Abstract
Graphical Abstract
Figures and Tables
Fig. 1
Study population. A total of 2,254 very-low-birth-weight (VLBW) infants registered in the Korean Neonatal Network (KNN) from 2013.1. to 2014.6. were included. "No PDA group" was 1,206 patients (53.5%) and "PDA group" was 1,048 patients (46.5%).

Fig. 2
Preterm patent ductus arteriosus (PDA) treatment. Proportion of patients who received pharmacological (with or without surgery) and surgical treatment (both primary and secondary ligation) was decreased with the increase in gestational age and birthweight. (A) According to the gestational age, (B) According to the birthweight.

Fig. 3
Proportion of patients according to the postnatal days of the first day of patent ductus arteriosus (PDA) treatment (Proportion of patients in each treatment modality: pharmacological treatment, primary ligation, and secondary ligation). Mean postnatal days at the first dose of pharmacological treatment was 4.6±6.4 days. Mean postnatal days of surgery was 10.9±10.4 days in primary ligation group and 21.3±16.2 days in secondary ligation group.

Table 1
Patient characteristics according to the patent ductus arteriosus (PDA) treatment strategies in very-low-birth-weight infants in Korea

*P<0.05 in one-way ANOVA with Tukey or chi-square test with Bonferroni correction between no PDA, PT, PST, ST, and CT groups; †P<0.05 when compared with no PDA group; ‡P<0.05 when compared with CT group; §P<0.05 when compared with ST group; ∥P<0.05 in chi-square test or t-test between no PDA group and PDA group. PT, prophylactic treatment; PST, pre-symptomatic treatment; ST, symptomatic treatment; CT, conservative treatment without any intervention; PDA, patent ductus arteriosus; GA, gestational age at birth; HCA, histologic chorioamnionitis; PIH, pregnancy induced hypertension; SGA, small for gestational age; DR, delivery room; AS, apgar score, NA: not appropriate.
Table 2
Patent ductus arteriosus (PDA) treatment strategies according to the gestational age group

*In multinomial logit model, the odds ratio (OR) of the ratio of CT vs. PST was 5.3 [95% confidence interval (CI) 1.56-18.18] and OR of the ratio of CT vs. ST was 2.9 (95% CI 1.03-8.13) in GA <24 weeks group when compared with GA≥32 weeks group. P<0.001 in 4 PDA treatment subgroups according to GA by chi-square test. PT, prophylactic treatment; PST, pre-symptomatic treatment; ST, symptomatic treatment; CT, conservative treatment; PDA, patent ductus arteriosus; GA, gestational age; OR, odds ratio.
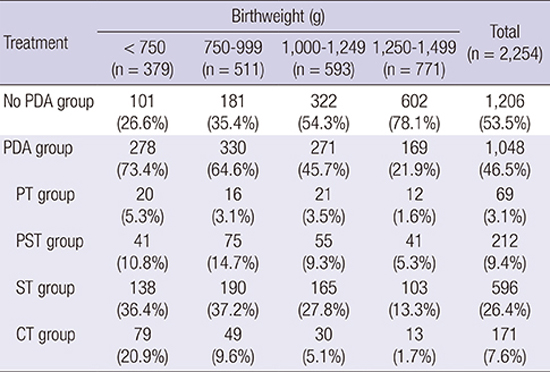
Table 3
Patent ductus arteriosus (PDA) treatment strategies according to the birthweight group

*In multinomial logit model, the odds ratio (OR) of the ratio of CT vs. PT was 3.6 (95% CI 1.45-9.17), OR of the ratio of CT vs. PST was 9.2 (95% CI 2.93-12.66), and the OR of the ratio of CT vs. ST was 4.5 (95% CI 2.39-8.62) in birthweight <750 g group when compared with birthweight 1,250-1,499 g group. P<0.001 in 4 PDA treatment subgroups according to the birthweight by chi-square test. PT, prophylactic treatment; PST, pre-symptomatic treatment; ST, symptomatic treatment; CT, conservative treatment without any intervention; PDA, patent ductus arteriosus; OR, odds ratio.
Table 4
Pharmacological treatment of preterm PDA according to the PDA treatment strategies

Table 5
Postnatal days of the first days of pharmacological treatment according to the different strategies of patent ductus arteriosus (PDA) treatment (days)

P<0.05 compared between oral, intravenous, and both oral and intravenous treatment of ibuprofen groups by one-way ANOVA test. PT, prophylactic treatment; PST, pre-symptomatic treatment; ST, symptomatic treatment; IV, intravenous; NA, not applicable; others, other medications used for treatment of preterm patent ductus arteriosus such as acetaminophen.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download