1. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Hanna SE, Makarski J. Development of the AGREE II, part 1: performance, usefulness and areas for improvement. CMAJ. 2010; 182:1045–1052.
2. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Hanna SE, Makarski J. Development of the AGREE II, part 2: assessment of validity of items and tools to support application. CMAJ. 2010; 182:E472–8.
3. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, et al. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010; 182:E839–42.
4. Makarski J, Brouwers MC, Enterprise AG. The AGREE Enterprise: a decade of advancing clinical practice guidelines. Implement Sci. 2014; 9:103.
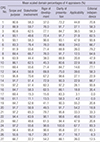
5. Jo MW, Lee JY, Kim NS, Kim SY, Sheen S, Kim SH, Lee SI. Assessment of the quality of clinical practice guidelines in Korea using the AGREE Instrument. J Korean Med Sci. 2013; 28:357–365.
6. Ahn HS, Kim HJ. Development and implementation of clinical practice guidelines: current status in Korea. J Korean Med Sci. 2012; 27:Suppl. S55–60.
7. Lee YK, Shin ES, Shim JY, Min KJ, Kim JM, Lee SH; Executive Committee for CPGs. Korean Academy of Medical Sciences. Developing a scoring guide for the Appraisal of Guidelines for Research and Evaluation II instrument in Korea: a modified Delphi consensus process. J Korean Med Sci. 2013; 28:190–194.
8. Oh MK, Jo H, Lee YK. Improving the reliability of clinical practice guideline appraisals: effects of the Korean AGREE II scoring guide. J Korean Med Sci. 2014; 29:771–775.
9. Sinclair D, Isba R, Kredo T, Zani B, Smith H, Garner P. World Health Organization guideline development: an evaluation. PLoS One. 2013; 8:e63715.
10. Tudor KI, Kozina PN, Marušić A. Methodological rigour and transparency of clinical practice guidelines developed by neurology professional societies in Croatia. PLoS One. 2013; 8:e69877.
11. Gagliardi AR, Brouwers MC. Do guidelines offer implementation advice to target users? A systematic review of guideline applicability. BMJ Open. 2015; 5:e007047.
12. Jokhan S, Whitworth MK, Jones F, Saunder A, Heazell AE. Evaluation of the quality of guidelines for the management of reduced fetal movements in UK maternity units. BMC Pregnancy Childbirth. 2015; 15:54.
13. Sabharwal S, Patel V, Nijjer SS, Kirresh A, Darzi A, Chambers JC, Malik I, Kooner JS, Athanasiou T. Guidelines in cardiac clinical practice: evaluation of their methodological quality using the AGREE II instrument. J R Soc Med. 2013; 106:315–322.
14. Burgers JS, Cluzeau FA, Hanna SE, Hunt C, Grol R. Characteristics of high-quality guidelines: evaluation of 86 clinical guidelines developed in ten European countries and Canada. Int J Technol Assess Health Care. 2003; 19:148–157.
15. Al-Ansary LA, Tricco AC, Adi Y, Bawazeer G, Perrier L, Al-Ghonaim M, AlYousefi N, Tashkandi M, Straus SE. A systematic review of recent clinical practice guidelines on the diagnosis, assessment and management of hypertension. PLoS One. 2013; 8:e53744.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download