Abstract
Spinal cord injury (SCI) causes not only loss of sensory and motor function below the level of injury but also chronic pain, which is difficult and challenging of the treatment. Repetitive transcranial magnetic stimulation (rTMS) to the motor cortex, of non-invasive therapeutic methods, has the motor and sensory consequences and modulates pain in SCI-patients. In the present study, we studied the effectiveness of rTMS and the relationship between the modulation of pain and the changes of neuroglial expression in the spinal cord using a rat SCI-induced pain model. Elevated expressions of Iba1 and GFAP, specific microglial and astrocyte markers, was respectively observed in dorsal and ventral horns at the L4 and L5 levels in SCI rats. But in SCI rats treated with 25 Hz rTMS for 8 weeks, these expressions were significantly reduced by about 30%. Our finding suggests that this attenuation of activation by rTMS is related to pain modulation after SCI. Therefore, rTMS might provide an alternative means of attenuating neuropathic pain below the level of SCI.
Spinal cord injury (SCI) results in initial loss of motor, sensory, and autonomic functions below the lesion (1, 2). In the long-term, variable degrees of motor and sensory recovery have been shown, and about 70% of SCI patients develop central neuropathic pain syndrome within several months to years after injury. In primary lesions, there have been reported interruptions of axons in the spinothalamic tract and dorsal column, and sensitizations of these spinothalamic neurons due to increases in synaptic gain at glutamatergic (3-5). The mechanism of neuropathic pain after SCI has been recently reported to involve activated microglia and astrocytes (6). Furthermore, their activations have been reported to result in the consistent up-regulations of proinflammatory cytokines, chemokines, and their receptors, and the concomitant development of chronic allodynia after nerve injury (7-11).
Of the available non-invasive therapeutic tools, repetitive transcranial magnetic stimulation (rTMS) had been shown to have various therapeutic effects on diseases, such as, depression, stroke, and acute and chronic pain (12-17). rTMS has also been reported to reduce central neuropathic pain after SCI, although its mechanism of action is unclear (18). In our previous pain study, rTMS significantly changed cold allodynia (19). In particular, a significant difference in hypersensitivity was observed from 4 to 8 weeks between rTMS and sham groups. At 4 weeks, response frequency to acetone-induced stimuli was reduced in rTMS group and continuously reduced until 8 weeks. However, the sham group showed a higher frequency than the rTMS group at 4 weeks and a much higher frequency at 8 weeks.
The deactivation of activated neuroglia, that is, microglia and astrocytes, might reduce neuropathic pain (20). Furthermore, the mechanism whereby rTMS induces pain behavioral changes might be related to the deactivation of microglia and astrocytes in the spinal dorsal horn in SCI models. However, few rTMS studies have been conducted on neuroglial cellular changes in spinal cord associated with neuropathic amelioration. Hence, the aim of this study was to determine whether the attenuation of microglial and astroglial activations in the spinal cord below injuries by rTMS is related to the modulation of pain after SCI.
All experiments with adult Sprague-Dawley rats (230-250 g, female) were carried out in accordance with the guidelines issued by the institutional animal care and use committee at the Yeungnam University, Daegu, Korea. Animals were acclimatized to controlled laboratory environments (12 hr light/dark cycles) with free access to food and water. Contusive SCI was induced using a weight-drop device developed at New York University (NYU) (21, 22). Briefly, animals were anesthetized with intraperitoneal injections of Zoletil (Virbac Laboratories, France, 50 mg/kg) and laminectomy was performed at T9 to expose the dorsal surface of dura matter. The exposed spinal cord was compressed by dropping a 10 g rod (2.5 mm in diameter) from a height of 25 mm using a NYU impactor. All rats received a daily intramuscular injection of antibiotics (prophylactic kanamycin, 1 mg/kg) for 1 week after injury to prevent urinary tract infections. Manual bladder evacuation was carried out twice per a day until voiding reflexes were reestablished.
A Basso, Beattie, and Bresnahan (BBB) scores were measured as previously described (23). Rats with BBB score of 6 within 4 days after SCI were selected and randomly assigned to rTMS (n = 5) or sham (n = 5) group. The transcranial rTMS was delivered using a customized magnetic stimulator (Biocon-1000Pro, Mcube Technology, Seoul) (19). A round prototype coil (7 cm diameter) of biphasic current waveform was positioned around bregma at 1 cm from the skull. The stimulation parameters were used as the follow: biphasic current waveform, 370 µs (pulse width), 0.2 T (intensity at 100% stimulation), 25 Hz (repetition rate), and 3 sec per on/off cycle for 20 min (total stimulation time). Sham stimulation was applied using the same method, but to prevent magnetic stimulation of the brain, the coil was not activated and the active surface of the coil was placed perpendicularly to the skull. Real and sham stimulations were applied daily 5 days per week for 8 weeks from the 4th day after SCI.
For immunohistochemical staining for microglial and astrocytic markers in spinal cords, all experimental animals were sacrificed at 8 weeks after SCI. After the deep anesthesia with intraperitoneal injections of Zoletil, a catheter was inserted into the left ventricle, and whole blood stream was rinsed with 100 mL of saline, followed by 500 mL of 4% paraformaldehyde (PFA), as previously described (8, 10). Spinal cords at the L3-S1 level, that is, below the sites of injury were removed, post-fixed for 2 days in 4% PFA, stored in 20% sucrose for at least 1 day, and then, transversely sectioned at 40-50 µm using a cryostat (Leica, Germany). To enhance the penetration of antibody to tissue, spinal cord sections were reacted with 50% ethanol for 30 min, and then treated with 10% normal donkey serum (NDS, Jackson Immunoresearch, USA) to block non-specific secondary antibody reactions. Sections were then incubated overnight in a mixture of primary antibodies; anti-Iba1 (Chemicon; 1:1,000) and anti-GFAP (Chemicon, 1:1,000), and then incubated with Alexa 488-conjugated donkey anti-rabbit (Invitrogen, USA, 1:200) and Cy3-conjugated donkey anti-mouse (Jackson Immunoresearch, USA, 1:200) for 3 hr. At each step, sections were rinsed with 1 × PBS 3 times. Finally, sections were mounted with Vectashield (Vector Lab, USA) and immunofluorescent images were acquired using a cooled CCD camera (F-View II, Soft imaging system, Germany) attached to a light microscope (Leica DMR) for the quantitative analysis of Iba1 and GFAP immunoreactivities in the dorsal horn. Images obtained from 6 spinal cord sections (4 sections from L4 and L5 segments) per rat in rTMS and sham groups were analyzed using image analysis software (Analysis Pro, SIS, Germany).
Collected data were inputted into SPSS/PC version 14.0 and continuously analyzed. Results are expressed as mean ± standard deviations (SDs). Wilcoxon Signed Rank test was used to analyze baseline measures and the Mann Whitney U test was used for behavioral tests. P values of < 0.05 were considered statistically significant.
To investigate cellular changes in spinal cord caused by rTMS in SCI rats, we treated rats with 25 Hz of rTMS for 8 weeks. During the experimental periods, cold allodynia sensitivity was checked once weekly. Significant differences were observed between rTMS and sham groups from 4 to 8 weeks after injury. Hindpaw withdrawal frequency to cold allodynia was significantly lower in the rTMS than in the sham group (data not shown). After that observation, we wondered whether there is a relationship between the observed behavioral change and the concomitant cellular change of spinal cord induced by rTMS as an etiological phenomenon.
In the spinal cord of L4 and L5 segments, microglial cells, which were immunoreactive for Iba1, were evenly distributed in the gray matter and showed the wavy pattern in naïve control (Fig. 1A, a). At 8 weeks after SCI, Iba1 positive microglia numbers were higher, especially in the lamina II and III, than in naive control (Fig. 1B). In high power view of the dorsal horn at lamina II, activated microglia included round-shaped perikaryon were observed (Fig. 1b). Numbers of activated microglia increased after SCI was subsequently reduced in the spinal dorsal horn of rTMS rats (Fig. 1C, c). Quantitative analysis of Iba1-positive areas revealed a significant reduction (27.2%) in Iba1-positive areas in dorsal horn of rTMS group (average area = 26,283.32 µm2), as compared with sham group (average area = 36,104.2 µm2) (P = 0.028) (Fig. 1D).
In astrocytes, the immunoreactivity of GFAP increased in spinal dorsal horn after SCI, versus naive control (Fig. 2A, B). In particular, the density of GFAP positive cells in lamina II was notably increased, indicating that numbers of reactive astrocytes were elevated after the injury (Fig. 2b). In spinal dorsal horn, reactive astrocytes numbers subsequently decreased in the rTMS group, but not in sham group (Fig. 2C). Quantitative analysis of GFAP-positive areas confirmed a significant decrease of GFAP expression (29.2%) in the dorsal horn from rTMS group (average area = 48,192.7 µm2), as compared with sham group (average area = 68,058.6 µm2) (P = 0.009) (Fig. 2D).
In the present study, the application of 25 Hz of rTMS ameliorated cold allodynia and produced more motor recovery than others (10 or 50 Hz) in our SCI rat model (data not shown), and thus we used this frequency to cellular changes in spinal dorsal horn induced by rTMS in SCI rats. This injury noticeably induced the activations of microglia and astrocytes, as reflected by increases in the expressions of Iba1 and GFAP. Furthermore, these activations were found to be efficiently attenuated by rTMS.
Various frequencies of magnetic stimulation have been reported to be effective in different symptoms of diseases, but no uniform guidance has been issued regarding effective frequencies (24). In human studies, high frequency stimulation (20 Hz) was reported to reduce hypersensitivity and chronic pain (25). Furthermore, it has been suggested that rTMS relieves central pain by inducing GABA-mediated renormalization of cortical activity (26), and by producing a change in the balance between serotonergic and noradrenergic systems (15, 16, 27). Recently, an action mechanism was suggested for magnetic stimulation, based on signal mediation by brain-derived neurotrophic factor (BDNF) and tyrosine receptor kinase B (TrkB) (28). In addition, it has been suggested that alterations induced by magnetic stimulation in the cortical brain may affect remote subcortical structures, the thalamus or brainstem nucleus, and finally the spinal cord, which could modulate pain perception and transmission (29-32). However, few studies have addressed to spinal cord changes, after stimulation of the cortical brain.
Activated neuroglia in the spinal dorsal horn had been reported to contribute to hyper-responsive states in cases of chronic neuropathic pain. Furthermore, attenuations of the activation of microglia and astrocytes are essential for pain reduction (20). In the present study, the expression of Iba1 was elevated by SCI, and then dramatically fell in the spinal dorsal horn in rTMS group, as compared with sham group. Gwak and Hulsebosch (20) reported that spinal hemisection significantly increased GFAP expression and those of other cellular and chemical mediators, and subsequently activated astrocyte and microglia and caused chronic neuropathic pain (CNP). After nerve injury, star-shaped active astrocytes maintain persistent pain. In this study, the expressions of GFAP were markedly elevated in sham group, as compared with naive control, and the elevation was attenuated by rTMS. Furthermore, this reduction in GFAP and Iba1 expression could influence nerve fibers and nociceptors transmitting cold allodynia after spinal cord damage (33, 34).
The concomitant reduction of neuroglial activation and cold allodynia could be important in the context of the hypersensitivity of pain response. Here, we found a relation between cold allodynia and neuroglial cellular activation in spinal cord in SCI-rats. Our findings suggest that rTMS reduces pain by attenuating the activations of microglia and astrocytes, and to the best of our knowledge, this is the first report to claim that rTMS reduces spinal cord neuroglial cellular activity below level CNP after SCI (20). Furthermore, rTMS could be used to control CNP after SCI. Further mechanistic studies are required to identify detail signal pathways.
Figures and Tables
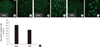 | Fig. 1Expression of Iba1 in the spinal dorsal horns. Immunofluorescent images of spinal dorsal horns from (A) the naive control, (B) the sham, and (C) rTMS groups. Numbers of Iba1 positive microglia were increased at 8 weeks after injury in sham group (B). Activated microglia with round-shaped perikaryon were observed in high power view (a, b, and c). However, activated microglia were less numerous in spinal dorsal horn in the rTMS group than in the sham group (C). Size of scale bar: 100 µm (D). Quantitative analysis of numbers of Iba1 immunoreactive cells in spinal dorsal horns. Iba1 positive areas were significantly attenuated by rTMS (P = 0.028). Results are presented as mean ± SDs. |
 | Fig. 2Expression of GFAP in the spinal dorsal horns. The spinal dorsal horns were from (A) naive control, (B) sham, and (C) rTMS groups. Immunoreactity for GFAP increased in the spinal dorsal horns of L4-5 segments after SCI (B). At higher magnification, reactive astrocytes with increased immunoreactivity and hypertrophy were noted (b). Reactive astrocyte numbers, GFAP expression, were lower in rTMS than in sham group (C and c). Size of scale bar: 100 µm (D). Quantitative analysis of the number of GFAP immunereactive cells in spinal dorsal horns. In quantitative analysis of numbers of GFAP, the immunoreactive positive area was significantly lower in the rTMS group, than in sham group (P = 0.009). Results are presented as mean ± SDs. |
References
1. Hulsebosch CE. From discovery to clinical trials: treatment strategies for central neuropathic pain after spinal cord injury. Curr Pharm Des. 2005. 11:1411–1420.
2. Nakae A, Nakai K, Yano K, Hosokawa K, Shibata M, Mashimo T. The animal model of spinal cord injury as an experimental pain model. J Biomed Biotechnol. 2011. 2011:939023.
3. Willis A, Mihalevich M, Neff RA, Mendelowitz D. Three types of postsynaptic glutamatergic receptors are activated in DMNX neurons upon stimulation of NTS. Am J Physiol. 1996. 271:R1614–R1619.
4. Salter MW. Cellular neuroplasticity mechanisms mediating pain persistence. J Orofac Pain. 2004. 18:318–324.
5. Wasner G, Lee BB, Engel S, McLachlan E. Residual spinothalamic tract pathways predict development of central pain after spinal cord injury. Brain. 2008. 131:2387–2400.
6. Hains BC, Waxman SG. Activated microglia contribute to the maintenance of chronic pain after spinal cord injury. J Neurosci. 2006. 26:4308–4317.
7. Colburn RW, Rickman AJ, DeLeo JA. The effect of site and type of nerve injury on spinal glial activation and neuropathic pain behavior. Exp Neurol. 1999. 157:289–304.
8. Kim SJ, Park SM, Cho YW, Jung YJ, Lee DG, Jang SH, Park HW, Hwang SJ, Ahn SH. Changes in expression of mRNA for interleukin-8 and effects of interleukin-8 receptor inhibitor in the spinal dorsal horn in a rat model of lumbar disc herniation. Spine. 2011. 36:2139–2146.
9. Lindia JA, McGowan E, Jochnowitz N, Abbadie C. Induction of CX3CL1 expression in astrocytes and CX3CR1 in microglia in the spinal cord of a rat model of neuropathic pain. J Pain. 2005. 6:434–438.
10. Park HW, Ahn SH, Kim SJ, Seo JM, Cho YW, Jang SH, Hwang SJ, Kwak SY. Changes in spinal cord expression of fractalkine and its receptor in a rat model of disc herniation by autologous nucleus pulposus. Spine (Phila Pa 1976). 2011. 36:E753–E760.
11. Sweitzer SM, Colburn RW, Rutkowski M, DeLeo JA. Acute peripheral inflammation induces moderate glial activation and spinal IL-1beta expression that correlates with pain behavior in the rat. Brain Res. 1999. 829:209–221.
12. Grunhaus L, Dannon PN, Schreiber S, Dolberg OH, Amiaz R, Ziv R, Lefkifker E. Repetitive transcranial magnetic stimulation is as effective as electroconvulsive therapy in the treatment of nondelusional major depressive disorder: an open study. Biol Psychiatry. 2000. 47:314–324.
13. Kim YH, You SH, Ko MH, Park JW, Lee KH, Jang SH, Yoo WK, Hallett M. Repetitive transcranial magnetic stimulation-induced corticomotor excitability and associated motor skill acquisition in chronic stroke. Stroke. 2006. 37:1471–1476.
14. Lomarev MP, Kim DY, Richardson SP, Voller B, Hallett M. Safety study of high-frequency transcranial magnetic stimulation in patients with chronic stroke. Clin Neurophysiol. 2007. 118:2072–2075.
15. Pridmore S, Oberoi G. Transcranial magnetic stimulation applications and potential use in chronic pain: studies in waiting. J Neurol Sci. 2000. 182:1–4.
16. Pridmore S, Oberoi G, Marcolin M, George M. Transcranial magnetic stimulation and chronic pain: current status. Australas Psychiatry. 2005. 13:258–265.
17. Saitoh Y, Hirayama A, Kishima H, Shimokawa T, Oshino S, Hirata M, Tani N, Kato A, Yoshimine T. Reduction of intractable deafferentation pain due to spinal cord or peripheral lesion by high-frequency repetitive transcranial magnetic stimulation of the primary motor cortex. J Neurosurg. 2007. 107:555–559.
18. Defrin R, Grunhaus L, Zamir D, Zeilig G. The effect of a series of repetitive transcranial magnetic stimulations of the motor cortex on central pain after spinal cord injury. Arch Phys Med Rehabil. 2007. 88:1574–1580.
19. Bae YK, Kim SJ, Seo JM, Cho YW, Ahn SH, Kang IS, Park HW, Hwang S. Effects of continuous repetitive transcranial magnetic stimulation on pain response in spinal cord injured rat. J Korean Acad Rehabil Med. 2010. 34:259–264.
20. Gwak YS, Hulsebosch CE. Remote astrocytic and microglial activation modulates neuronal hyperexcitability and below-level neuropathic pain after spinal injury in rat. Neuroscience. 2009. 161:895–903.
21. Gruner JA. A monitored contusion model of spinal cord injury in the rat. J Neurotrauma. 1992. 9:123–128.
22. Anderson AJ, Robert S, Huang W, Young W, Cotman CW. Activation of complement pathways after contusion-induced spinal cord injury. J Neurotrauma. 2004. 21:1831–1846.
23. Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995. 12:1–21.
24. Canovero S, Barbara MM, Zollino G. Canovero S, editor. Noninvasive stimulation for choronic pain. Textbook of therapeutic cortical stimulation. 2009. NY: Nova Science Publishers Inc.;113–179.
25. Johnson S, Summers J, Pridmore S. Changes to somatosensory detection and pain thresholds following high frequency repetitive TMS of the motor cortex in individuals suffering from chronic pain. Pain. 2006. 123:187–192.
26. Canavero S, Bonicalzi V, Dotta M, Vighetti S, Asteggiano G, Cocito D. Transcranial magnetic cortical stimulation relieves central pain. Stereotact Funct Neurosurg. 2002. 78:192–196.
27. Ben-Shachar D, Gazawi H, Riboyad-Levin J, Klein E. Chronic repetitive transcranial magnetic stimulation alters beta-adrenergic and 5-HT2 receptor characteristics in rat brain. Brain Res. 1999. 816:78–83.
28. Wang HY, Crupi D, Liu J, Stucky A, Cruciata G, Di Rocco A, Friedman E, Quartarone A, Ghilardi MF. Repetitive transcranial magnetic stimulation enhances BDNF-TrkB signaling in both brain and lymphocyte. J Neurosci. 2011. 31:11044–11054.
29. Leo RJ, Latif T. Repetitive transcranial magnetic stimulation (rTMS) in experimentally induced and chronic neuropathic pain: a review. J Pain. 2007. 8:453–459.
30. Lefaucheur JP, Drouot X, Menard-Lefaucheur I, Keravel Y, Nguyen JP. Motor cortex rTMS restores defective intracortical inhibition in chronic neuropathic pain. Neurology. 2006. 67:1568–1574.
31. Lefaucheur JP, Hatem S, Nineb A, Menard-Lefaucheur I, Wendling S, Keravel Y, Nguyen JP. Somatotopic organization of the analgesic effects of motor cortex rTMS in neuropathic pain. Neurology. 2006. 67:1998–2004.
32. Pleger B, Janssen F, Schwenkreis P, Volker B, Maier C, Tegenthoff M. Repetitive transcranial magnetic stimulation of the motor cortex attenuates pain perception in complex regional pain syndrome type I. Neurosci Lett. 2004. 356:87–90.
33. Yoon YW, Dong H, Arends JJ, Jacquin MF. Mechanical and cold allodynia in a rat spinal cord contusion model. Somatosens Mot Res. 2004. 21:25–31.
34. Choi Y, Yoon YW, Na HS, Kim SH, Chung JM. Behavioral signs of ongoing pain and cold allodynia in a rat model of neuropathic pain. Pain. 1994. 59:369–376.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download