Abstract
We report the first case of ocular benign lymphoid hyperplasia (BLH) treated with subconjunctival injection of bevacizumab (Avastin). A 27-year-old man presented to our clinic with conjunctival masses and limbal neovascularization. An incisional biopsy yielded the diagnosis of BLH. The patient was subsequently given a subconjunctival injection of bevacizumab (1.25 mg / 0.1 mL). The patient did not experience recurrence or malignant metaplasia during the one-year follow-up period. In patients with conjunctival BLH, subconjunctival injection of bevacizumab can be a useful treatment option in patients unable to undergo a surgical procedure due to limbal neovascularization.
Ocular adnexal lymphoproliferative lesions are lymphohistologic masses that can appear in various locations, including the conjunctiva, orbit, eyelid, lacrimal duct, and lacrimal gland. They can be both primary or secondary and encompass a wide disease spectrum ranging from benign lymphoid hyperplasia (BLH) to malignant lymphoma. Conjunctival lymphoproliferative lesions have the best prognosis among ocular lymphoproliferative lesions; the majority are diagnosed as BLH. It has been reported that more than 90% of such lesions do not go on to develop into systemic lymphoma [1]. There is no definitive recommended treatment for conjunctival BLH, but reported treatments include cryotherapy and the combination of surgical excision and oral steroid administration [2,3]. There have also been reports of using local radiotherapy to prevent malignant proliferation and systemic invasion [4]. However, cryotherapy or surgical excision carry the risk of cosmetic problems due to scar formation. Furthermore, localized radiotherapy, cryotherapy, or surgical excision would be difficult to perform in lesions with limbal neovascularization. As such, we report the first case of conjunctival BLH treated with bevacizumab injection.
A 27-year-old man with a two year history of bilateral medial conjunctival masses was referred to our hospital. His chief complaint was injection of both eyes. His best corrected visual acuity was 20/20 and the intraocular pressures in both eyes were within normal limits. Color vision testing, automated visual fields, fundus examination, and extraocular muscle function were normal bilaterally. There were no palpable masses or edema in the eyelids and there was no exophthalmos. Slit lamp examination revealed a protruding hypervascular mass with combined medial limbal neovascularization on each medial conjunctival surface. Specifically, examination revealed a salmon colored, elevated (2 mm), moderately firm patch (6 mm × 5 mm) on the nasal conjunctiva of the right eye with neovascularization. A faint, salmon colored, elevated (1 mm) mass (5 mm × 4 mm) on the nasal conjunctiva of the left eye, with thinner new vessels than in the right eye, was also observed (Fig. 1A and 1B).
Incisional biopsy was performed on the mass in the right eye for definitive diagnosis. Pathological examination revealed benign lymphohistiocytic infiltrates (Fig. 2). The lymphoid reaction showed T cells (CD3+) and B cells (CD20+) with no evidence of atypical malignant cells. The patient subsequently underwent a complete physical exam, including serology (thyroid function tests) and radiology (chest radiography and abdominal unltrasonography), to rule out systemic disease. There were no significant findings.
Given the combination of BLH with hypervascular masses and medial limbal neovascularization, bevacizumab (2.5 mg / 0.1 mL) (Avastin; Genentech, South San Francisco, CA, USA) was injected into both medial subconjunctival spaces. Two months after injection, both masses had almost completely disappeared and the accompanying neovascularization was reduced (Fig. 1D and 1E). The patient did not experience recurrence or any other complications during the one-year follow-up period.
Appropriate treatment for ocular BLH remains controversial. Many physicians simply recommend observation. The reported treatments for orbital BLH include surgical excision, radiation therapy, systemic corticosteroid treatment, chemotherapy, and cryotherapy. Chemotherapy can be used for lymphoma in the conjunctiva if it coexists with systemic lymphoma. One report found the final remission rate to be 98% for cryotherapy used to treat lymphoma localized in the conjunctiva [5]. Additionally, local excision or oral or topical corticosteroids can also be considered as treatment options. However, in the present case, localized radiotherapy, cryotherapy, or surgical excision would have been inappropriate treatment options because the lesion was accompanied by limbal neovascularization and surgical excision carries the risk of postoperative cosmetic defects. As such, bevacizumab injection was administered under the assumption that the conjunctival BLH, which is often difficult to treat using existing treatment methods, could be treated by altering the ocular surface blood vessels and lymphatic vessel formation.
Bevacizumab is an anti-vascular endothelial growth factor (VEGF)-A antibody approved to treat metastatic colorectal cancer [6]. Recently, it has been reported that anti-VEGF antibody may be effective in treating age-related macular degeneration and diabetic retinopathy [7,8]. Bevacizumab has recently been studied as a treatment option for neovascularization in ocular surface diseases [9-11]. However, it is unclear if bevacizumab inhibits lymphangiogenesis. Only a handful of experimental approaches deal with the inhibition of lymphangiogenesis (e.g., the use of VEGFR1R2-Trap [12] or a blocking anti-VEGFR3-antibody [13]). Recently, several studies have reported that VEGF-A not only mediates hemangiogenesis, but also lymphangiogenesis [14-16]. Given this, bevacizumab has been reported to be an inhibitor of angiogenesis and lymphangiogenesis on the ocular surface [17]. According to this report, bevacizumab also inhibits the proliferation of lymphatic endothelial cells. However, there has not yet been a report of conjunctival BLH treated with bevacizumab.
In this case, we hypothesized that with the inhibition of new blood and lymphatic vessel formation due to bevacizumab injection, the influx of polyclonal lymphocytes would be blocked and the polyclonal lymphocytes that had already accumulated would be drained by the existing lymphatic vessels. We subsequently observed a dramatic response to the local subconjunctival injection of bevacizumab. The bevacizumab near the reactive follicle must have been sufficient to suppress the inflow of lymphocytes. In the treatment of BLH, a common lymphoma in the conjunctiva, subconjunctival injection of bevacizumab can be a useful treatment option for patients who cannot tolerate a surgical procedure because of limbal neovascularization.
Figures and Tables
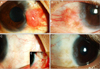 | Fig. 1(A) Slit lamp photograph of the patient's right eye on initial presentation. Note the size, salmon color, and elevated appearance of the hypervascular lesion. (B) Slit lamp photograph of the patient's left eye on initial presentation. Note the size, faint salmon color, and mildly protruding mass. (C,D) Slit lamp photograph of both eyes 2 months following subconjunctival injection of bevacizumab. The lesions appear to be almost completely resolved with no obvious remnants seen on the sclera or conjunctiva. (E) Slit lamp photograph of the patient's right eye after biopsy. |
References
1. Jo YJ, Yim JH, Park KS. MALT (mucosa associated lymphoid tissue) type lymphoma of the ocular adnexa. J Korean Ophthalmol Soc. 2002. 43:357–362.
2. Yu SB, Lew HL, Yun YS. A case of conjunctival benign lymphoid hyperplasia associated with ptosis of the eyelid in a child. J Korean Ophthalmol Soc. 2005. 46:560–564.
3. Chang WS, Park JB, Kim KH, Kim SY. Two cases of conjunctival mucosa-associated lymphoid tissue (MALT) lymphomas treated by cryotherapy. J Korean Ophthalmol Soc. 2008. 49:1165–1172.
4. Bolek TW, Moyses HM, Marcus RB Jr, et al. Radiotherapy in the management of orbital lymphoma. Int J Radiat Oncol Biol Phys. 1999. 44:31–36.
5. Eichler MD, Fraunfelder FT. Cryotherapy for conjunctival lymphoid tumors. Am J Ophthalmol. 1994. 118:463–467.
6. Ferrara N, Hillan KJ, Novotny W. Bevacizumab (Avastin), a humanized anti-VEGF monoclonal antibody for cancer therapy. Biochem Biophys Res Commun. 2005. 333:328–335.
7. Lazic R, Gabric N. Intravitreally administered bevacizumab (Avastin) in minimally classic and occult choroidal neovascularization secondary to age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2007. 245:68–73.
8. Jorge R, Costa RA, Calucci D, et al. Intravitreal bevacizumab (Avastin) for persistent new vessels in diabetic retinopathy (IBEPE study). Retina. 2006. 26:1006–1013.
9. Manzano RP, Peyman GA, Khan P, et al. Inhibition of experimental corneal neovascularisation by bevacizumab (Avastin). Br J Ophthalmol. 2007. 91:804–807.
10. DeStafeno JJ, Kim T. Topical bevacizumab therapy for corneal neovascularization. Arch Ophthalmol. 2007. 125:834–836.
11. Hosseini H, Nejabat M. A potential therapeutic strategy for inhibition of corneal neovascularization with new anti-VEGF agents. Med Hypotheses. 2007. 68:799–801.
12. Cursiefen C, Cao J, Chen L, et al. Inhibition of hemangiogenesis and lymphangiogenesis after normal-risk corneal transplantation by neutralizing VEGF promotes graft survival. Invest Ophthalmol Vis Sci. 2004. 45:2666–2673.
13. Cursiefen C, Ikeda S, Nishina PM, et al. Spontaneous corneal hem- and lymphangiogenesis in mice with destrin-mutation depend on VEGFR3 signaling. Am J Pathol. 2005. 166:1367–1377.
14. Björndahl MA, Cao R, Burton JB, et al. Vascular endothelial growth factor-a promotes peritumoral lymphangiogenesis and lymphatic metastasis. Cancer Res. 2005. 65:9261–9268.
15. Hong YK, Lange-Asschenfeldt B, Velasco P, et al. VEGF-A promotes tissue repair-associated lymphatic vessel formation via VEGFR-2 and the alpha1beta1 and alpha2beta1 integrins. FASEB J. 2004. 18:1111–1113.
16. Nagy JA, Vasile E, Feng D, et al. Vascular permeability factor/vascular endothelial growth factor induces lymphangiogenesis as well as angiogenesis. J Exp Med. 2002. 196:1497–1506.
17. Bock F, Onderka J, Dietrich T, et al. Bevacizumab as a potent inhibitor of inflammatory corneal angiogenesis and lymphangiogenesis. Invest Ophthalmol Vis Sci. 2007. 48:2545–2552.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download