repair by transplantation of photoreceptor precursors. Nature. 2006; 444:203–207.
2. Foerster O. Contributions to the pathophysiology of the visual pathway and visual area. J Psychol Neurol. 1929; 39:463–485.
3. Krieg WJ. Functional neuroanatomy. 2nd ed. New York: Blakiston;1953. p. 207–208.
4. Rizzo JF 3rd, Wyatt J, Loewenstein J, et al. Perceptual efficacy of electrical stimulation of human retina with a microelectrode array during short-term surgical trials. Invest Ophthalmol Vis Sci. 2003; 44:5362–5369.
5. Zrenner E. Will retinal implants restore vision? Science. 2002; 295:1022–1025.
6. Qu J, Wang D, Grosskreutz CL. Mechanisms of retinal ganglion cell injury and defense in glaucoma. Exp Eye Res. 2010; 91:48–53.
7. Schiefer U, Hart W. Functional anatomy of the human visual pathway. In : Schiefer U, Hart W, Wilhelm H, editors. Clinical neuro-ophthalmology: a practical guide. Berlin: Springer;2007. p. 19–28.
8. Krause M, Fogel W, Heck A, et al. Deep brain stimulation for the treatment of Parkinson's disease: subthalamic nucleus versus globus pallidus internus. J Neurol Neurosurg Psychiatry. 2001; 70:464–470.
9. Gloesmann M, Hermann B, Schubert C, et al. Histologic correlation of pig retina radial stratification with ultrahigh-resolution optical coherence tomography. Invest Ophthalmol Vis Sci. 2003; 44:1696–1703.
10. Kara P, Pezaris JS, Yurgenson S, Reid RC. The spatial receptive field of thalamic inputs to single cortical simple cells revealed by the interaction of visual and electrical stimulation. Proc Natl Acad Sci U S A. 2002; 99:16261–16266.
11. Pezaris JS, Reid RC. Demonstration of artificial visual percepts generated through thalamic microstimulation. Proc Natl Acad Sci U S A. 2007; 104:7670–7675.
12. Pezaris JS, Reid RC. Simulations of electrode placement for a thalamic visual prosthesis. IEEE Trans Biomed Eng. 2009; 56:172–178.
13. Cohen ED. Prosthetic interfaces with the visual system: biological issues. J Neural Eng. 2007; 4:R14–R31.
14. Logothetis NK, Pauls J, Augath M, et al. Neurophysiological investigation of the basis of the fMRI signal. Nature. 2001; 412:150–157.
15. Schneider KA, Richter MC, Kastner S. Retinotopic organization and functional subdivisions of the human lateral geniculate nucleus: a high-resolution functional magnetic resonance imaging study. J Neurosci. 2004; 24:8975–8985.
16. Okada Y, Lahteenmaki A, Xu C. Comparison of MEG and EEG on the basis of somatic evoked responses elicited by stimulation of the snout in the juvenile swine. Clin Neurophysiol. 1999; 110:214–229.
17. Laube T, Schanze T, Brockmann C, et al. Chronically implanted epidural electrodes in Gottinger minipigs allow function tests of epiretinal implants. Graefes Arch Clin Exp Ophthalmol. 2003; 241:1013–1019.
18. Fang M, Li J, Rudd JA, et al. fMRI mapping of cortical centers following visual stimulation in postnatal pigs of different ages. Life Sci. 2006; 78:1197–1201.
19. Gizewski ER, Schanze T, Bolle I, et al. Visualization of the visual cortex in minipigs using fMRI. Res Vet Sci. 2007; 82:281–286.
20. Jelsing J, Hay-Schmidt A, Dyrby T, et al. The prefrontal cortex in the Gottingen minipig brain defined by neural projection criteria and cytoarchitecture. Brain Res Bull. 2006; 70:322–336.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download