Abstract
Purpose
To investigate the role of focal adhesion kinase (FAK) in transforming growth factor (TGF)-β-induced myofibroblast transdifferentiation of human Tenon's fibroblasts.
Methods
Primary cultured human Tenon's fibroblasts were exposed to TGF-β1 for up to 48 hours. The mRNA levels of FAK, α smooth muscle actin (αSMA), and β-actin were determined by quantitative real time reverse transcription polymerase chain reaction. The protein levels of collagen type I, FAK, phospho-FAK, αSMA, and β-actin were determined by Western immunoblots. After the small interfering RNA targeting FAK (siRNAFAK) molecules were delivered into the cells, the expressions of αSMA proteins were determined by Western immunoblots.
Myofibroblast transdifferentiation of activated fibroblasts is an essential step of the fibrotic process in most living tissues. Myofibroblasts have characteristics that are taken from both smooth muscle cells and fibroblasts; they express the contractile phenotype of α smooth muscle actin (αSMA) and synthesize extracellular matrix proteins including collagens [1-3]. In subconjunctival fibrosis, the myofibroblast transdifferentiation of Tenon's fibroblasts is known to be mainly caused by transforming growth factor (TGF)-β [4-6]. Many ophthalmic researchers have sought to identify an anti-fibrotic mechanism by inhibiting TGF-β signaling [7-10].
Focal adhesion kinase (FAK), a cytosolic protein tyrosine kinase, provides scaffolding functions at sites of integrin adhesion and promotes cell migration [11,12]. The kinase modulates several basic processes in normal development and cancer metastasis. In addition, it also plays a crucial role in the transdifferentiation of fibroblasts to myofibroblasts induced by TGF-β [13-15]. However, because the precise action of the kinase is somewhat variable based on the tissue [14,15], the role of FAK in myofibroblast transdifferentiation in human Tenon's fibroblasts must be confirmed.
Using primary cultured human Tenon's fibroblasts, the role of FAK in TGF-β-induced myofibroblast transdifferentiation of human Tenon's fibroblasts was investigated in the present study.
After the Institutional Review Board approved our protocol, selected patients received comprehensive information and provided written consent for inclusion. In compliance with the tenets of the Declaration of Helsinki, small human Tenon's capsule specimens were obtained during strabismus surgery, and primary human Tenon's fibroblasts were cultured from the explants. Cells were incubated in Dulbecco's modified Eagle's medium (DMEM; Life Technologies, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (Life Technologies), 100 units/mL penicillin, and 100 µg/mL streptomycin (Life Technologies) at 37℃ in 5% CO2.
After 12 hours of incubation in serum-free media, the cells were treated with 5 ng/mL of recombinant human TGF-β1 (R&D Systems Inc., Minneapolis, MN, USA) for up to 48 hours. For the control group, the same volume of DMEM was added to the media instead of TGF-β1.
Small interfering RNA molecules targeting FAK mRNA (siRNAFAK) were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA) and were delivered into cells according to the manufacturer's instructions. Briefly, the fibroblasts were incubated in media containing 50 nM of siRNAFAK and 2 µg/mL of Lipofectamine 2000 (Life Technologies) for 16 hours at 37℃ in a CO2 incubator.
Total RNA was extracted using an RNeasy Mini Kit (Qiagen, Venlo, Netherlands), and cDNAs were synthesized using the SuperScript III First-Strand Synthesis System (Life Technologies). Real-time polymerase chain reaction (PCR) was performed with 50 ng cDNA per reaction using 25 µL of iQ SYBR Green Supermix (Bio-Rad Laboratories, Hercules, CA, USA) containing 500 nM of specific primers (Table 1) in the iCycler iQ Real-Time PCR Detection System (Bio-Rad Laboratories). The SYBER green data were analyzed with a relative standard curve of β-actin.
Whole cellular proteins were extracted from the cells, and equal amounts of protein (10 µg) were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis. The proteins were transferred to polyvinylidene fluoride membranes and probed overnight with primary antibodies against human collagen type I, FAK, phospho-FAK (pFAK), αSMA, and β-actin (Santa Cruz Biotechnology). Immunoreactive bands were detected with horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology) and visualized with an enhanced chemiluminescent system.
Quantitative reverse transcription (RT)-PCR data is expressed as mean ± SEM and compared with the Kruskal-Wallis one-way analysis of variance using the MedCalc ver. 11.4.2 (MedCalc Software bvba, Mariakerke, Belgium). Image analysis of the immunobands was performed using the ImageJ ver. 1.43u (National Institutes of Health, Bethesda, MD, USA). A p-value less than 0.05 was considered statistically significant.
The quantitative data of RT-PCR for FAK and αSMA are presented in Fig. 1. In the human Tenon's fibroblasts, the 16-hour treatment with TGF-β1 significantly increased the mRNA levels of αSMA (5.11 ± 1.37 times to control, p = 0.023) but not of FAK (1.16 ± 0.06 times to control, p = 0.204).
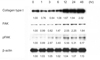
On the Western immunoblots, the TGF-β1 treatment increased the protein expression of FAK as well as that of collagen type I. Expression of pFAK, an active form of FAK, was also increased. These changes were observed after 12 hours. Representative immunobands for collagen type I, FAK, pFAK, and β-actin are shown in Fig. 2.
Regarding αSMA, though serum starvation itself slightly induced the expression of αSMA, the exposure to TGF-β1 for 48 hours stimulated αSMA expression in human Tenon's fibroblasts. When the siRNAFAK molecules were introduced into the cells, the TGF-β1-induced αSMA expression was attenuated. Representative immunobands for αSMA in the control group, TGF-β1 only treatment group, and siRNAFAK/TGF-β1 treatment group are shown in Fig. 3.
FAK, also known as protein tyrosine kinase 2, participates in the focal adhesion between the cytoskeleton and extracellular matrix and elicits intracellular signal transductions associated with cell migration and survival [11,12,16-18]. It is activated by autophosphorylation via Smad- and p38 MAPK-dependent mechanisms [19-22] and plays a key role in cancer metastasis as well as normal development [11,12]. Even though FAK is also linked to the myofibroblast transdifferentiation of fibroblasts in response to TGF-β1 [13-15], the precise role of the kinase has not been well studied in the fibrotic process in the subconjunctival space. Excessive subconjunctival fibrosis causes ocular morbidity in not only patients with ocularcutaneous disorders, but also patients who have undergone strabismus surgery and/or glaucoma-filtering surgery. A better understanding and modulation of FAK signaling in this process may result in a novel therapeutic strategy for those patients.
Using primary cultured human Tenon's fibroblasts, we investigated the role of FAK in TGF-β-induced myofibroblast transdifferentiation in the present study and found that silencing of FAK action using siRNAFAK duplex significantly attenuated the αSMA expression induced by TGF-β1 in human Tenon's fibroblasts. Our data implies that FAK may participate in the myofibroblast transdifferentiation of those cells.
Myofibroblasts contain characteristics found in smooth muscle cells and fibroblasts in that they possess a contractile phenotype of αSMA and synthesize extracellular matrix proteins [1-3]. Since they play a crucial role in most fibrotic responses, myofibroblast transdifferentiation from activated fibroblasts is an essential step in the fibrotic process. In the subconjunctival space, TGF-β typically initiates this myofibroblast transdifferentiation of fibroblasts [4-6].
On quantitative RT-PCR, the mRNA expression of αSMA was dramatically increased by TGF-β1, but FAK was not significantly altered. However, on Western immunoblots, the protein levels of both FAK and pFAK were minimally stimulated by TGF-β1 treatment. The increase in FAK seems to result from an increase in translation rather than transcription.
Currently, many researchers have tried to develop anti-TGF-β neutralizing antibodies to modulate postoperative scarring in patients undergoing ocular surface surgery [23-25]. FAK, as a participant in TGF-β-associated intracellular signaling of myofibroblast transdifferentiation from fibroblasts, might also be valuable as a novel therapeutic strategy for anti-fibrosis.
Figures and Tables
Fig. 1
Quantitative reverse transcription polymerase chain reaction for focal adhesion kinase (FAK) and α smooth muscle actin (αSMA) after exposure to 5 ng/mL of transforming growth factor-β1 for up to 16 hours. The level of target mRNA was calculated using a relative standard curve of β-actin and expressed as the mean ± SEM. *p < 0.05.

Fig. 2
Representative Western immunoblots and densitometric data for collagen type I (190 kDa), focal adhesion kinase (FAK, 125 kDa), phospho-FAK (pFAK, 125 kDa), and β-actin (43 kDa) in human Tenon's fibroblasts stimulated by 5 ng/mL of transforming growth factor-β1 for up to 48 hours.
Acknowledgements
This work was supported by a Faculty Research Grant from the Yonsei University College of Medicine, Seoul, Republic of Korea (6-2008-0274).
References
1. Eyden B. The myofibroblast: an assessment of controversial issues and a definition useful in diagnosis and research. Ultrastruct Pathol. 2001. 25:39–50.
2. Eyden B. Electron microscopy in the study of myofibroblastic lesions. Semin Diagn Pathol. 2003. 20:13–24.
3. Desmouliere A, Guyot C, Gabbiani G. The stroma reaction myofibroblast: a key player in the control of tumor cell behavior. Int J Dev Biol. 2004. 48:509–517.
4. Meyer-Ter-Vehn T, Grehn F, Schlunck G. Localization of TGF-beta type II receptor and ED-A fibronectin in normal conjunctiva and failed filtering blebs. Mol Vis. 2008. 14:136–141.
5. Saika S, Yamanaka O, Okada Y, et al. TGF beta in fibroproliferative diseases in the eye. Front Biosci (Schol Ed). 2009. 1:376–390.
6. Seong GJ, Hong S, Jung SA, et al. TGF-beta-induced interleukin-6 participates in transdifferentiation of human Tenon’s fibroblasts to myofibroblasts. Mol Vis. 2009. 15:2123–2128.
7. Nakamura H, Siddiqui SS, Shen X, et al. RNA interference targeting transforming growth factor-beta type II receptor suppresses ocular inflammation and fibrosis. Mol Vis. 2004. 10:703–711.
8. Meyer-Ter-Vehn T, Gebhardt S, Sebald W, et al. p38 inhibitors prevent TGF-beta-induced myofibroblast transdifferentiation in human tenon fibroblasts. Invest Ophthalmol Vis Sci. 2006. 47:1500–1509.
9. Yamanaka O, Saika S, Ikeda K, et al. Interleukin-7 modulates extracellular matrix production and TGF-beta signaling in cultured human subconjunctival fibroblasts. Curr Eye Res. 2006. 31:491–499.
10. Meyer-Ter-Vehn T, Katzenberger B, Han H, et al. Lovastatin inhibits TGF-beta-induced myofibroblast transdifferentiation in human tenon fibroblasts. Invest Ophthalmol Vis Sci. 2008. 49:3955–3960.
11. Chatzizacharias NA, Kouraklis GP, Theocharis SE. The role of focal adhesion kinase in early development. Histol Histopathol. 2010. 25:1039–1055.
12. Schwock J, Dhani N, Hedley DW. Targeting focal adhesion kinase signaling in tumor growth and metastasis. Expert Opin Ther Targets. 2010. 14:77–94.
13. Thannickal VJ, Lee DY, White ES, et al. Myofibroblast differentiation by transforming growth factor-beta1 is dependent on cell adhesion and integrin signaling via focal adhesion kinase. J Biol Chem. 2003. 278:12384–12389.
14. Mimura Y, Ihn H, Jinnin M, et al. Constitutive phosphorylation of focal adhesion kinase is involved in the myofibroblast differentiation of scleroderma fibroblasts. J Invest Dermatol. 2005. 124:886–892.
15. Brenmoehl J, Miller SN, Hofmann C, et al. Transforming growth factor-beta 1 induces intestinal myofibroblast differentiation and modulates their migration. World J Gastroenterol. 2009. 15:1431–1442.
16. McLean GW, Carragher NO, Avizienyte E, et al. The role of focal-adhesion kinase in cancer: a new therapeutic opportunity. Nat Rev Cancer. 2005. 5:505–515.
17. Cox BD, Natarajan M, Stettner MR, Gladson CL. New concepts regarding focal adhesion kinase promotion of cell migration and proliferation. J Cell Biochem. 2006. 99:35–52.
18. Liu S, Xu SW, Kennedy L, et al. FAK is required for TGF-beta-induced JNK phosphorylation in fibroblasts: implications for acquisition of a matrix-remodeling phenotype. Mol Biol Cell. 2007. 18:2169–2178.
19. Horowitz JC, Rogers DS, Sharma V, et al. Combinatorial activation of FAK and AKT by transforming growth factor-beta1 confers an anoikis-resistant phenotype to myofibroblasts. Cell Signal. 2007. 19:761–771.
20. Parsons CJ, Takashima M, Rippe RA. Molecular mechanisms of hepatic fibrogenesis. J Gastroenterol Hepatol. 2007. 22:Suppl 1. S79–S84.
21. Walsh MF, Ampasala DR, Hatfield J, et al. Transforming growth factor-beta stimulates intestinal epithelial focal adhesion kinase synthesis via Smad- and p38-dependent mechanisms. Am J Pathol. 2008. 173:385–399.
22. Walsh MF, Ampasala DR, Rishi AK, Basson MD. TGF-beta1 modulates focal adhesion kinase expression in rat intestinal epithelial IEC-6 cells via stimulatory and inhibitory Smad binding elements. Biochim Biophys Acta. 2009. 1789:88–98.
23. Cordeiro MF. Technology evaluation: lerdelimumab, Cambridge Antibody Technology. Curr Opin Mol Ther. 2003. 5:199–203.
24. Mead AL, Wong TT, Cordeiro MF, et al. Evaluation of anti-TGF-beta2 antibody as a new postoperative anti-scarring agent in glaucoma surgery. Invest Ophthalmol Vis Sci. 2003. 44:3394–3401.
25. CAT-152 0102 Trabeculectomy Study Group. Khaw P, Grehn F, et al. A phase III study of subconjunctival human anti-transforming growth factor beta(2) monoclonal antibody (CAT-152) to prevent scarring after first-time trabeculectomy. Ophthalmology. 2007. 114:1822–1830.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download