1. Choi HK, Mount DB, Reginato AM; American College of Physicians; American Physiological Society. Pathogenesis of gout. Ann Intern Med. 2005; 143:499–516.
2. Bloch C, Hermann G, Yu TF. A radiologic reevaluation of gout: a study of 2,000 patients. AJR Am J Roentgenol. 1980; 134:781–787.
3. Dalbeth N, Collis J, Gregory K, Clark B, Robinson E, McQueen FM. Tophaceous joint disease strongly predicts hand function in patients with gout. Rheumatology (Oxford). 2007; 46:1804–1807.
4. Dalbeth N, Clark B, Gregory K, Gamble G, Sheehan T, Doyle A, McQueen FM. Mechanisms of bone erosion in gout: a quantitative analysis using plain radiography and computed tomography. Ann Rheum Dis. 2009; 68:1290–1295.
5. Dalbeth N, Aati O, Kalluru R, Gamble GD, Horne A, Doyle AJ, McQueen FM. Relationship between structural joint damage and urate deposition in gout: a plain radiography and dual-energy CT study. Ann Rheum Dis. 2015; 74:1030–1036.
6. Kavanaugh A, Han C, Bala M. Functional status and radiographic joint damage are associated with health economic outcomes in patients with rheumatoid arthritis. J Rheumatol. 2004; 31:849–855.
7. Gladman DD, Farewell VT, Wong K, Husted J. Mortality studies in psoriatic arthritis: results from a single outpatient center. II. Prognostic indicators for death. Arthritis Rheum. 1998; 41:1103–1110.
8. Shi D, Xu JX, Wu HX, Wang Y, Zhou QJ, Yu RS. Methods of assessment of tophus and bone erosions in gout using dual-energy CT: reproducibility analysis. Clin Rheumatol. 2015; 34:755–765.
9. McQueen FM, Doyle A, Reeves Q, Gao A, Tsai A, Gamble GD, Curteis B, Williams M, Dalbeth N. Bone erosions in patients with chronic gouty arthropathy are associated with tophi but not bone oedema or synovitis: new insights from a 3 T MRI study. Rheumatology (Oxford). 2014; 53:95–103.
10. van der Heijde D. How to read radiographs according to the Sharp/van der Heijde method. J Rheumatol. 2000; 27:261–263.
11. Dalbeth N, Clark B, McQueen F, Doyle A, Taylor W. Validation of a radiographic damage index in chronic gout. Arthritis Rheum. 2007; 57:1067–1073.
12. Dalbeth N, Doyle A, Boyer L, Rome K, Survepalli D, Sanders A, Sheehan T, Lobo M, Gamble G, McQueen FM. Development of a computed tomography method of scoring bone erosion in patients with gout: validation and clinical implications. Rheumatology (Oxford). 2011; 50:410–416.
13. Conaghan P, Bird P, Ejbjerg B, O’Connor P, Peterfy C, McQueen F, Lassere M, Emery P, Shnier R, Edmonds J, et al. The EULAR-OMERACT rheumatoid arthritis MRI reference image atlas: the metacarpophalangeal joints. Ann Rheum Dis. 2005; 64:Suppl 1. i11–i21.
14. Wallace SL, Robinson H, Masi AT, Decker JL, McCarty DJ, Yü TF. Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum. 1977; 20:895–900.
15. Shrout PE, Fleiss JL. Intraclass correlations: uses in assessing rater reliability. Psychol Bull. 1979; 86:420–428.
16. Hallgren KA. Computing inter-rater reliability for observational data: an overview and tutorial. Tutor Quant Methods Psychol. 2012; 8:23–34.
17. Lu WS, Wang CH, Lin JH, Sheu CF, Hsieh CL. The minimal detectable change of the simplified stroke rehabilitation assessment of movement measure. J Rehabil Med. 2008; 40:615–619.
18. Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007-2008. Arthritis Rheum. 2011; 63:3136–3141.
19. Dalbeth N, Doyle AJ. Imaging of gout: an overview. Best Pract Res Clin Rheumatol. 2012; 26:823–838.
20. Dalbeth N, Smith T, Nicolson B, Clark B, Callon K, Naot D, Haskard DO, McQueen FM, Reid IR, Cornish J. Enhanced osteoclastogenesis in patients with tophaceous gout: urate crystals promote osteoclast development through interactions with stromal cells. Arthritis Rheum. 2008; 58:1854–1865.
21. Lee SJ, Nam KI, Jin HM, Cho YN, Lee SE, Kim TJ, Lee SS, Kee SJ, Lee KB, Kim N, et al. Bone destruction by receptor activator of nuclear factor κB ligand-expressing T cells in chronic gouty arthritis. Arthritis Res Ther. 2011; 13:R164.
22. Dalbeth N, Pool B, Gamble GD, Smith T, Callon KE, McQueen FM, Cornish J. Cellular characterization of the gouty tophus: a quantitative analysis. Arthritis Rheum. 2010; 62:1549–1556.
23. Muehleman C, Li J, Aigner T, Rappoport L, Mattson E, Hirschmugl C, Masuda K, Rosenthal AK. Association between crystals and cartilage degeneration in the ankle. J Rheumatol. 2008; 35:1108–1117.
24. Pascual E, Ordóñez S. Orderly arrayed deposit of urate crystals in gout suggest epitaxial formation. Ann Rheum Dis. 1998; 57:255.
25. Dalbeth N, Milligan A, Doyle AJ, Clark B, McQueen FM. Characterization of new bone formation in gout: a quantitative site-by-site analysis using plain radiography and computed tomography. Arthritis Res Ther. 2012; 14:R165.
26. Roddy E. Revisiting the pathogenesis of podagra: why does gout target the foot? J Foot Ankle Res. 2011; 4:13.
27. Kim SK, Lee H, Kim JH, Park SH, Lee SK, Choe JY. Potential interest of dual-energy computed tomography in gout: focus on anatomical distribution and clinical association. Rheumatology (Oxford). 2013; 52:402–403.








 PDF
PDF ePub
ePub Citation
Citation Print
Print



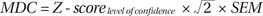
 XML Download
XML Download