Abstract
Purpose
To evaluate the characteristics of fluid accumulation in the uveitic stage of Vogt-Koyanagi-Harada (VKH) disease using high resolution optical coherence tomography (OCT3).
Methods
Twenty-eight eyes in 14 patients with VKH disease were reviewed retrospectively. These 28 eyes were divided into 19 eyes with intraretinal fluid (C group) and 9 eyes without intraretinal fluid (N group). Changes in visual acuity and fluid accumulation observed using OCT were compared between the two groups.
Results
Visual acuity at the time of presentation was significantly worse in the C group than in the N group (p=0.005). The photoreceptor layer appeared to be double-layered due to a cystoid space in the C group. Layered structures and strands found in the cystoid space. Expanding sponge-form edema led to the development of a cystoid space in the photoreceptor layer. Intraretinal fluid resolved earlier than subretinal fluid. There were no observed differences in visual acuity between the two groups after four days of treatment.
Vogt-Koyanagi-Harada (VKH) disease is a panuveitis involving both eyes in patients with no history of penetrating ocular lesions. There is often involvement of other organs including the ears, skin, and meninges.1 The acute phase of VKH disease is characterized by diffuse choroidal inflammation and serous retinal detachment in the posterior pole.2
Round yellowish subretinal structures have recently been described in patients with VKH. Application of a new diagnostic technique, optical coherence tomography (OCT), has revealed intraretinal fluid accumulation in 40% of the eyes afflicted with VKH disease.3,4 This intraretinal fluid accumulates in the outer retina, leaving a well-preserved inner retina,4 in contrast to the cystoid macular edema that is seen in other types of uveitis such as intermediate uveitis.5 Thin structures that may represent subretinal fibrosis or fibrin membranesare found within the cystoid space.6,7
Previous studies using OCT1 or OCT2, which have lower resolution than the more recently developed OCT3, suffered from limitations of imaging and visualization. We examined cystoid spaces typical of VKH using OCT3 and investigated the clinical characteristics related to these findings.
We reviewed the medical records of 14 consecutive patients who were diagnosed with acute VKH disease based on systemic symptoms, ocular findings, and fluorescein angiography (FA) at Pusan National University Hospital from 2003 to 2006, following the revised diagnostic criteria proposed by the First International Workshop on Vogt-Koyanagi-Harada disease.1
All 14 patients were treated with intravenous methylprednisolone for three to seven days, and then treated with oral steroids for three to four months. Supplemental regional and/or topical steroids were prescribed at the physicians' discretion. The clinical data recorded included best corrected visual acuity, biomicroscopic exam, fundus findings, FA, and results of OCT scans.
OCT scans were taken with a high resolution OCT3 scanner (StratusOCT®, Carl Zeiss, USA) every day during intravenous steroid therapy, and then at one to two week intervals until subretinal fluid accumulation resolved. Vertical and horizontal scans of 5 mm or 9 mm through the fovea were obtained based on the extent of serous detachment. Skilled technicians administered follow-up scans of the same orientation and size as the scans at the presentation.
The 28 eyes we examined were divided into two groups based on the findings of the OCT scans. In the C group there was a cystoid space regardless of the presence of subretinal fluid. In the N group there was only subretinal fluid and no cystoid space. A cystoid space was defined as a hypo-reflective space in the outer retina. Cystoid spaces exhibited an outer wall that was identified on OCT scans as a hyper-reflective line over the retinal pigment epithelium (RPE)-choriocapillaris complex (Fig. 1).
Snellen acuity was converted to a corresponding logMAR acuity. Changes in visual acuity, subretinal fluid accumulation, and cystoid space were analyzed and compared between the groups. Statistical analysis was performed with T-test using SPSS 12.0.
Twenty-eight eyes were examined in 14 patients (eight male, six female). The average age was 34.8±12.6 years (range, 16-53). Three patients were confirmed to have complete VKH disease. Eight had incomplete VKH disease and three had probable VKH disease. None of the patients had anterior uveitis severe enough to affect visual acuity.
The C group included 19 eyes (67.9%). 18 of which simultaneously exhibited a cystoid space and subretinal fluid accumulation. The N group included 9 eyes (32.1%, Table 1). Eleven patients had both of their eyes categorized in the same group. Three patients had the right eye in the N group and the left eye in the C group.
The baseline characteristics of the patients are summarized in Table 1. Visual acuity at presentation was significantly worse in the C group compared to the N group (p=0.005).
Although the findings of a cystoid space observed using OCT varied, several common features were identified. First, cystoid spaces that were located subfoveally were related to poor visual acuity; visual acuity remained good if the cystoid space did not involve the fovea (Fig. 1A). Second, the cystoid spaces observed in these patients lacked septa which are commonly observed in cystoid macular edema caused by other diseases such as intermediate uveitis or diabetic retinopathy (Fig. 1 A to D). Third, the cystoid spaces in some eyes had inner structures that were layered or had the appearance of vertical strands (Fig. 1 C and D). No similar structures were found in the subretinal fluid.
In many cases, the outer wall (RPE side) of the cystoid space continued to the photoreceptor layer (Fig. 1 A and C). The photoreceptor layer appeared to be separated or double-layered, and exhibited a sponge-like edema adjacent to the cystoid space (Fig. 1 A and B).
One eye showed early development of the cystoid space (Fig. 2). Sponge-like edema of the photoreceptor layer was seen in this eye. We observed a layer that was thought to be a junction between the inner and the outer segments of the photoreceptor. The next day, just before the initiation of steroid treatment, the edema of the outer segments expanded to form a cystoid space. The edema of the inner segments and the subretinal fluid appeared to be compressed by the expansion of the cystoid space.
After initiation of the steroid treatment, the cystoid space resolved before the subretinal fluid. In 12 eyes the cystoid space became confluent with the subretinal space with degradation of the outer boundary (Fig. 3). In three eyes layered structures were temporarily observed during the process of resolution (Fig. 4).
Differences in visual acuity between the two groups were found no later than four days after steroid treatments began. After three months, the median visual acuity in both groups was 20/20 (Fig. 5).
Serous retinal detachment or subretinal fluid accumulation is regarded as a typical feature of diffuse choroiditis in acute VKH disease. The presence of an intraretinal cystoid space observed on OCT was recently described by Maruyama.4 The incidence of intraretinal fluid was reported at about 40%. This finding was supported by the results of another report in which round yellowish structures were found in 43% of cases of acute VKH disease.6 OCT scans obtained in six of those eyes showed fluid accumulation in the retina.
We observed a cystoid space in over two-thirds (67.9%) of the eyes examined using OCT3, a higher incidence than observed in previous studies that used OCT2. The incidence of cystoid spaces may be even higher considering that only the vertical and horizontal scans were analyzed due to the retrospective nature of the present study. The higher incidence of cystoid spaces that we observed may be explained by differences in the baseline characteristics of the patients in those studies and in ours, or by the higher resolution images made available by OCT3.
Previous researchers suspected that intraretinal fluid accumulation represents cystoid edema in the outer plexiform layer.4,6 However, the findings in the intraretinal spaces observed using OCT3 had several features that are different from those characteristic of cystoid macular edema. The septa, which are thought to be composed of Mueller cells, were not observed. A foveal depression that persisted in the presence of intraretinal fluid suggests that the outer plexiform layer is not the site of fluid accumulation. This is because the outer plexiform layer and the photoreceptor cells are the only components of the foveal depression.
Tsujikawa6 described common features of cystoid spaces. A large subfoveal cystoid space was separated from the subretinal fluid by a thin septum. Weak reflectivity was noted in the cystoid space, whereas no reflectivity was seen in the subretinal fluid. de Smet et al. believed that the thin septum represented subretinal fibrosis or a fibrin membrane.7
Yamaguchi et al. proposed a hypothesis supporting the subretinal fibrin theory.8 They found numerous dot reflexes in the cystoid space and suspected that the fibrin membrane is formed on the RPE in the subretinal fluid. In addition, they believed that septa are formed by a detachment of the fibrin membrane caused by the additional fluid flux through the RPE. The septa were resolved after steroid treatment. We also observed resolution of septa after steroid treatment. Yamaguchi et al. believed that this process represented the resolution of the fibrin membrane.
However, several findings of the present study contradict the fibrin membrane theory. First, no fibrin membrane was demonstrated on OCT other than the outer wall of the cystoid space. The organized internal reflectivity we observed, including strands and layered structures (Fig. 1C, 1D and 4C), was different from the mere dot reflexes or protein-rich fluid that represent fibrin formation. Second, the theory proposed by Yamaguchi et al. cannot explain the direct formation of a cystoid space in the detached retina caused by expanding sponge-form edema (Fig. 2). The volume of subretinal fluid would have become larger than the volume of the cystoid space if the cystoid space had been formed by fluid influx, because the fibrin membrane would have acted as a barrier to fluid influx.
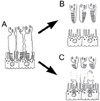
The higher resolution offered by OCT3 enabled visualization of the photoreceptor layer in the present study.9 Based on the results of our OCT3 scans, we interpreted at least some of the cystoid spaces we observed as fluid accumulation in the photoreceptor layer. Acute inflammation induce the swelling of the photoreceptors. Because the discs of the cone are the part of the plasma membrane of the cells, unfolding of the discs may enable the swelling of outer segments without rupturing the cell membrane (Fig. 6A). Although cystoid spaces are not actually vacant spaces, their reflectivity should be very low because the edematous photoreceptors have a low optical density and the cell membrane is arranged parallel to the scan beam. The OCT findings shown in Figure 2 strongly support our hypothesis. The cystoid space was "ballooned" with increasing intraretinal fluid in the photoreceptor layer. The edema progressed from the junction of the inner and outer segments to the distal photoreceptors. In addition, our hypothesis can explain why initial visual acuity was worse in the C group. This is likely because the subretinal fibrin membrane that was proposed by Yamguchi et al. is separate from the photoreceptors and would have less effect on the proper function of the sensory retina. This status could be maintained by the depressed phagocytotic activity of RPE which was damaged by inflammation.
The steroid treatment will resolve the conditions proposed above. If the changes of the photoreceptors are reversible, the outer segments can regain their normal folded structures and the intraretinal space will be resolved simply (Fig. 4 and 6B). The layered structure in Figure 4 is suspected to be the photoreceptor layer being recovered to folded structures. If the outer segments are irreversibly damaged, the distal ends of the photoreceptors should be removed by phagocytosis of the RPE. This process can be seen on OCT as a mergence of the separated photoreceptor layer with the RPE layer. As a consequence, the outer wall of the cystoid space disappears and the intraretinal space is connected to the subretinal space. Then only subretinal fluid is observed (Fig. 3 and Fig. 6C).
We questioned what caused fluid to accumulate in the photoreceptor layer but not in the subretinal space. We propose that the immunologic response seen in VKH disease may target not only the pigmented tissue, but also the photoreceptors directly. A study of photopigment regeneration reported that VKH disease is a disorder of the foveal cone.10 No photopigment regeneration took place after VKH disease was found using foveal cone densitometry. This was found despite good visual acuity 15 months after the last episode of active inflammation. The regeneration of photopigments was delayed more than healing after rhegmatogenous retinal detachment.11
Our hypothesishas several limitations. Subretinal fibrin theory is supported by biomicroscopic observations7 and OCT scans taken during the chronic phase,3 while our hypothesis is based entirely on OCT findings. It is difficult to obtain a specimen that provides histological evidence of active VKH disease. Electrophysiologic studies, such as focal eletroretinograms of cystoid spaces, may provide information regarding damage to the photoreceptors. In addition, a very large cystoid space would have difficulty forming due to swelling of the photoreceptors alone. It is likely that both mechanisms, subretinal fibrin formation and photoreceptor layer edema, are related to the formation of a cystoid space.
A cystoid space in the outer retina is a common presentation in acute VKH disease. Our results suggest that edema of the photoreceptor layer plays a role in the formation of cystoid spaces. These features may represent inflammatory damage to the photoreceptors. We expect that more information will be obtained using higher resolution OCT machines such as an ultra-high resolution OCT or a spectral domain OCT.
Figures and Tables
Fig. 1
Cross-sectional images taken using high resolution optical coherence tomography (OCT) show the various features of the cystoid space in acute Vogt-Koyanagi-Harada disease. (A) A cystoid space that does not involve the center of the fovea on a 9 mm vertical OCT scan of the left eye from a 33-year-old man. Visual acuity was 20/20. Sponge-like edema is noted in the photoreceptor layer (arrows). (B) Optically empty cystoid spaces with thin outer walls (arrowheads) are seen on a 9 mm horizontal OCT scan from a 43-year-old woman. Sponge-like edema expanding toward the cystoid space is seen in the photoreceptor layer (arrows). (C) A 9 mm horizontal OCT scan from a 21 year-old man. A layered structure is noted in the cystoid space (short arrows). (D) A 9 mm horizontal OCT scan from a 46 year-old woman demonstrates a large cystoid space with a thin outer wall (arrowheads). Multiple vertical strands are noted in the space (short arrows). In the every case, all of the retinal layers can be identified inside the cystoid space. The outer boundary of a portion of the cystoid space continues to the photoreceptor layers (A and C, arrowheads).
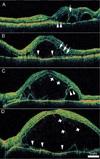
Fig. 2
Serial optical coherence tomography (OCT) scans from a 47 year-old woman with Vogt-Koyanagi-Harada disease demonstrate the development of a cystoid space in the photoreceptor layer. Serous retinal detachment and sponge-like edematous changes of the photoreceptor layer are noted in the horizontal (A) and vertical (B) scans. A highly-reflective band representing the junction of the inner and outer segments is noted (arrows). The next day, before the initiation of intravenous steroid treatment, horizontal (C) and vertical (D) OCT scans show that the intraretinal cystoid space had ballooned to oppress the subretinal space (arrowheads). All retinal layers are identified, including the outer nuclear layer (arrow, ON) and the outer plexiform layer (arrow, OP).
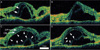
Fig. 3
Serial optical coherence tomography (OCT) scans from a 32-year-old man diagnosed with Vogt-Koyanagi-Harada disease. (A) Intraretinal fluid is noted on a 9 mm horizontal OCT scan of the right eye after three days of treatment with oral prednisolone. The margins of the cystoid space are indicated by arrows. (B) Methylprednisolone (125 mg) was infused intravenously for three days. On day six, as the outer boundary of the intraretinal space degraded, the cystoid and the subretinal spaces became interconnected (arrows). Defects or notches are seen at the margins of the previous cystoid space (arrowheads). An oral steroid was prescribed again. (C and D) The subretinal fluid gradually resolved over one month.
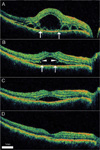
Fig. 4
Serial 9 mm vertical optical coherence tomography (OCT) scans from the right eye of a 43-year-old woman diagnosed with Vogt-Koyanagi-Harada disease. (A) An OCT scan before steroid treatment demonstrates a large cystoid space involving the fovea (arrows). (B) On the third day, multiple layered structures are noted in the cystoid space. (C) On day seven, the cystoid space decreased in size and the layered structure became more distinct. (D) After two weeks of steroid treatment, a small volume of subretinal fluid was noted under the fovea without a cystoid space.
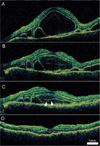
Fig. 5
Changes in visual acuity during acute episodes of Vogt-Koyanagi-Harada disease during systemic steroid therapy according to location of the fluid. The visual acuity of the group with a cystoid space was significantly worse compared to the group without a cystoid space. The differences in visual acuity between the two groups was found no later than four days after treatment began (*p<0.05).
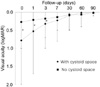
Fig. 6
An explanation of the formation of an outer retinal cystoid space caused by photoreceptor layer edemain acute Vogt-Koyanagi-Harada (VKH) disease. (A) Acute swelling of the photoreceptors unfolds the discs of the outer segments. (B) If the changes in the photoreceptor are reversible, the photoreceptors recover as the edema improves. (C) If the changes are irreversible, the plasma membrane is degraded and the separated outer segments are removed by phagocytosis of the RPE. In either case, subretinal fluid will emerge in the area of the cystoid space after the resolution.
References
1. Read RW, Holland GN, Rao NA, et al. Revised diagnostic criteria for Vogt-Koyanagi-Harada disease: report of an international committee on nomenclature. Am J Ophthalmol. 2001. 131:647–652.
2. Rubsamen PE, Gass JD. Vogt-Koyanagi-Harada syndrome. Clinical course, therapy, and long-term visual outcome. Arch Ophthalmol. 1991. 109:682–687.
3. Parc C, Guenoun JM, Dhote R, Brezin A. Optical coherence tomography in the acute and chronic phases of Vogt-Koyanagi-Harada disease. Ocul Immunol Inflamm. 2005. 13:225–227.
4. Maruyama Y, Kishi S. Tomographic features of serous retinal detachment in Vogt-Koyanagi-Harada syndrome. Ophthalmic Surg Lasers Imaging. 2004. 35:239–242.
5. Hassenstein A, Bialasiewicz AA, Richard G. Optical coherence tomography in uveitis patients. Am J Ophthalmol. 2000. 130:669–670.
6. Tsujikawa A, Yamashiro K, Yamamoto K, et al. Retinal cystoid spaces in acute Vogt-Koyanagi-Harada syndrome. Am J Ophthalmol. 2005. 139:670–677.
7. de Smet MD, Rao NA. Retinal cystoid spaces in acute Vogt-Koyanagi-Harada syndrome. Am J Ophthalmol. 2005. 140:962–963.
8. Yamaguchi Y, Otani T, Kishi S. Tomographic Features of Serous Retinal Detachment With Multilobular Dye Pooling in Acute Vogt-Koyanagi-Harada Disease. Am J Ophthalmol. 2007. 144:260–265.
9. Ko TH, Fujimoto JG, Schuman JS, et al. Comparison of ultrahigh- and standard-resolution optical coherence tomography for imaging macular pathology. Ophthalmology. 2005. 112:1922.e1–1922.e15.
10. Okamoto Y, Miyake Y, Horio N, et al. Delayed regeneration of foveal cone photopigments in Vogt-Koyanagi-Harada disease at the convalescent stage. Invest Ophthalmol Vis Sci. 2004. 45:318–322.
11. Liem AT, Keunen JE, van Meel GJ, van Norren D. Serial foveal densitometry and visual function after retinal detachment surgery with macular involvement. Ophthalmology. 1994. 101:1945–1952.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download