Abstract
Purpose
To introduce autologous tragal perichondrium transplantation as a novel surgical modality for the management of intractable symptomatic bullous keratopathy.
Methods
In three eyes of three patients with painful bullous keratopathy, autologous tragal perichondria were transplanted on the corneal surface with the human amniotic membrane transplanted above. We included an additional three eyes of three patients with painful bullous keratopathy who received amniotic membrane transplantation only to serve as controls. Clinical symptom outcomes were assessed using a visual analogue scale at postsurgical months 1, 3, 5, 7, and 9. In addition, transplanted tragal perichondrium and amniotic membrane complex tissue button obtained from one patient who underwent penetrating keratoplasty was evaluated by immunohistochemical analysis of CD34, vimentin, and alcian blue staining.
Results
All three patients who underwent autologous tragal perichondrium and human amniotic membrane co-transplantation showed improvements in pain and tearing. However, all three patients in the control group experienced aggravation of tearing and no further improvement of pain 3 months after surgery. In addition, one patient in the control group developed premature degradation of the amniotic membrane. Histopathologic and immunohistochemical analysis showed intact surface epithelization and positive CD34, vimentin and alcian blue staining of transplanted tragal perichondria.
Conclusions
The tragal perichondrium has a high mechanical structural force and high potency due to well-organized epithelization and the presence of mesenchymal stem cells. Autologous tragal perichondrium transplantation may be an effective modality for the management of painful bullous keratopathy.
Bullous keratopathy, the leading cause for corneal transplantation, is a painful end-stage corneal disorder of numerous ocular conditions caused by endothelial decompensation. Normally the corneal endothelium regulates stromal hydration through its ion pumping function, but in conditions such as aging, trauma, intraocular lens implantation, corneal graft failure, endothelial dystrophy, absolute glaucoma, chronic contact lens wear, or decrease of endothelial cell density and pumping function, the corneal stroma becomes edematous with bullae formation [1]. Clinical features of bullous keratopathy can vary from severe vision loss caused by stromal edema and subepithelial scarring to symptoms such as pain, foreign body sensation, photophobia, and epiphora due to the formation and rupture of blisters [2,3].
Currently, penetrating keratoplasty (PKP) is the definitive treatment for bullous keratopathy [4-6]. However, patients usually have to wait many years before undergoing surgery due to a shortage of donor corneas, especially in Asian countries. Thus, in practice, the primary aim of treatment is to relieve its associated symptoms.
Several modalities are being applied for the management of pain in symptomatic bullous keratopathy including medical (e.g., hyperosmotic agents, lubricants, topical corticosteroids, and therapeutic soft contact lens) and surgical (e.g., diffuse anterior stromal puncture, human amniotic membrane transplantation, excimer laser phototherapeutic keratectomy [PTK], diamond burr polishing of Bowman's membrane, and conjunctival flap) [2,3,7-11]. However, these treatments are not always satisfactory or acceptable. The effects of applied drugs are transient and imperfect and the use of steroids may predispose patients to bacterial keratitis [12]. The surgical procedures are also unsatisfactory because of the regression of epithelial bullae, the recurrence of pain, and discomfort [2,10,13].
The tragal perichondrium is used widely for reconstructive ear surgery (i.e., myringoplasty) in the otorhinolaryngology field and is also used in eyelid reconstruction [14-16]. Originally, the tragal perichondrium differentiates from mesenchymal tissue and due to its stem cell potency [17,18], it has high tissue compatibility yielding excellent epithelization after transplantation [15]. In this study, we transplanted autologous tragal perichondria on the cornea in conjunction with human amniotic membrane transplantation in patients with painful bullous keratopathy who were awaiting PKP. Although this study included only a small number of cases, we achieved successful postoperative graft viability and compatibility, and associated symptoms such as pain and tearing improved after transplantation.
Amniotic membrane transplantation (AMT) has been shown to be a safe, effective, and long-lasting treatment modality for intractable pain associated with chronic bullous keratopathy with poor visual potential [13]. However, there are several reported complications after AMT, including hematoma formation in the postoperative period, granuloma formation, or premature degradation of the membrane requiring a repeat procedure [19]. For this reason, we compared the effect of autologous tragal perichondrium (aTPC) and amniotic membrane (AM) co-transplantation with the effect of AMT alone in patients with bullous keratopathy. The aim of our study was to evaluate the therapeutic efficacy of aTPC as a bridge until PKP.
Autologous tragal perichondrium transplantation was performed on three eyes of three patients with chronic intractable painful bullous keratopathy with symptoms of tearing for over 2 years and who were awaiting PKP. Patient demographics and clinical data are given in Table 1. Given the opacity of the perichondrium, the surgery was confined to patients with low visual acuity or potential. The institutional review board for Human Studies of Chung-Ang University Hospital approved this research and protocols adhered to the Declaration of Helsinki. Informed consent was obtained from all patients after explaining the nature of the study and its possible consequences.
Gauze soaked in 2% lidocaine with 1:100,000 epinephrine was packed in the external auricular canal for 5 minutes for anesthetization and decongestion of the external auricular skin. After disinfecting the periauricular area, 0.2 mL of a local anesthetic agent was injected at the posterior surface of the tragus to be harvested. After 5 minutes, a 1.2-cm-long incision was made 2 mm posterior to the free edge of tragus with a no. 10 scalpel blade (Fig. 1A). After the skin and subcutaneous tissues were incised parallel to the free edge of the tragus, the tragal perichondrium and cartilage were immediately seen under the skin. Using a pair of small blunt curved scissors, the subcutaneous tissue was dissected and the anterior surface of the tragal perichondrium was dissected from the tragal cartilage (Fig. 1B). After the entire anterior perichondrium was maximally elevated and cleaned on both sides, the perichondrium to be cut was gently held by fine tissue forceps (Fig. 1C). Thereafter, the tragal perichondrium was harvested from the tragal tissue and the skin incision was sutured with 4-0 nylon material. The free tragal perichondrium tissues were washed with normal saline and prepared for grafting (Fig. 1D). Harvested autologous tragal perichondria were processed to be round in shape by trephination with a 9-mm diameter corneal trephine. After applying retrobulbar anesthesia, a central 9 mm of the cornea was treated with 30% alcohol, and the relevant corneal epithelium was removed by manual debridement with a cotton tip and a Weck-Cell sponge. The prepared 9-mm-diameter tragal perichondrium was sutured to the peripheral cornea using simple interrupted suturing technique with 10-0 nylon, skin side up. Because the tragal perichondrium is a whitish-opaque tissue, we performed corneal tattooing using a 1:1 mixture of black and brown pigments (Micropigment; BioTouch, La Puente, CA, USA) for cosmetic purposes. Permanent AM transplantation with basement membrane side down and a successive temporary AM patch procedure with the basement membrane side up were then performed over the attached perichondrial tissue using a suturing technique with 10-0 nylon. In all control group patients, corneal epithelial peeling, permanent AM transplantation, and temporary AM patching were performed in the same way as in the perichondrium transplantation group. After the surgery, all patients in both groups were instructed to use a topical levofloxacin eye drop (Cravit; Santen, Osaka, Japan) 4 times a day, a steroid-antibiotic mixture ointment (Maxitrol; Alcon Laboratories, Fort Worth, TX, USA) 2 times a day for 2 weeks followed by a tapered dose over the following 2 weeks, a topical 40% autoserum made in our hospital, and artificial tear eye drops without preservatives (Hyalein mini 0.1%, Santen) every 2 hours for 4 weeks. The attached AM patch was removed 1 to 2 weeks after surgery.
This study had two major outcomes with respect to the effect of tragal perichondrium transplantation: 1) post-transplant alleviation of pre-existing pain and tearing; and 2) post-operative compatibility and viability of graft tissue. The most common and intractable symptoms, pain and tearing, were assessed at postsurgical months 1, 3, 5, 7, and 9 and were compared with pre-procedure values. Symptom severity was estimated with a questionnaire that rated patient symptoms with a visual analogue scale (VAS), with 0 representing no pain or discomfort and 10 representing unbearable symptoms. All six patients were asked about the postoperative degree of symptoms by the same interviewer and were instructed to indicate the degree on the VAS.
One (case 3) patient in the aTPC-AMT group underwent PKP 5 months after surgery. The excised transplanted perichondrium-amniotic membrane complex from this patient was detached from the underlying cornea button, fixed in 10% formaldehyde, bisected, and subjected to routine paraffin processing. Hematoxylin and eosin staining, alcian blue staining, and immunohistochemical staining of vimentin and CD34 were performed to determine the postoperative structural stability of the cornea and to evaluate transplanted aTPC as a compatible, proliferative graft tissue.
The follow-up period after aTPC-AMT or AMT was 9 months for all patients. In addition, one male patient in the aTPC-AMT group underwent PKP 5 months after aTPC-AMT. All patients experienced pain and tearing relief as a result of graft epithelium healing 2 to 3 weeks after surgery. Fig. 2 shows the symptom scale score of each patient in the both groups. Preoperatively, all patients in the aTPC-AMT group had uncontrollable pain and tearing, despite long-term use of hyperosmotic agents, lubricants, and topical corticosteroids. In contrast, during the postoperative period, the pain and tearing were relieved within the first month. Thereafter, this improvement continued until postsurgical month 9 (Fig. 2A and 2B). However, in the control group, although all patients experienced mild relief of their symptoms within the first postoperative month, there was an increase in pain at the 3-month follow-up in two patients (cases 4 and 6) and pain persisted at the 9-month follow-up. The remaining patient in this group (case 5) showed further improvement at the third month, but symptoms deteriorated thereafter (Fig. 2C). In all patients of this group, tearing was aggravated after mild improvement (Fig. 2D).
Comparing graft tissue viability and compatibility, adverse effects were not observed in either group on corneal examination up to 1 month postoperatively. The transplanted aTPC-AM complex was maintained well without damage up to 9 months postoperatively (the maximum follow-up period) (Fig. 3). However, one male patient in the control group on whom a permanent AMT and temporary AM patch procedure was performed had developed premature degradation of the amniotic membrane and central corneal ulceration at the 6-week follow-up (Fig. 4). After 1 month of conservative treatment, the central corneal ulceration improved, but the extent of degradation of the amniotic membrane remained the same on slit lamp examination.
No signs of tissue rejection, necrosis, or infection were noted in the aTPC-AMT group up to 5 months postoperatively. One of these patients underwent PKP 5 months after transplantation. In this patient, visual acuity was improved from hand movements close to the face to 20 / 200. In addition, there was no complaint of pain or tearing, and no graft rejection up to 9 months post-PKP, with maintenance of the transparency of the donor cornea (Fig. 5).
Histologic and immunohistochemical analyses were performed on tissues from one patient (case 3) in the aTPC-AMT group who underwent PKP 5 months after the primary surgery. Sections of the excised transplanted aTPC-AM complex obtained at the time of PKP revealed complete multi-layered epithelization over the transplanted aTPC and AM, which was morphologically very similar to stratified squamous epithelium of a normal cornea (Fig. 6).
Immunohistochemical staining demonstrated that the transplanted aTPC had a proliferative and regenerative potential as a graft tissue. Alcian blue staining was positive and vimentin was expressed throughout the inner chondrogenic layer (Fig. 7). Immunohistochemical staining of CD34, which is expressed in vascular endothelial cells, showed vessel formation in fibrous connective tissue in the outer region of the chondrogenic layer (Fig. 8).
Symptomatic bullous keratopathy, which occurs as a result of endothelial decompensation, is one of the major indications for PKP [4-6]. However, PKP is a treatment option that is often unavailable because of the insufficient number of donor corneas and the high cost of the procedure. Therefore, in cases with poor visual potential or low visual acuity, the focus is on life-enhancing symptom alleviation.
Several management methods are used to control the symptoms of patients who are awaiting PKP. However, these are not totally satisfactory because of therapeutic limitations or possible side effects. Contact lens usage and topical steroid application may predispose a patient to bacterial keratitis. Surgically, anterior stromal micropuncture (ASP), PTK, and AMT, which are well recognized and commonly used procedures to manage painful bullous keratopathy, can cause several side effects. Fernandes et al. [20] reported exaggerated subepithelial fibrosis after ASP that led to symptom recurrence. In addition, possible complications such as persistent epithelial defect, delayed epithelial healing, and microbial keratitis have been reported after PTK [21], and AMT can result in complications associated with premature membrane degradation [2,9,10,13]. In our study, one patient in the AMT group experienced degradation of the transplanted AM just over the area of the ulcerated cornea 6 weeks after AMT.
Although the tragal perichondrium is generally used in myringoplasty and eyelid reconstruction [14-16], there is no report on the efficacy of tragal perichondrium grafting on the cornea. The perichondrium is composed of collagen type I and II fibers, elastic fibers, and glycoproteins including aggrecan [22] which are similar to the composition of sclera. And autologous tragal perichondrium graft was reported to be an effective treatment for replacing the necrotized sclera via neovascularization and rapid epithelization in refractory necrotizing scleritis [23]. In addition, Migirov et al. [24] showed that closure of the tympanic membrane using the tragal perichondrium achieved water tolerance in all cases. That is, due to the mechano-structural force and scant water penetration [24] due to the good epithelization over the transplanted perichondrium (Fig. 6), the perichondrium graft could act as a substrate compatible with the underlying corneal stroma.
When choosing an ideal graft tissue, many factors must be taken into account, including availability of the material, immune reactions due to allogenic antigens, secondary infections, foreign body reactions, and inflammation. The most commonly used patch grafts such as the sclera, dura, pericardium and fascia lata in repairing of scleral defects have some limitations [25-32]. Scleral or pericardial tissue grafts may induce immune rejection leading to graft failure. Fascia lata, dura or dermis grafts are not widely used because of harvesting and processing difficulties. However, the tragal perichondrium is easily obtained without deformity or dysfunction of the donor site and is free from immune rejection. Furthermore, the tragal perichondrium is known to contain stem cells, which may assist the viability of grafted tissues [17,18]. Ljung et al. [33] reported structural, functional, and proliferative characteristics of regenerating cartilage, in addition to vivid vessel regeneration, in a rabbit perichondrial graft tissue model. Arai et al. [17] showed that mesenchymal stem cells in the perichondrium of the hyaline cartilage of fetal limbs are pluripotent. Togo et al. [18] differentiated perichondrocytes harvested from rabbit ears into adipocytes and osteocytes and reported the efficacy of perichondria containing such tissue progenitor cells as a potential candidate tissue for reconstructing cartilage. In this study, based on two unique characteristics; mechanical structural force and stem cell-bearing feature, we co-transplanted autologous tragal perichondria and human amniotic membranes on the corneal surfaces of patients with painful bullous keratopathy. All patients who underwent aTPC-AMT experienced improvements in pain and tearing. Moreover, aTPC was maintained without degradation for 5 months postoperatively.
In bullous keratopathy, painful episodes associated with photophobia and tearing are attributed to nerve stretching and irritation by epithelial and subepithelial bullae and to the rupture of surface bullae exposing nerve endings [34]. Therefore, symptom alleviation can be achieved by the removal of the bullae and by preventing the corneal nerve endings from touching the eyelid margin.
We were able to examine the aTPC-AM complex button removed at the time of PKP in one patient and correlate this patient's clinical symptom improvements with histologic observations. In this case, we observed a well-organized multilayered surface epithelium over the transplanted aTPC-AM complex. AMT is generally considered an effective treatment for ocular surface reconstruction. However, in a study by Tosi et al. [35], five eyes that underwent AMT (epithelial side up) and subsequent PKP or enucleation were subjected to histopathological analysis after a mean of 8.2 months (range, 2 to 22 months). None of the eyes showed remnants of AM in the cornea and all showed disruption of Bowman's membrane despite ocular symptom improvement.
We suspect that the good epithelization observed in the aTPC-AMT group relieved the pain and tearing by functioning as a key barrier that prevents the nerve endings on the cornea from being exposed to the external environment. The high expression of CD34 on immunohistochemical analysis indicated vessel regeneration in the transplanted aTPC and the positive vimentin and alcian blue staining in the inner chondrogenic layer suggests the existence of viable mesenchymal stem cell-like perichondrocytes. Based on the CD34-positive staining of cells in the inner chondrogenic layer, we suggest that the inner layer, which contains mesenchymal stem cells, may also play a role in the origin of angiogenesis (Fig. 8). The results of the histologic and immunohistochemical analyses in our study implies that aTPC could act as a feeder layer promoting overlying epithelization and subsequent formation of a multilayered intact epithelium. Rapid and well-organized epithelization in graft tissues is a unique feature of the tragal perichondrium and seems to be attributable to the presence of endothelial and mesenchymal stem cells.
Our study is limited in that the number of patients in each group was small and the follow-up period was short. Also, we cannot exclude the possible impact of undiscovered factors in postoperative tissue healing. Nevertheless, this is the first study that has utilized aTPC in corneal disease, specifically bullous keratopathy. Our results suggest that aTPC encourages intact epithelial regeneration and symptom alleviation because it contains stem cells.
Autologous tragal perichondrium transplantation represents a novel treatment method for symptom control in patients with intractable painful bullous keratopathy who are awaiting PKP. For extensive and standardized use of this treatment modality, further studies of the mechanism underlying the therapeutic effect of tragal perichondrium are required based on additional standardized prospective trials.
Figures and Tables
Fig. 1
Harvest of autologous tragal perichondrium. (A) Skin incision parallel to the free edge of the tragus. (B) Anterior surface (arrow) of the tragal perichondrium dissected from tragal cartilage. (C) Tragal perichondrium (arrow) to be cut held with forceps. (D) Tagal perichondrium harvested intraoperatively.
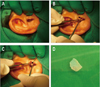
Fig. 2
Visual analogue scales of symptoms (pain and tearing) before and after treatment. (A,B) Symptom improvement continued until postsurgical 9 months in autologous tragal perichondrium + amniotic membrane transplantation group. (C,D) However, in the control group (amniotic membrane transplantation alone), symptom was aggravated after mild improvement in post-surgical early period.

Fig. 3
Slit lamp photograph of the cornea with bullous keratopathy before (A) and after autologous tragal perichondrium transplantation, followed by corneal tattooing and amniotic membrane transplantation (B). (B) There was an area of epithelial defect (arrow) 2 weeks after surgery. (C,D) After surgery, transplanted autologous tragal perichondrium was well maintained without degradation 5 months after surgery (C), and up to follow-up month 9.
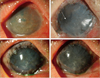
Fig. 4
Slit lamp photograph of the cornea 6 weeks after amniotic membrane transplantation. (A) Transplanted amniotic membrane degraded in one patient who underwent only amniotic membrane transplantation. (B) Central cornea was ulcerated with fluorescein dye staining.
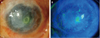
Fig. 5
Clinical appearance before and after autologous tragal perichondrium transplantation in conjunction with corneal tattooing and amniotic membrane transplantation, thereafter penetrating keratoplasty (PKP) in one patient (case 3). (A) Preoperatively, there was central corneal opacity with severe corneal neovascularization. (B) Until post-PKP month 9, there was no graft rejection, maintaining transparency.

Fig. 6
Histology of postoperative epithelization underlying transplanted amniotic membrane and autologous tragal perichondrium (×400). Multi-layered generated stratified squamous epithelium was morphologically similar to normal corneal epithelium.
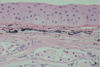
Fig. 7
Histology and immunohistochemistry of the transplanted autologous tragal perichondrium. (A) Inner chondrogenic layer of transplanted autologous tragal perichondrium (inside of black rectangle) with hematoxylin and eosin staining (×200). (B) Immunohistochemical staining of vimentin (arrow) and (C) alcian blue staining (arrow head) of the inner chondrogenic layer showed existence of mesenchymal stem cells in transplanted autologous tragal perichondrium (×400).
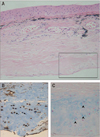
Fig. 8
Immunohistochemical staining of CD34 of the transplanted autologous tragal perichondrium. (A) Formation of vascular structures are shown (arrow) in transplanted autologous tragal perichondrium (×200), and (B) this is supposed to be attributable to CD34-positive endothelial cells (arrow head) in the inner chondrogenic layer (inside of black rectangle in A) (×400).

Acknowledgements
The Basic Science Research Program of the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (2012-0005222) supported this research.
References
1. Joyce NC. Cell cycle status in human corneal endothelium. Exp Eye Res. 2005. 81:629–638.
2. Pires RT, Tseng SC, Prabhasawat P, et al. Amniotic membrane transplantation for symptomatic bullous keratopathy. Arch Ophthalmol. 1999. 117:1291–1297.
3. Gomes JA, Haraguchi DK, Zambrano DU, et al. Anterior stromal puncture in the treatment of bullous keratopathy: six-month follow-up. Cornea. 2001. 20:570–572.
4. Brady SE, Rapuano CJ, Arentsen JJ, et al. Clinical indications for and procedures associated with penetrating keratoplasty, 1983-1988. Am J Ophthalmol. 1989. 108:118–122.
5. Cursiefen C, Kuchle M, Naumann GO. Changing indications for penetrating keratoplasty: histopathology of 1,250 corneal buttons. Cornea. 1998. 17:468–470.
6. Mamalis N, Anderson CW, Kreisler KR, et al. Changing trends in the indications for penetrating keratoplasty. Arch Ophthalmol. 1992. 110:1409–1411.
7. Alino AM, Perry HD, Kanellopoulos AJ, et al. Conjunctival flaps. Ophthalmology. 1998. 105:1120–1123.
8. Lamberts DW. Topical hyperosmotic agents and secretory stimulants. Int Ophthalmol Clin. 1980. 20:163–169.
9. Mejía LF, Santamaria JP, Acosta C. Symptomatic management of postoperative bullous keratopathy with nonpreserved human amniotic membrane. Cornea. 2002. 21:342–345.
10. Sridhar MS, Vemuganti GK, Bansal AK, Rao GN. Anterior stromal puncture in bullous keratopathy: a clinicopathologic study. Cornea. 2001. 20:573–579.
11. Thomann U, Meier-Gibbons F, Schipper I. Phototherapeutic keratectomy for bullous keratopathy. Br J Ophthalmol. 1995. 79:335–338.
12. Kent HD, Cohen EJ, Laibson PR, Arentsen JJ. Microbial keratitis and corneal ulceration associated with therapeutic soft contact lenses. CLAO J. 1990. 16:49–52.
13. Espana EM, Grueterich M, Sandoval H, et al. Amniotic membrane transplantation for bullous keratopathy in eyes with poor visual potential. J Cataract Refract Surg. 2003. 29:279–284.
14. Cavaliere M, Mottola G, Rondinelli M, Iemma M. Tragal cartilage in tympanoplasty: anatomic and functional results in 306 cases. Acta Otorhinolaryngol Ital. 2009. 29:27–32.
15. Nigro MV, Friedhofer H, Natalino RJ, Ferreira MC. Comparative analysis of the inf luence of perichondrium on conjunctival epithelialization on conchal cartilage grafts in eyelid reconstruction: experimental study in rabbits. Plast Reconstr Surg. 2009. 123:55–63.
16. Yotsuyanagi T, Urushidate S, Watanabe M, Sawada Y. Reconstruction of a three-dimensional structure using cartilage regenerated from the perichondrium of rabbits. Plast Reconstr Surg. 1999. 103:1120–1123.
17. Arai F, Ohneda O, Miyamoto T, et al. Mesenchymal stem cells in perichondrium express activated leukocyte cell adhesion molecule and participate in bone marrow formation. J Exp Med. 2002. 195:1549–1563.
18. Togo T, Utani A, Naitoh M, et al. Identification of cartilage progenitor cells in the adult ear perichondrium: utilization for cartilage reconstruction. Lab Invest. 2006. 86:445–457.
19. Dua HS, Gomes JA, King AJ, Maharajan VS. The amniotic membrane in ophthalmology. Surv Ophthalmol. 2004. 49:51–77.
20. Fernandes M, Moreker MR, Shah SG, Vemuganti GK. Exaggerated subepithelial fibrosis after anterior stromal puncture presenting as a membrane. Cornea. 2011. 30:660–663.
21. Maini R, Sullivan L, Snibson GR, et al. A comparison of different depth ablations in the treatment of painful bullous keratopathy with phototherapeutic keratectomy. Br J Ophthalmol. 2001. 85:912–915.
22. Madsen K, von der Mark K, van Menxel M, Friberg U. Analysis of collagen types synthesized by rabbit ear cartilage chondrocytes in vivo and in vitro. Biochem J. 1984. 221:189–196.
23. Koo H, Jeong JH, Chun YS, Kim JC. Ocular reconstruction using autologous tragal perichondrium for a refractory necrotizing scleral perforation: a case report. J Korean Ophthalmol Soc. 2011. 52:1227–1231.
24. Migirov L, Shapira Y, Horowitz Z, Wolf M. Exclusive endoscopic ear surgery for acquired cholesteatoma: preliminary results. Otol Neurotol. 2011. 32:433–436.
25. Ahn BH, Lee EJ, Sung KH. The use of a temporal muscle fascia in the treatment of scleral defect. J Korean Ophthalmol Soc. 1983. 24:785–791.
26. Kwak JY, Chang HK. Autogenous temporalis fascia grafting and conjunctival flap transposition in scleromalacia after pterygium excision. J Korean Ophthalmol Soc. 2004. 45:180–186.
27. Kim JH. Scleral grafting on necrotic scleritis following pterygium excision. J Korean Ophthalmol Soc. 1982. 23:29–36.
28. Lee CO, Jong SH, Lee JJ. Autologous simple conjunctival graft and conjunctiva/tenon graft on focal scleromalacia. J Korean Ophthalmol Soc. 1997. 38:1737–1741.
29. Na YS, Joo MJ, Kim JH. Results of scleral allografting on scleral necrosis following pterygium excision. J Korean Ophthalmol Soc. 2005. 46:402–409.
30. Mauriello JA Jr, Fiore PM, Pokorny KS, Cinotti DJ. Use of split-thickness dermal graft in the surgical treatment of corneal and scleral defects. Am J Ophthalmol. 1988. 105:244–247.
31. Smith MF, Doyle JW, Ticrney JW Jr. A comparison of glaucoma drainage implant tube coverage. J Glaucoma. 2002. 11:143–147.
32. Pinto LM, Regatieri CV, Tavares IM, Rigueiro MP. Porcine pericardium as glaucoma implant tube coverage: an experimental study. Arq Bras Oftalmol. 2008. 71:623–628.
33. Ljung A, Ohlsen L, Widenfalk B, Gerdin B. Characterisation of cells in regenerating cartilage from autotransplanted perichondrium: immunohistochemical expression of smooth-muscle actin, desmin, vimentin, and Ki-67. Scand J Plast Reconstr Surg Hand Surg. 1999. 33:257–266.
34. Borboli S, Colby K. Mechanisms of disease: Fuchs' endothelial dystrophy. Ophthalmol Clin North Am. 2002. 15:17–25.
35. Tosi GM, Traversi C, Schuerfeld K, et al. Amniotic membrane graft: histopathological findings in five cases. J Cell Physiol. 2005. 202:852–857.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download