This article has been
cited by other articles in ScienceCentral.
Abstract
With increase of multi-drug resistant Escherichia coli in community-acquired urinary tract infections (CA-UTI), other treatment option with a therapeutic efficacy and a low antibiotic selective pressure is necessary. In this study, we evaluated in vitro susceptibility of E. coli isolates from CA-UTI to fosfomycin (FM), nitrofurantoin (NI), temocillin (TMO) as well as trimethoprim-sulfamethoxazole (SMX), ciprofloxacin (CIP) and cefepime (FEP). The minimal inhibitory concentrations were determined by E-test or agar dilution method according to the Clinical and Laboratory Standards Institute guidelines, using 346 E. coli collected in 12 Korean hospitals from March 2010 to February 2011. FM, NI and TMO showed an excellent susceptibility profile; FM 100% (346/346), TMO 96.8% (335/346), and NI 99.4% (344/346). Conversely, resistance rates of CIP and SMX were 22% (76/346) and 29.2% (101/349), respectively. FEP still retained an activity of 98.5%. In Korea, NI and TMO in addition to FM are a good therapeutic option for uncomplicated CA-UTI, especially for lower UTI.
Keywords: Community-Acquired Infections, Urinary Tract, Escherichia coli, Fosfomycin, Nitrofurantoin, Temocillin, Trimethoprim-Sulfamethoxazole combination, Ciprofloxacin, Cefepime, Minimal Inhibitory Concentration
Urinary tract infections (UTIs) are common with an annual global incidence of at least 250 million cases; and
Escherichia coli is the most common pathogen (
1). Trimethoprim/sulfamethoxazole (SMX) has been used successfully to treat community-acquired UTI (CA-UTI) previously, but fluoroquinolones (FQ) are currently being used more frequently as the first-line treatment because of the increasing resistance to SMX among uropathogens (
2). However, FQ-resistant
E. coli is increasing recently in most countries, which becomes a great concern for public health worldwide. Several studies have reported that FQ resistance rate of
E. coli in Korea reached 30%, indicating that Korea is one of hot spots for antimicrobial resistance of
E. coli (
3). Increasing trend of multi-drug resistance of
E. coli from CA-UTI requires other treatment options.
In terms of candidate antibiotics for CA-UTI, especially for cystitis which is very common but not serious infection in community, selective pressure by antibiotics as well as treatment efficiency should be carefully considered. With this perspective, fosfomycin (FM), nitrofurantoin (NI) and temocillin (TMO) would be good candidate antibiotics for uncomplicated cystitis from community. In brief, FM has been extensively used in several European countries for the treatment of uncomplicated UTI since 1988. Despite many years of treatment use, FM continues to be characterized by low resistance of 1% against
E. coli strains worldwide (
4). NI is also an old drug used for uncomplicated UTI, but its use is limited because of nephrotoxicity. Nevertheless, the potential role of NI for uncomplicated UTI in the growing resistance era has been presented (
5,
6). In Korea, there has been a report for in vitro activity of FM (
7), but no report for TMO or NI against
E. coli isolates from urine. Objective of this study was to figure out the minimal inhibitory concentration (MIC) distribution of candidate antibiotics against uropathogenic
E. coli from CA-UTI in Korea.
As part of a nationwide multicenter surveillance study, a total of 346 community-acquired
E. coli from patients with UTI were collected from April 2010 to March 2011. The isolates were from 12 hospitals with 582-250 beds each, located throughout the Korean peninsula (3 in Seoul, 4 in Gyeonggi-do, 2 in Incheon, 2 in Daegu and 1 in Busan), and 11 were academic hospitals. The study was approved by the institutional review board (IRB) of Hanyang University Hospital (2010-007). Only the first isolate from each patient was included in the study. Susceptibility to FM, TMO, NI, ciprofloxacin (CIP), SMX and cefepime (FEP) was examined. The MIC of FM, TMO, NI, CIP, and FEP was determined by E-test (bioMérieux, Marcy L'Etoile, France), and SMX by agar dilution method according to Clinical and Laboratory Standards Institute (CLSI) guildeline (
8). Quality control was performed using
E. coli ATCC 25922. Extended-spectrum beta-lactamase (ESBL)-positivity was defined by extended-spectrum beta-lactamase test in either a semi-automated system or double disk diffusion test. We considered the isolates as plasmid-mediated AmpC beta-lactamase (PABL)-producers when they were resistant to extended-spectrum cephalosporins (ESC) but Extended-spectrum beta-lactamase test was negative.
A total of 346
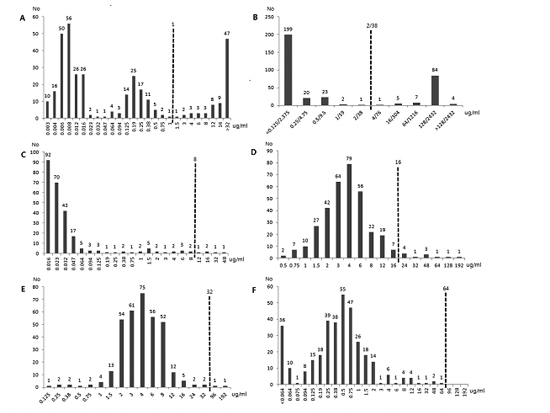
E. coli isolates were collected in urine and/or blood cultures. The susceptibility rates of antibiotics were as follows: FM 100% (346/346), TMO 96.8% (335/346), NI 99.4% (344/346), FEP 98.5% (341/346), CIP 78% (270/346) and SMX 70.8% (245/346). The MIC
50 and MIC
90 values (µg/mL) of each antibiotic were measured: FM, 0.5, 1.5; TMO, 4, 12; NI, 4, 8; FEP, 0.016, 0.064; CIP 0.016, >32; SMX, <0.125/2.375, 128/2,432.
Fig. 1 shows the MIC distribution of above antibiotics against 346
E. coli isolates.
We compared the antimicrobial resistance of 76 (22%) CIP-resistant (CIP-R) and 270 (78%) CIP-susceptible (CIP-S) isolates (
Table 1). Resistance rates of FEP, TMO and SMX were significantly higher in CIP-R than CIP-S isolates: 6.6% vs. 0% (
P<0.001); 9.2% vs. 1.5% (
P<0.001) and 55.3% vs. 21.9% (
P<0.001), respectively. On the other hand, the resistance rate of FM and NI did not differ significantly between the CIP-R and CIP-S isolates: 0% vs. 0%,
P=0.397; 0% vs. 0.7%,
P=0.637. In accordance with the co-resistance trend between CIP and FEP, TMO or SMX, the MIC
50 and MIC
90 values of those antibiotics in CIP-R isolates were higher than CIP-S isolates. By the same token, the MIC
50 and MIC
90 of NI and FM did not differ between the two groups.
With respect to extended-spectrum beta-lactamase or plasmid-mediated AmpC beta-lactamase phenotypes, 24
E. coli (6.9%) were tested positive for extended-spectrum beta-lactamase or plasmid-mediated AmpC beta-lactamase and 322 isolates (93.1%), negative. Comparing the resistance rate of antibiotics according to the ESBL/PABL positivity, resistance rates of TMO, FEP, CIP and SMX were significantly higher in ESBL/PABL-positive than ESBL/PABL-negative isolates: 12.5% vs. 2.5% (
P= 0.033); 20.8% vs. 0% (
P<0.001); 83.3% vs. 17.4% (
P<0.001) and 50% vs. 27.6% (
P=0.02), respectively (
Table 2). Conversely, the resistance rate of FM and NI did not differ: 0% vs. 0%,
P=0.143; 0% vs. 0.6%,
P=0.267. Likewise, MIC
50 and MIC
90 values of TMO, FEP, CIP and SMX were higher in ESBL/PABL-positive than ESBL/PABL-negative
E. coli isolates, but those values of FM and NI did not differ between the 2 groups.
In this study, we evaluated the in vitro susceptibility of
E. coli isolates from CA-UTI to candidate antibiotics for lower UTI such as FM, NI and TMO, which have a therapeutic efficacy but give a relatively low antibiotic selective pressure. The three antibiotics had an excellent susceptibility profile, mainly because FM is not commonly prescribed, and TMO and NI are not available in Korea. We compared the susceptibility of the three antibiotics with CIP, SMX and FEP which are major antibiotics for the treatment of CA-UTI as well as other serious infections. CIP, SMX and FEP belong to a different family of antibiotics with a different resistance mechanism, but uropathogenic
E. coli commonly shows a co-resistance against CIP, SMX and FEP. Co-resistance rates of CIP and SMX or CIP, SMX and extended-spectrum cephalosporins were 10.6%-12.8% or 1.3%-3.8% in our previous report (
9). Interestingly, FM and NI did not show a tendency for co-resistance with CIP or FEP, but ESBL/PABL-positive isolates were more resistant to TMO in this study. Temocillin is a narrow-spectrum penicillin and known to be resistant to hydrolysis by penicillinase, extended-spectrum beta-lactamases, and AmpC enzymes (
10). However, 3 of 24 ESBL/PABL-positive isolates, possibly with change of membrane permeability, were resistant to TMO in a previous study (
11). In this study, 11 of 346
E. coli were resistant to TMO at the breakpoint of 8 µg/mL, and 6 of the eleven were resistant at the British Society for Antimicrobial Chemotherapy (BSAC) urinary breakpoint (32 mg/L) (
12). In terms of NI and FM, only 1 isolate was resistant to NI, which was resistant to TMO and SMX but susceptible to CIP, FM and FEP, and there was no
E. coli resistant to FM among 346 isolates from CA-UTI. Based on these findings, FM, NI and TMO are an excellent therapeutic option for lower UTI in Korea. As we described, FQ and extended-spectrum cephalosporins are major antibiotics for treatment of UTI as well as other serious infections, and uropathogens showed a tendency for co-resistance to both major drugs. Therefore, it is mandatory to decrease the use of FQ and extended-spectrum cephalosporins for common but not-serious infections such as lower UTI. Instead, nationwide introduction of TMO, NI in addition to FM for treatment of lower UTI should be considered.
In conclusion, resistance rate of E. coli from CA-UTI to CIP and SMX were above 20% in Korea. However, TMO, FM and NI showed an excellent activity to them. Therefore, we propose TMO, FM and NI as the first-line antimicrobial agent for CA-lower UTI.







 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download