1. Kim JE, Sykes JM. Hyaluronic acid fillers: history and overview. Facial Plast Surg. 2011; 27:523–528.
2. Narins RS, Beer K. Liquid injectable silicone: a review of its history, immunology, technical considerations, complications, and potential. Plast Reconstr Surg. 2006; 118:77s–84s.
3. Lemperle G, Morhenn V, Charrier U. Human histology and persistence of various injectable filler substances for soft tissue augmentation. Aesthetic Plast Surg. 2003; 27:354–366. discussion 67.
4. Kinney BM, Hughes CE 3rd. Soft tissue fillers: an overview. Aesthet Surg J. 2001; 21:469–471.
5. Funt D, Pavicic T. Dermal fillers in aesthetics: an overview of adverse events and treatment approaches. Clin Cosmet Investig Dermatol. 2013; 6:295–316.
6. Vartanian AJ, Frankel AS, Rubin MG. Injected hyaluronidase reduces restylane-mediated cutaneous augmentation. Arch Facial Plast Surg. 2005; 7:231–237.
7. Goldberg DJ. Legal ramifications of off-label filler use. Clin Plast Surg. 2006; 33:597–601.
8. Cahill KV, Burns JA. Volume augmentation of the anophthalmic orbit with cross-linked collagen (Zyplast). Arch Ophthalmol. 1989; 107:1684–1686.
9. Carr A, Miller J, Law M, Cooper DA. A syndrome of lipoatrophy, lactic acidaemia and liver dysfunction associated with HIV nucleoside analogue therapy: contribution to protease inhibitor-related lipodystrophy syndrome. AIDS. 2000; 14:F25–F32.
10. Chan RW, Titze IR. Viscosities of implantable biomaterials in vocal fold augmentation surgery. Laryngoscope. 1998; 108:725–731.
11. Flint PW, Corio RL, Cummings CW. Comparison of soft tissue response in rabbits following laryngeal implantation with hydroxylapatite, silicone rubber, and Teflon. Ann Otol Rhinol Laryngol. 1997; 106:399–407.
12. Hallen L, Johansson C, Laurent C. Cross-linked hyaluronan (Hylan B gel): a new injectable remedy for treatment of vocal fold insufficiency--an animal study. Acta Otolaryngol. 1999; 119:107–111.
13. Dayan SH. Complications from toxins and fillers in the dermatology clinic: recognition, prevention, and treatment. Facial Plast Surg Clin North Am. 2013; 21:663–673.
14. De Boulle K. Management of complications after implantation of fillers. J Cosmet Dermatol. 2004; 3:2–15.
15. Jones D. Volumizing the face with soft tissue fillers. Clin Plast Surg. 2011; 38:379–390. v
16. Rohrich RJ, Monheit G, Nguyen AT, Brown SA, Fagien S. Soft-tissue filler complications: the important role of biofilms. Plast Reconstr Surg. 2010; 125:1250–1256.
17. Cohen JL, Bhatia AC. The role of topical vitamin K oxide gel in the resolution of postprocedural purpura. J Drugs Dermatol. 2009; 8:1020–1024.
18. Lemperle G, Rullan PP, Gauthier-Hazan N. Avoiding and treating dermal filler complications. Plast Reconstr Surg. 2006; 118:92s–107s.
19. Lemperle G, Duffy DM. Treatment options for dermal filler complications. Aesthet Surg J. 2006; 26:356–364.
20. Alam M, Gladstone H, Kramer EM, Murphy JP Jr, Nouri K, Neuhaus IM, Spencer JM, Spenceri E, Van Dyke S, Ceilley RI, et al. ASDS guidelines of care: injectable fillers. Dermatol Surg. 2008; 34:S115–S148.
21. Sclafani AP, Fagien S. Treatment of injectable soft tissue filler complications. Dermatol Surg. 2009; 35:1672–1680.
22. Alam M, Dover JS. Management of complications and sequelae with temporary injectable fillers. Plast Reconstr Surg. 2007; 120:98s–105s.
23. Cohen JL. Understanding, avoiding, and managing dermal filler complications. Dermatol Surg. 2008; 34:S92–S99.
24. Graivier MH, Bass LS, Busso M, Jasin ME, Narins RS, Tzikas TL. Calcium hydroxylapatite (Radiesse) for correction of the mid- and lower face: consensus recommendations. Plast Reconstr Surg. 2007; 120:55s–66s.
25. Beer KR. Radiesse nodule of the lips from a distant injection site: report of a case and consideration of etiology and management. J Drugs Dermatol. 2007; 6:846–847.
26. Berlin A, Cohen JL, Goldberg DJ. Calcium hydroxylapatite for facial rejuvenation. Semin Cutan Med Surg. 2006; 25:132–137.
27. Kang BS, Na YC, Jin YW. Comparison of the wound healing effect of cellulose and gelatin: an in vivo study. Arch Plast Surg. 2012; 39:317–321.
28. Mioton LM, Jordan SW, Hanwright PJ, Bilimoria KY, Kim JY. The relationship between preoperative wound classification and postoperative infection: a multi-institutional analysis of 15,289 patients. Arch Plast Surg. 2013; 40:522–529.
29. DeLorenzi C. Complications of injectable fillers, part I. Aesthet Surg J. 2013; 33:561–575.
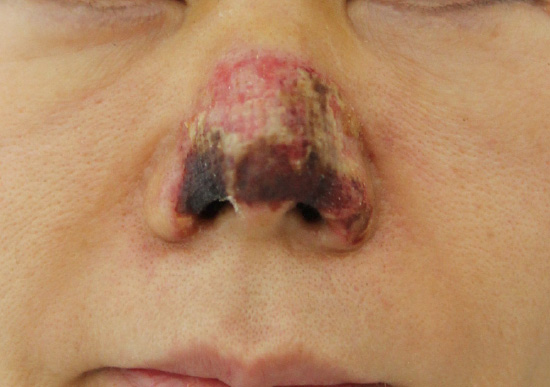
30. Kassir R, Kolluru A, Kassir M. Extensive necrosis after injection of hyaluronic acid filler: case report and review of the literature. J Cosmet Dermatol. 2011; 10:224–231.
31. Kim SG, Kim YJ, Lee SI, Lee CJ. Salvage of nasal skin in a case of venous compromise after hyaluronic acid filler injection using prostaglandin E. Dermatol Surg. 2011; 37:1817–1819.
32. Dayan SH, Arkins JP, Mathison CC. Management of impending necrosis associated with soft tissue filler injections. J Drugs Dermatol. 2011; 10:1007–1012.
33. Salibian AA, Widgerow AD, Abrouk M, Evans GR. Stem cells in plastic surgery: a review of current clinical and translational applications. Arch Plast Surg. 2013; 40:666–675.
34. Sung HM, Suh IS, Lee HB, Tak KS, Moon KM, Jung MS. Case reports of adipose-derived stem cell therapy for nasal skin necrosis after filler injection. Arch Plast Surg. 2012; 39:51–54.
35. Choi J, Minn KW, Chang H. The efficacy and safety of platelet-rich plasma and adipose-derived stem cells: an update. Arch Plast Surg. 2012; 39:585–592.
36. Bailey SH, Cohen JL, Kenkel JM. Etiology, prevention, and treatment of dermal filler complications. Aesthet Surg J. 2011; 31:110–121.
37. Ko WJ, Na YC, Suh BS, Kim HA, Heo WH, Choi GH, Lee SU. The effects of topical agent (kelo-cote or contractubex) massage on the thickness of post-burn scar tissue formed in rats. Arch Plast Surg. 2013; 40:697–704.
38. Carruthers A, Carruthers J. Non-animal-based hyaluronic acid fillers: scientific and technical considerations. Plast Reconstr Surg. 2007; 120:33s–40s.
39. Brody HJ. Use of hyaluronidase in the treatment of granulomatous hyaluronic acid reactions or unwanted hyaluronic acid misplacement. Dermatol Surg. 2005; 31:893–897.
40. Lemperle G, Gauthier-Hazan N. Foreign body granulomas after all injectable dermal fillers: part 2. Treatment options. Plast Reconstr Surg. 2009; 123:1864–1873.
41. Christensen LH. Host tissue interaction, fate, and risks of degradable and nondegradable gel fillers. Dermatol Surg. 2009; 35:1612–1619.
42. Goldman MP. Pressure-induced migration of a permanent soft tissue filler. Dermatol Surg. 2009; 35:403–405. discussion 5-6.
43. Pecharki D, Petersen FC, Scheie AA. Role of hyaluronidase in Streptococcus intermedius biofilm. Microbiology. 2008; 154:932–938.
44. Bjarnsholt T, Tolker-Nielsen T, Givskov M, Janssen M, Christensen LH. Detection of bacteria by fluorescence in situ hybridization in culture-negative soft tissue filler lesions. Dermatol Surg. 2009; 35:1620–1624.
45. Yoon TH, Yun IS, Rha DK, Lee WJ. Reconstruction of various perinasal defects using facial artery perforator-based nasolabial island flaps. Arch Plast Surg. 2013; 40:754–760.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download