1. Brown GW, Robinson DM, Huxsoll DL, Ng TS, Lim KJ, Sannasey G. Scrub typhus: a common cause of illness in indigenous populations. Trans R Soc Trop Med Hyg. 1976; 70:444–448.
2. Chang WH, Kang JS, Lee WK, Choi MS, Lee JH. Serological classification by monoclonal antibodies of Rickettsia tsutsugamushi isolated in Korea. J Clin Microbiol. 1990; 28:685–688.
3. Ebisawa I. Current epidemiology and treatment of tsutsugamushi disease in Japan. J Travel Med. 1995; 2:218–220.
4. Ree HI, Lee IY, Jeon SH, Yoshida Y. Geographical distribution of vectors and sero-strains of tsutsugamushi disease at mid-south inland of Korea. Korean J Parasitol. 1997; 35:171–179.
5. Watt G, Walker DH. Scrub typhus. In : Guerrant RL, Walker DH, Weller PF, editors. Tropical Infectious Diseases: Principles, Pathogens, and Practice: Vol 1. Philadelphia, PA: Churchill Livingstone;1999. p. 592–597.
6. Brown GW. Scrub typhus: pathogenesis and clinical syndrome. In : Walker DH, editor. Biology of Rickettsial Diseases: Vol I. Boca Raton, FL: CRS Press;1988. p. 93–100.
7. Munro-Faure AD, Andrew R, Missen GA, Mackay-Dick J. Scrub typhus in Korea. J R Army Med Corps. 1951; 97:227–229.
8. Groves MG. Scrub typhus. In : Beran GW, editor. Handbook of Zoonoses: Section A: Bacterial, Rickettsial, Chlamydial and Mycotic. 2nd ed. Boca Raton, FL: CRC Press;1994. p. 663–668.
9. Lee JS, Ahn C, Kim YK, Lee MH. Thirteen cases of rickettsial infection including nine cases of tsutsugamushi disease, first confirmed in Korea. J Korean Med Assoc. 1986; 29:430–438.
10. Chang WH, Kang JS. Isolation of Rickettsia tsutsugamushi from Korean patients. J Korean Med Assoc. 1987; 30:999–1008.
11. Ree HI, Cho MK, Lee IY, Jeon SH. Comparative epidemiological studies on vector/reservoir animals of tsutsugamushi disease between high and low endemic areas in Korea. Korean J Parasitol. 1995; 33:27–36.
12. Kitaoka M, Tanaka Y. Rickettsial toxin and its specificity in 3 prototype strains, Karp, Gilliam and Kato, of Rickettsia orientalis
. Acta Virol. 1973; 17:426–434.
13. Lee YM, Kim DM, Lee SH, Jang MS, Neupane GP. Phylogenetic analysis of the 56 kDa protein genes of Orientia tsutsugamushi in Southwest Area of Korea. Am J Trop Med Hyg. 2011; 84:250–254.
14. Min CH, Chang WH, Kang JS, Cho SI, Choi MK, Cho MK, Yoon CS, Kim YW. Murine typhus and scrub typhus in Kangwon-do Korea. Korean J Infect Dis. 1988; 20:105–116.
15. Bozeman FM, Elisberg BL. Serological diagnosis of scrub typhus by indirect immunofluorescence. Proc Soc Exp Biol Med. 1963; 112:568–573.
16. Elisberg BL, Campbell JM, Bozeman FM. Antigenic diversity of Rickettsia tsutsugamushi: epidemiologic and ecologic significance. J Hyg Epidemiol Microbiol Immunol. 1968; 12:18–25.
17. Jiang J, Marienau KJ, May LA, Beecham HJ 3rd, Wilkinson R, Ching WM, Richards AL. Laboratory diagnosis of two scrub typhus outbreaks at Camp Fuji, Japan in 2000 and 2001 by enzyme-linked immunosorbent assay, rapid flow assay, and Western blot assay using outer membrane 56-kD recombinant proteins. Am J Trop Med Hyg. 2003; 69:60–66.
18. Brown GW, Shirai A, Rogers C, Groves MG. Diagnostic criteria for scrub typhus: probability values for immunofluorescent antibody and Proteus OXK agglutinin titers. Am J Trop Med Hyg. 1983; 32:1101–1107.
19.
YW Kim
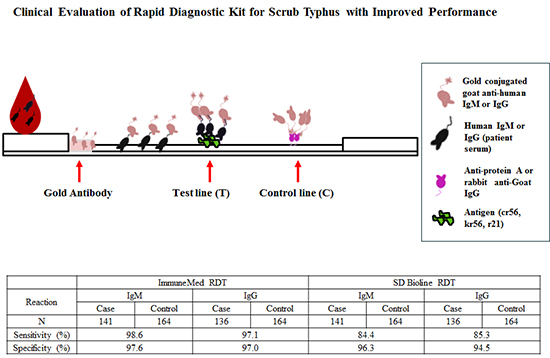
IS Kim
IA Chang
SD Woo
YJ Kim
JM Chun
WC Kim
YH Byun
MK Cho
. IMMUNEMED Inc.Diagnostic formulation for tsutsugamushi disease. Korea patent. KR 10-2006-0084490. 2008. 01. 15.
20. Rapmund G. Rickettsial diseases of the Far East: new perspectives. J Infect Dis. 1984; 149:330–338.
21. Kim YJ, Yeo SJ, Park SJ, Woo YJ, Kim MW, Kim SH, Chang IA, Jeon SH, Park BJ, Song GJ, et al. Improvement of the diagnostic sensitivity of scrub typhus using a mixture of recombinant antigens derived from Orientia tsutsugamushi serotypes. J Korean Med Sci. 2013; 28:672–679.







 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download