Abstract
Purpose
The effects of amiloride on cellular toxicity caused by tissue plasminogen activator (tPA) in mouse primary retinal cells were investigated.
Methods
Primary retinal cell cultures were maintained using glial conditioned medium. Commercial tPA and L-arginine were added, and the level of cyclic guanosine monophosphate (cyclic-GMP) in the culture supernatant was assessed using an ELISA assay. We measured the cell viability of cultured retinal cells pretreated with three different concentrations of amiloride (1, 10, and 100 µm) in addition to commercial tPA or L-arginine treatment.
Results
After exposing the cultured mouse retinal cells to tPA plus L-arginine or L-arginine alone, cyclic-GMP concentrations were 61.9 ± 5.1 pmole/mL and 63.1 ± 6.1 pmole/mL, respectively. However, the control group had a significantly lower concentration of cyclic-GMP (37.2 ± 3.4 pmole/mL, p < 0.01). The cyclic GMP-dissolved solution did not cause retinal cell death. In the control group and the group treated with 1 µm amiloride and tPA containing L-arginine, the cell viability was 43.7% and 44.5%, respectively. However, cell viability increased to 70.6% with 10 µm amiloride and 78.4% with 100 µm amiloride (p = 0.015).
Tissue plasminogen activator (tPA), an effective thrombolytic agent, is commercially available for the treatment of vitreous hemorrhage, subfoveal hemorrhage, suprachoroidal hemorrhage, and retinal vascular occlusive disease in the field of ophthalmology [1,2]. In particular, intravitreal or subretinal injections of tPA have been used when performing surgery for submacular hemorrhages or subfoveal choroidal neovascularization [3-8]. Dose-dependent retinal toxicity has been reported with the injection of intravitreal tPA; this toxicity has been reported to be due to the L-arginine vehicle rather than the tPA itself [9]. The retinal toxicity of L-arginine has been reported to increase nitric oxide (NO) in cells [10]. NO activates guanylate cyclase in the photoreceptor layer of the retina to produce more cyclic guanosine monophosphate (cyclic-GMP) [11]. In the rd/rd mouse, photoreceptor degeneration is due to a mutation of the rod-specific enzyme cyclic-GMP phosphodiesterase, resulting in permanently open cyclic-GMP-gated cation channels in the rod outer segment membrane, allowing Na+ and Ca2+ ions to enter the cell and leading to possibly toxic levels of Ca2+ [12]. Therefore, the increase in NO that is caused by L-arginine leads to the accumulation of cyclic-GMP, thus leading to toxicity.
Amiloride has an inhibitory function on the Na+/H+ exchanger in the apical plasma membrane of the distal nephron and the cyclic-GMP gated channel of the outer membrane of retinal photoreceptor cells [13]. Therefore, inhibition of the cyclic-GMP gated channels of retinal cells may result in a decrease in intracellular Ca2+, which then leads to increased retinal cell survival after tPA-induced toxicity.
The purpose of this study was to examine the effect of amiloride on cellular toxicity caused by tPA-arginine in mouse primary retinal cells.
ICR mice were purchased from Samtako Co. (Osan, Korea). The care, use, and treatment of all animals in this study were in strict agreement with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. The recombinant human tPA preparation used in this study was commercially obtained from Boehringer Ingelheim (Ridgefield, CT, USA) and had an average activity of 580 IU/µg. The reconstituted preparation had a recombinant tPA concentration of 1 mg/mL. The vehicle consisted of L-arginine (35 mg/mL or 0.2 mol/L), phosphoric acid (10 mg/L, 0.1 mmol/L), and polysorbate 80 (≤110 µg/mL, 83 µmol/mL). Small aliquots of recombinant tPA were maintained at -70℃ until just prior to use when they were warmed to room temperature and diluted in sterile balanced salt solution to concentrations of 25, 125, or 250 µg/mL. Sodium bicarbonate, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid, glutamine, and minimum essential medium (MEM) were purchased from Gibco BRL (Grand Island, NY, USA). The fetal bovine serum was purchased from Hyclone Co. (Logan, UT, USA). L-Arginine, nicotinamide adenine dinucleotide phosphate, and poly-D-lysine were purchased from Sigma Co. (St. Louis, MO, USA).
The retinas from two-day-old mouse litters were dissected from other ocular tissues and digested for 20 minutes at 37℃ with 0.1% trypsin in Hanks' balanced salt solution. The cells were suspended in MEM supplemented with 10% heat-inactivated fetal bovine serum and then seeded in poly-D-lysine-coated 24-well plates at a density of 12 retinas per plate. The cells were maintained at 37℃ in a humidified incubator with 95% air and 5% CO2. The glial conditioned medium (GCM) solution was replaced with a 60% medium 24 hours after plating the retina cells. The GCM solution was prepared as previously described [10]. The retinal cells were used after ten days in culture.
Based on the form of the nucleus and the loss of the plasma membrane, apoptotic cells can be differentiated from non-apoptotic cells using propidium iodide and Hoechst 33258, as described previously [13]. Highly charged propidium iodide cannot enter an intact cell, whereas Hoechst 33258 can pass through both intact and dead cell membranes. The Hoechst stain solution was added to the growth media at 2 to 5 µg/mL and then allowed to incubate for 30 minutes at 37℃. The propidium iodide solution was then added to the media at the same concentration as the Hoechst stain. Cell damage was observed by fluorescent microscopy (Olympus microscope, Tokyo, Japan).
To examine whether cyclic-GMP caused cell death, the lactate dehydrogenase (LDH) secreted into the culture media was measured by a spectrophotometer [14]. The total LDH release, which corresponds to complete neuronal cell death, was assessed at the end of each experiment after freezing the culture at -70℃ for two hours with a subsequent rapid thaw. The relative LDH activity was calculated as a percentage of the total LDH release as follows: relative LDH activity = ([sample LDH - basal LDH] / [total LDH - basal LDH]) × 100 (%). All data are presented as mean ± SE and statistically analyzed by a one-way ANOVA and the Student-Newman-Keul's test using SigmaStat software (Jandel Scientific, San Rafael, CA, USA).
The levels of cyclic-GMP in the culture supernatants were assessed by ELISA (Medicorp Inc., Montreal, QB, Canada). The supernatants were collected at 0, 12, or 24 hours after adding 10 µg/400 µL of tPA (with L-arginine) and 5 mM L-arginine to the primary retinal cell culture. One hundred microliters of culture supernatant was measured using the ELISA kit (sensitivity of 1 pmole/mL) and read in an automated plate reader at a wavelength of 405 nm.
Cell death in cultures treated with various concentrations of cyclic-GMP was measured by LDH assay. At 24 hours after adding cyclic-GMP in concentrations of 0, 0.01, 0.05, 0.1, 0.5, or 1.0 mM, the levels of cell death in the primary cultured retinal cells were assessed. Statistically, all data was analyzed by a one-way ANOVA and the Student-Newman-Keul's test using SigmaStat software (Jandel Scientific).
To examine the possible mechanism involved in tPA plus L-arginine-induced retinal cell toxicity, we measured cell viability using the LDH assay of cultured retinal cells pretreated with various concentrations of amiloride followed by tPA plus L-arginine (10 µg/400 µL) or L-arginine alone (5 mM). The data represent the mean ± SD of independent experiments and were statistically analyzed by a one-way ANOVA and the Student-Newman-Keul's test using SigmaStat software (Jandel Scientific).
To examine retinal toxicity, primary retinal cell cultures were treated with L-arginine. Hoechst 33258 and propidium iodide staining revealed that L-arginine treatment induced nuclear condensation and fragmentation in the primary retinal cells. Cells with blue fragmented nuclei were early apoptotic cells, and cells with pink fragmented nuclei were late apoptotic cells (Fig. 1A).
As we previously demonstrated, the LDH release assay showed that the treatment of retinal cells with 10 ug/400 µL of tPA (containing L-arginine) and 5 mM L-arginine resulted in a significantly larger number of dead cells after 24 hours compared to untreated control cells and tPA treatment alone. LDH release of control, tPA, tPA with L-arginine, and L-arginine were as follows: 19.7%, 20.4%, 49.7%, and 40.1%, respectively (Fig. 1B).
Twenty-four hours after treatment with 10 ug/400 µL of tPA plus L-arginine and 5 mM L-arginine alone, the cyclic-GMP concentrations in the primary retinal cell cultures were 61.9 ± 5.1 pmole/mL and 63.1 ± 6.1 pmole/mL, respectively, revealing significant differences from the control (37.2 ± 3.4 pmole/mL, p < 0.01).
LDH was measured 24 hours after treatment with cyclic-GMP dissolved in PBS at concentrations of 0.01, 0.05, 0.1, 0.5, or 1 mM, after which each was treated in primary cultured retinal cells to verify whether the treatment caused cell death (Fig. 1A). The LDH measures showed that cyclic-GMP added to this solution did not cause cell death (Fig. 2B).
Amiloride was used to identify the link between increased cyclic-GMP and cell death. In the control group and group treated with 1 µm amiloride with tPA containing L-arginine, the cell viabilities were 43.7% and 44.5% respectively. However, cell viability increased to 70.6% with 10 µm amiloride to and 78.4% with 100 µm amiloride (p = 0.015). In the case of the L-arginine treated group, cell survival increased significantly with 10 µm amiloride compared to that of the control group (81.7% vs. 63.4%) (Fig. 3).
We previously reported that the arginine vehicle used in commercial tPA formulations is responsible for the observed retinal toxicity reactions and the excessive NO involved in retinal cell death [10]. L-Arginine is the main source of the synthesis of NO in many cell types [15]. NO is generated from the terminal guanidine nitrogen of L-arginine by nitric oxide synthetase. The generated NO stimulates soluble guanylate cyclase, resulting in an increase in cyclic-GMP [11]. This mechanism has been found to modulate voltage-gated ion channels in rod photoreceptors [16].
Retinal cell death was demonstrated by LDH assay, and the product, which includes L-arginine (tPA with L-arginine and L-arginine alone), revealed higher LDH release than that of the control and tPA. These results are similar to a previous study [10].
To further explore the possible mechanisms involved in tPA plus L-arginine-induced cell death, we analyzed cyclic-GMP production in the retinal cell cultures treated with tPA plus L-arginine or L-arginine alone. Exposure of cultured mouse retinal cells to tPA plus L-arginine or L-arginine alone led to marked increases in cyclic-GMP.
Amiloride may have several effects on intracellular cyclic-GMP accumulation. Amiloride is a well known Na+/H+ exchanger [17], a major transporter responsible for the regulation of intracellular pH. When the Na+/H+ exchanger is activated, intracellular Na+ is increased, which will then stimulate the Na+/Ca2+ exchanger. These changes increase intracellular Ca2+. Thus, if amiloride inhibits the Na+/H+ exchanger, the influx of Ca2+ may be decreased; this outcome is one of the mechanisms involved in protecting against cell death under L-arginine exposure.
Furthermore, amiloride is responsible for blocking the cyclic GMP-activated conductance of rod photoreceptors [13]. When amiloride blocks the Na+/H+ exchanger, the compound also causes intracellular pH changes. These changes in intracellular pH result in changes in the level of cyclic-GMP due to inhibition of the cyclic-GMP channel, leading to decreased tPA plus L-arginine- or L-arginine alone-induced cell death for unknown reasons. These findings indicate that the opening of cyclic-GMP channels might play a role in tPA (containing L-arginine)-induced retinal cell death. The selective rod functional deficits and degeneration may be caused by the following cellular mechanisms: inhibition of retinal cyclic-GMP phosphodiesterase causing an increase in cyclic-GMP, an inhibition of intermediary metabolism, and/or an alteration in calcium metabolism [18]. These mechanisms are very similar to the L-arginine-induced cell death mechanism. However, we could not confirm selective rod cell death, so further study is necessary to confirm the mechanism in this cell culture system.
In summary, our data suggests that L-arginine increases intracellular cyclic-GMP and may give rise to retinal cells through this mechanism. In addition, amiloride in concentrations greater than 10 µm protected against L-arginine-induced retinal cell death.
Figures and Tables
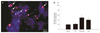 | Fig. 1(A) Fluorescence micrograph (Hoechst-PI stain) of cultured retinal cells treated with L-arginine (L-arg). L-arg-induced cell death showed nuclear fragmentation, the typical apoptotic nuclear pattern. Cells with blue fragmented nuclei are early apoptotic cells, and cells with pink fragmented nuclei are late apoptotic cells (arrows). (B) The graph shows the level of cell death in primary cultured retinal cells at 24 hours after adding tissue plasminogen activator (tPA) alone, tPA plus L-arg, and L-arg alone. Cell death was assessed by lactate dehydrogenase (LDH) assay. Significant cell death occurred at 24 hours after treatment with tPA plus L-arg and L-arg alone. The data represent the means ± SD of the three independent experiments and were statistically analyzed by a one-way ANOVA and the Student-Newman-Keul's test using SigmaStat software. *p < 0.01 compared with control. |
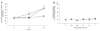 | Fig. 2(A) Cyclic guanosine monophosphate (cyclic-GMP) formation after exposure to tissue plasminogen activator (tPA) plus L-arginine (L-arg) and L-arg alone. The graph shows the cyclic-GMP concentration (pmole/mL) in primary cultured retinal cell media at 0, 12, or 24 hours after adding 10 µg/400 µL tPA (containing L-arg) and 5.0 mM L-arg. Cyclic-GMP concentration was assessed by ELISA assay. The concentration of cyclic-GMP was significantly elevated at 12 hours after treatment with 5.0 mM L-arg and at 24 hours after adding 10 µg/400 µL tPA plus L-arg and 5.0 mM L-arg. *p < 0.05 and **p < 0.01 compared with same time control groups (closed circle; control group, open circle; tPA-arg and reverse triangle; L-arg). (B) Measurement of cell death in cultures treated with various concentrations of cyclic-GMP. The graph shows the level of cell death in primary cultured retina cells at 24 hours after adding cyclic-GMP in various concentrations. Cell death was assessed by lactate dehydrogenase (LDH) assay. Concentrations of 0, 0.01, 0.05, 0.1, 0.5, or 1.0 mM cyclic-GMP were added, respectively. Added cyclic-GMP did not induce cell death. All data are presented as mean ± SE and statistically analyzed by a one-way ANOVA and the Student-Newman-Keul's test using SigmaStat software. conc = concentration. |
 | Fig. 3Measurement of cell viability of cultured retinal cells pretreated with various concentrations of the cyclic nucleotide-gated channel blocker, amiloride, followed by tissue plasminogen activator (tPA) plus L-arginine (L-arg, 10 µg/400 µL) or L-arg alone (5 mM). The data represent the mean ± SD of independent experiments and were statistically analyzed by one-way ANOVA and the Student-Newman-Keul's test using SigmaStat software. *p < 0.05 compared with control (first column, no treatment; second column, 1 µm amiloride; third column, 10 µm amiloride; and fourth column, 100 µm amiloride). LHD = lactate dehydrogenase. |
References
1. Pennica D, Holmes WE, Kohr WJ, et al. Cloning and expression of human tissue-type plasminogen activator cDNA in E. coli. Nature. 1983. 301:214–221.
2. Collen D, Stassen JM, Marafino BJ Jr, et al. Biological properties of human tissue-type plasminogen activator obtained by expression of recombinant DNA in mammalian cells. J Pharmacol Exp Ther. 1984. 231:146–152.
3. Hassan AS, Johnson MW, Schneiderman TE, et al. Management of submacular hemorrhage with intravitreous tissue plasminogen activator injection and pneumatic displacement. Ophthalmology. 1999. 106:1900–1906.
4. Hattenbach LO, Klais C, Koch FH, Gumbel HO. Intravitreous injection of tissue plasminogen activator and gas in the treatment of submacular hemorrhage under various conditions. Ophthalmology. 2001. 108:1485–1492.
5. Peyman GA, Nelson NC Jr, Alturki W, et al. Tissue plasminogen activating factor assisted removal of subretinal hemorrhage. Ophthalmic Surg. 1991. 22:575–582.
6. Kamei M, Tano Y, Maeno T, et al. Surgical removal of submacular hemorrhage using tissue plasminogen activator and perfluorocarbon liquid. Am J Ophthalmol. 1996. 121:267–275.
7. Lewis H, VanderBrug Medendorp S. Tissue plasminogen activator-assisted surgical excision of subfoveal choroidal neovascularization in age-related macular degeneration: a randomized, double-masked trial. Ophthalmology. 1997. 104:1847–1851.
8. Haupert CL, McCuen BW 2nd, Jaffe GJ, et al. Pars plana vitrectomy, subretinal injection of tissue plasminogen activator, and fluid-gas exchange for displacement of thick submacular hemorrhage in age-related macular degeneration. Am J Ophthalmol. 2001. 131:208–215.
9. Johnson MW, Olsen KR, Hernandez E, et al. Retinal toxicity of recombinant tissue plasminogen activator in the rabbit. Arch Ophthalmol. 1990. 108:259–263.
10. Oh HS, Kwon OW, Chung I, et al. Retinal toxicity of commercial tissue plasminogen activator is mediated by the induction of nitric oxide in the mouse retinal primary cells. Curr Eye Res. 2005. 30:291–297.
11. Moncada S, Palmer RM. Biosynthesis and actions of nitric oxide. Semin Perinatol. 1991. 15:16–19.
12. Rohrer B, Pinto FR, Hulse KE, et al. Multidestructive pathways triggered in photoreceptor cell death of the rd mouse as determined through gene expression profiling. J Biol Chem. 2004. 279:41903–41910.
13. McLatchie LM, Matthews HR. The effect of pH on the block by L-cis-diltiazem and amiloride of the cyclic GMP-activated conductance of salamander rods. Proc Biol Sci. 1994. 255:231–236.
14. Ho AK, Lalh SS, Young I, et al. Inhibitory effects of amilorides on pinealocyte adenosine 3',5'-monophosphate and guanosine 3',5'-monophosphate accumulation: possible involvement of postreceptor mechanisms. Endocrinology. 1990. 127:460–466.
15. Shimizu S, Eguchi Y, Kamiike W, et al. Induction of apoptosis as well as necrosis by hypoxia and predominant prevention of apoptosis by Bcl-2 and Bcl-XL. Cancer Res. 1996. 56:2161–2166.
16. Koh JY, Choi DW. Quantitative determination of glutamate mediated cortical neuronal injury in cell culture by lactate dehydrogenase efflux assay. J Neurosci Methods. 1987. 20:83–90.
17. Moncada S, Palmer RM, Higgs EA. Biosynthesis of nitric oxide from L-arginine: a pathway for the regulation of cell function and communication. Biochem Pharmacol. 1989. 38:1709–1715.
18. Kurenny DE, Moroz LL, Turner RW, et al. Modulation of ion channels in rod photoreceptors by nitric oxide. Neuron. 1994. 13:315–324.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download