Abstract
A 40-year-old female visited our clinic for visual disturbance of the right eye, in which a few creamy-yellow retinal lesions and visual field constrictions were noted. She had been treated for primary CNS lymphoma and was in complete remission. After failure to follow-up for three months, she lost vision in the right eye, at which time active panuveitis was seen. Decreased vision and field constriction was observed in the left eye. Her left eye showed a granular pattern and dye leakage from the vessels and disc on fluorescein angiography and small RPE humps were seen in optical coherence tomography (OCT). Diffuse large malignant B-cells with strong immunoreactivities with CD20 immunostaining were seen in the epiretinal membrane biopsy specimen. Intravitreal injections of methotrexate (MTX) (800 µg/0.1 ml in the right eye, 400 µg/0.05 ml in the left eye) were performed twice weekly for one month, once weekly for the following month, once every two weeks for the next month, followed by nine monthly injections. Both eyes were free from malignant cells on vitreous biopsy six months later. There was no leakage seen by angiography, but the granular pattern persisted. Visual field constriction was slightly improved, and the small RPE humpsdetachments seen in OCT disappeared. EOG Arden ratio was decreased in both eyes, and b wave amplitude of scotopic ERG was decreased in the left eye. She was free from recurrence until six months later. No ocular complications except minimal opacity of the crystalline lenses were noted in both eyes.
Primary intraocular lymphoma (PIOL) is considered to be a subset of primary CNS lymphoma (PCNSL).1 One-fifth of patients with PCNSL may demonstrate ocular involvement at the time of diagnosis, and the eye can be involved prior of the onset of CNS disease.2,3 The exact incidence of PIOL is unknown, but it is reported to be in the range of 30 to 50 cases per 10 million people in the United States.4,5 Only one case has been reported in Korea.6
So far, there are three main treatment options for PIOL; systemic high-dose MTX injection, orbital irradiation, and intravitreal chemotherapy with MTX injections. These may be combined or used separately.7,8 Initial approaches for the treatment of PIOL/PCNSL centered on radiotherapy, but it had a minimal effect on survival and may have been associated with radiation retinopathy.9 Isolated radiation therapy is currently less commonly used,10 and treatment can be limited to the eye in patients with PIOL limited to the eye.11 There are some reports of PIOL effectively treated with intravitreal chemotherapy, and it has been often recommended as a first-line therapy for initial and recurrent PIOL.11-13
To our knowledge, this is the first report of PIOL treated by intravitreal chemotherapy, and the first to describe fluorescein angiographic findings, visual field exams, OCT findings and the electrophysiological characteristics of PIOL in Koreas.
A 40-year-old female visited our clinic for agradual visual disturbance in her right eye in January 2007. Her best-corrected visual acuity (BCVA) was 20/160 OD and 20/20 OS. A few barely detectable creamy-yellow retinal lesions were seen in the right eye. Eighteen months prior, she had been diagnosed with PCNSL in the right temporo-parietal lobe which had been pathologically confirmed as malignant large B-cells. Systemic chemotherapy with MTX was used initially, but it failed. Complete remission was achieved by whole brain irradiation. A granular pattern of mixed hyperfluorescent and hypofluorescent spots, along with leakage from the retinal vessels and the optic disc, were observed in fluorescein angiography of the right eye. Her visual field was considerably constricted and OCT (STRATUS OCT model 3000, Zeiss Instruments INC. San Leandro, CA) showed small irregular RPE detachments in the right eye (Fig. 1). P1 amplitude and latency of the right eye were markedly decreased compared with those of the left eye, as determined by pattern visual evoked potentials (VEP). Prominent suppression of the central signal was noted in multifocal ERG of the right eye compared with that of the left eye (Fig. 2).
After three months of failure to follow-up, the patient's BCVA decreased to no light perception (NLP) OD and 20/200 OS, and active panuveitis was observed in the right eye. Shallow retinal detachment, vitreous debris and chorioretinal thickening were detected by ultrasonography. The left eye had mild uveitis and multiple vitreous seedings. It showed a granular pattern and leakage in angiography, small RPE humps in OCT, and considerable visual field constriction (Fig. 3). A thorough systemic evaluation was performed, but there was no evidence of intracranial or visceral involvement, and the hematologic exam was nonspecific except for an elevated erythrocyte sedimentation rate. Mega-dose intravenous steroid therapy was carried out, followed by diagnostic vitrectomy with retinal biopsy of the right eye.
Diffuse large malignant B-cells with strong immunoreactivities in CD20 immunostaining were seen in the preretinal membrane biopsy specimen otherwise, no abnormal cells were obtained from the vitreous biopsy (Fig. 4). Intravitreal injections of MTX (800 µg/0.1 ml in the right eye, 400 µg/0.05 ml in the left eye) were performed twice weekly for one month, once weekly for the next month, once every two weeks for the following month, followed by nine monthly injections. During the treatment, punctuate epithelial erosions and vortex keratopathy developed, but they subsided completely with topical instillation of 0.003% leucovorin eye drops and lubricants (Fig. 5).
The final BCVA was NLP OD, 20/20 OS, and both eyes were free from malignant cells in vitreous biopsy six months later. The granular pattern on fluorescein angiography was persistent but there was no dye leakage (Fig. 6). In the left eye, the visual field constriction was slightly improved, and the small RPE humps in OCT had disappeared. Although P1 latency was markedly delayed in the right eye compared with that of the left eye, the P1 amplitudes showed a minimal difference in flash VEP. The EOG Arden ratio was 0.9604 OD and 1.231 OS. The amplitude of b waves was markedly decreased in a scotopic ERG of the left eye the photopic ERG response, oscillatory potentials and 30 Hz flicker response were relatively well preserved, with ERG waves being flat in the right eye. The patient was free of intracranial and ocular recurrence until six months later. No ocular complications except minimal opacities of the crystalline lenses were noted in both eyes.
Of ocular masquerade syndromes, PIOL is reported to be the most common.14 PIOL typically presents as a posterior uveitis4 non-specific findings of vitreous debris, chorioretinal thickening, widening of the optic nerve, elevated chorioretinal lesions, and retinal detachment can be seen in ultrasonography.15 Subretinal and sub-RPE infiltrations of PIOL are reported to be common, and creamy-yellow lesions on a fundal exam might be associated with PED,16 which was demonstrated in our OCT images. On fluorescein angiography, small sub-RPE infiltrations may appear as blocked hypofluorescence, and as the tumor regresses, it can become an RPE window defect.16 In our case, vasculitis and disc leakage disappeared as the tumor regressed; the granular pattern which might have been due to clumps of pigment epithelial cells, RPE atrophy and subretinal fibrosis, were persistent. Vitreous biopsy can be a useful tool to diagnose PIOL, but prior steroid therapy might suppress the number of vitreous cells, including lymphoma cells, which may result in a negative vitreous cytology.17
Electrophysiological findings obtained from eyes with PIOL were commented on in a brief report by Wang et al.18 which showed subnormal rod and cone responses. In our case, the b wave amplitude of the scotopic ERG of the left eye was markedly decreased; other ERG waves might be subnormal but were relatively well preserved. The EOG Arden ratio was significantly reduced in both eyes and could represent abnormal outer retinal and RPE functions.
Intravitreal chemotherapy has been reported to have few complications,11,12 and because MTX is well tolerated by intraocular tissues, repeated injections are feasible without retinal toxicity.19 Toxic keratopathy can also be easily managed with topical leucovorin eye drops and lubricants.
We modified the chemotherapy protocol of a previous report12 by adding two additional once-every-two-week injections for consolidation because the patient had useful vision in only one eye and any other treatment choice was not readily available. We also used a double dose regimen in the right eye where tumor involvement of the vitreous and retina was massive, no useful vision remained, and, being a vitrectomized eye, a shorter half-life of MTX was expected. Intravitreal chemotherapy was effective even though the patient had a history of systemic chemotherapy failure for PCNSL.
In summary, primary intravitreal chemotherapy can be effectively used to treat PIOL and had minimal ocular complications in this patient.
Figures and Tables
Fig. 1
(A) Fluorescein angiography; some perivascular dye leakage, disc hyperfluorescence and granular patterns of scattered hypofluorescent and hyperfluorescent spots are seen in the right eye. (B) Visual field of the right eye; considerable peripheral constriction is seen in pattern deviation. (C) OCT of the right eye; small RPE humps are seen.
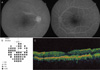
Fig. 2
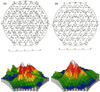
Multifocal ERG; trace arrays and 3D-topography ofthe response density in the right eye shows prominent suppression of the central signal compared with that of the left eye.
References
1. Chan CC, Buggage RR, Nussenblatt RB. Intraocular lymphoma. Curr Opin Ophthalmol. 2002. 13:411–418.
2. Peterson K, Gordon KB, Heinemann MH, DeAngelis LM. The clinical spectrum of ocular lymphoma. Cancer. 1993. 72:843–849.
3. Davis JL. Diagnosis of intraocular lymphoma. Ocul Immunol Inflamm. 2004. 12:7–16.
4. Nussenblatt RB, Chan CC, Wilson WH, et al. International Central Nervous System and Ocular Lymphoma Workshop: recommendations for the future. Ocul Immunol Inflamm. 2006. 14:139–144.
5. Hochberg FH, Miller DC. Primary central nervous system lymphoma. J Neurosurg. 1988. 68:835–853.
6. Lee SH, Kim DJ, Kim IT. A Case of Primary Central Nervous System Lymphoma with Ocular Involvement. J Korean Ophthalmol Soc. 2005. 46:565–571.
7. Nelson DF, Martz KL, Bonner H, et al. Non-Hodgkin's lymphoma of the brain: can high dose, large volume radiation therapy improve survival? Report on a prospective trial by the Radiation Therapy Oncology Group (RTOG): RTOG 8315. Int J Radiat Oncol Biol Phys. 1992. 23:9–17.
8. Batchelor TT, Kolak G, Ciordia R, et al. High-dose methotrexate for intraocular lymphoma. Clin Cancer Res. 2003. 9:711–715.
9. Berenbom A, Davila RM, Lin HS, Harbour JW. Treatment outcomes for primary intraocular lymphoma: implications for external beam radiotherapy. Eye. 2007. 21:1198–1201.
10. Margolis L, Fraser R, Lichter A, Char DH. The role of radiation therapy in the management of ocular reticulum cell sarcoma. Cancer. 1980. 45:688–692.
11. Sou R, Ohguro N, Maeda T, et al. Treatment of primary intraocular lymphoma with intravitreal methotrexate. Jpn J Ophthalmol. 2008. 52:167–174.
12. Frenkel S, Hendler K, Siegal T, et al. Intravitreal methotrexate for treating vitreoretinal lymphoma: 10 years of experience. Br J Ophthalmol. 2008. 92:383–388.
13. Behin A, Hoang-Xuan K, Carpentier AF, Delattre JY. Primary brain tumours in adults. Lancet. 2003. 361:323–331.
14. Rothova A, Ooijman F, Kerkhoff F, et al. Uveitis masquerade syndromes. Ophthalmology. 2001. 108:386–399.
15. Ursea R, Heinemann MH, Silverman RH, et al. Ophthalmic, ultrasonographic findings in primary central nervous system lymphoma with ocular involvement. Retina. 1997. 17:118–123.
16. Corriveau C, Easterbrook M, Payne D. Lymphoma simulating uveitis (masquerade syndrome). Can J Ophthalmol. 1986. 21:144–149.
17. Whitcup SM, de Smet MD, Rubin BI, et al. Intraocular lymphoma. Clinical and histopathologic diagnosis. Ophthalmology. 1993. 100:1399–1406.
18. Wang JK, Yang CM, Lin CP, et al. An Asian patient with intraocular lymphoma treated by intravitreal methotrexate. Jpn J Ophthalmol. 2006. 50:474–478.
19. Velez G, Yuan P, Sung C, et al. Pharmacokinetics and toxicity of intravitreal chemotherapy for primary intraocular lymphoma. Arch Ophthalmol. 2001. 119:1518–1524.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download