1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015; 65:5–29.
2. Weis SM, Cheresh DA. Tumor angiogenesis: molecular pathways and therapeutic targets. Nat Med. 2011; 17:1359–1370.
3. Des Guetz G, Uzzan B, Nicolas P, Cucherat M, Morere JF, Benamouzig R, Breau JL, Perret GY. Microvessel density and VEGF expression are prognostic factors in colorectal cancer. Meta-analysis of the literature. Br J Cancer. 2006; 94:1823–1832.
4. Chen J, Li T, Wu Y, He L, Zhang L, Shi T, Yi Z, Liu M, Pang X. Prognostic significance of vascular endothelial growth factor expression in gastric carcinoma: a meta-analysis. J Cancer Res Clin Oncol. 2011; 137:1799–1812.
5. Chen M, Cai E, Huang J, Yu P, Li K. Prognostic value of vascular endothelial growth factor expression in patients with esophageal cancer: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev. 2012; 21:1126–1134.
6. Yuan A, Yu CJ, Chen WJ, Lin FY, Kuo SH, Luh KT, Yang PC. Correlation of total VEGF mRNA and protein expression with histologic type, tumor angiogenesis, patient survival and timing of relapse in non-small-cell lung cancer. Int J Cancer. 2000; 89:475–483.
7. Yuan A, Yu CJ, Luh KT, Chen WJ, Lin FY, Kuo SH, Yang PC. Quantification of VEGF mRNA expression in non-small cell lung cancer using a real-time quantitative reverse transcription-PCR assay and a comparison with quantitative competitive reverse transcription-PCR. Lab Invest. 2000; 80:1671–1680.
8. O’Byrne KJ, Koukourakis MI, Giatromanolaki A, Cox G, Turley H, Steward WP, Gatter K, Harris AL. Vascular endothelial growth factor, platelet-derived endothelial cell growth factor and angiogenesis in non-small-cell lung cancer. Br J Cancer. 2000; 82:1427–1432.
9. Han H, Silverman JF, Santucci TS, Macherey RS, d’Amato TA, Tung MY, Weyant RJ, Landreneau RJ. Vascular endothelial growth factor expression in stage I non-small cell lung cancer correlates with neoangiogenesis and a poor prognosis. Ann Surg Oncol. 2001; 8:72–79.
10. Shimanuki Y, Takahashi K, Cui R, Hori S, Takahashi F, Miyamoto H, Fukurchi Y. Role of serum vascular endothelial growth factor in the prediction of angiogenesis and prognosis for non-small cell lung cancer. Lung. 2005; 183:29–42.
11. Lu H, Shu XO, Cui Y, Kataoka N, Wen W, Cai Q, Ruan ZX, Gao YT, Zheng W. Association of genetic polymorphisms in the VEGF gene with breast cancer survival. Cancer Res. 2005; 65:5015–5019.
12. Hefler LA, Mustea A, Könsgen D, Concin N, Tanner B, Strick R, Heinze G, Grimm C, Schuster E, Tempfer C, et al. Vascular endothelial growth factor gene polymorphisms are associated with prognosis in ovarian cancer. Clin Cancer Res. 2007; 13:898–901.
13. Bradbury PA, Zhai R, Ma C, Xu W, Hopkins J, Kulke MJ, Asomaning K, Wang Z, Su L, Heist RS, et al. Vascular endothelial growth factor polymorphisms and esophageal cancer prognosis. Clin Cancer Res. 2009; 15:4680–4685.
14. Heist RS, Zhai R, Liu G, Zhou W, Lin X, Su L, Asomaning K, Lynch TJ, Wain JC, Christiani DC.
VEGF polymorphisms and survival in early-stage non-small-cell lung cancer. J Clin Oncol. 2008; 26:856–862.
15. Guan X, Yin M, Wei Q, Zhao H, Liu Z, Wang LE, Yuan X, O’Reilly MS, Komaki R, Liao Z. Genotypes and haplotypes of the VEGF gene and survival in locally advanced non-small cell lung cancer patients treated with chemoradiotherapy. BMC Cancer. 2010; 10:431.
16. Masago K, Fujita S, Kim YH, Hatachi Y, Fukuhara A, Nagai H, Irisa K, Ichikawa M, Mio T, Mishima M. Effect of vascular endothelial growth factor polymorphisms on survival in advanced-stage non-small-cell lung cancer. Cancer Sci. 2009; 100:1917–1922.
17. Höckel M, Vaupel P. Tumor hypoxia: definitions and current clinical, biologic, and molecular aspects. J Natl Cancer Inst. 2001; 93:266–276.
18. Vaupel P. The role of hypoxia-induced factors in tumor progression. Oncologist. 2004; 9:Suppl 5. 10–17.
19. Vaupel P, Harrison L. Tumor hypoxia: causative factors, compensatory mechanisms, and cellular response. Oncologist. 2004; 9:Suppl 5. 4–9.
20. Eilertsen M, Pettersen I, Andersen S, Martinez I, Donnem T, Busund LT, Bremnes RM. In NSCLC, VEGF-A response to hypoxia may differ between squamous cell and adenocarcinoma histology. Anticancer Res. 2012; 32:4729–4736.
21. Postmus PE, Brambilla E, Chansky K, Crowley J, Goldstraw P, Patz EF Jr, Yokomise H; International Association for the Study of Lung Cancer International Staging Committee. The IASLC lung cancer staging project: proposals for revision of the M descriptors in the forthcoming (seventh) edition of the TNM classification of lung cancer. J Thorac Oncol. 2007; 2:686–693.
22. Vincenti V, Cassano C, Rocchi M, Persico G. Assignment of the vascular endothelial growth factor gene to human chromosome 6p21.3. Circulation. 1996; 93:1493–1495.
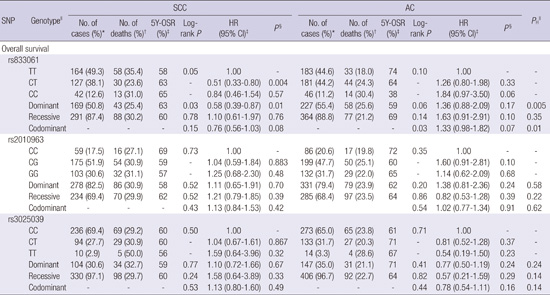
23. Tu J, Wang S, Zhao J, Zhu J, Sheng L, Sheng Y, Chen H, Tian J. rs833061 and rs699947 on promoter gene of vascular endothelial growth factor (VEGF) and associated lung cancer susceptibility and survival: a meta-analysis. Med Sci Monit. 2014; 20:2520–2526.
24. Chen MH, Tzeng CH, Chen PM, Lin JK, Lin TC, Chen WS, Jiang JK, Wang HS, Wang WS. VEGF− 460T→ C polymorphism and its association with VEGF expression and outcome to FOLFOX-4 treatment in patients with colorectal carcinoma. Pharmacogenomics J. 2011; 11:227–236.
25. Stevens A, Soden J, Brenchley PE, Ralph S, Ray DW. Haplotype analysis of the polymorphic human vascular endothelial growth factor gene promoter. Cancer Res. 2003; 63:812–816.
26. Hoeben A, Landuyt B, Highley MS, Wildiers H, Van Oosterom AT, De Bruijn EA. Vascular endothelial growth factor and angiogenesis. Pharmacol Rev. 2004; 56:549–580.
27. Ferah E, Ferda A, Sevinç B, Zuhal K, Ali A. Microvessel density as a marker of tumor angiogenesis in nonsmall cell lung cancer. Turk Respir J. 2000; 1:5–8.
28. Yuan A, Yang PC, Yu CJ, Lee YC, Yao YT, Chen CL, Lee LN, Kuo SH, Luh KT. Tumor angiogenesis correlates with histologic type and metastasis in non-small-cell lung cancer. Am J Respir Crit Care Med. 1995; 152:2157–2162.
29. Sekido Y, Fong KM, Minna JD. Molecular genetics of lung cancer. Annu Rev Med. 2003; 54:73–87.
30. Herbst RS, Heymach JV, Lippman SM. Lung cancer. N Engl J Med. 2008; 359:1367–1380.
31. Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, Lilenbaum R, Johnson DH. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006; 355:2542–2550.
32. Rizvi N, Rusch V, Zhao B, Senturk E, Schwartz L, Fury M, Downey R, Rizk N, Krug L, Kris MG. Single agent bevacizumab and bevacizumab in combination with docetaxel and cisplatin as induction therapy for resectable IB-IIIA non-small cell lung cancer. J Clin Oncol. 2007; 25:18045.
33. Schettino C, Bareschino MA, Rossi A, Maione P, Castaldo V, Mazzeo N, Sacco PC, Ferrara ML, Palazzolo G, Ciardiello F, et al. The potential role of bevacizumab in early stages and locally advanced non-small cell lung cancer. Ther Adv Med Oncol. 2009; 1:5–13.
34. Wozniak AJ, Gadgeel SM. Adjuvant therapy for resected non-small cell lung cancer. Ther Adv Med Oncol. 2009; 1:109–118.








 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download