1. Shoback D. Clinical practice. hypoparathyroidism. N Engl J Med. 2008; 359:391–403.
2. Yi HS, Choi B, Lee S. Molecular understanding and assessment of hypoparathyroidism. Endocrinol Metab. 2011; 26:25–32.
3. Marx SJ. Hyperparathyroid and hypoparathyroid disorders. N Engl J Med. 2000; 343:1863–1875.
4. Kobrynski LJ, Sullivan KE. Velocardiofacial syndrome, DiGeorge syndrome: the chromosome 22q11.2 deletion syndromes. Lancet. 2007; 370:1443–1452.
5. Husebye ES. Functional autoantibodies cause hypoparathyroidism. J Clin Endocrinol Metab. 2009; 94:4655–4657.
6. Masi L, Del Monte F, Gozzini A, De Feo ML, Gionata Gheri R, Neri A, Falchetti A, Amedei A, Imbriaco R, Mavilia C, et al. A novel polymorphism at the GNAS1 gene associated with low circulating calcium levels. Clin Cases Miner Bone Metab. 2007; 4:139–145.
7. Raue F, Pichl J, Dörr HG, Schnabel D, Heidemann P, Hammersen G, Jaursch-Hancke C, Santen R, Schöfl C, Wabitsch M, et al. Activating mutations in the calcium-sensing receptor: genetic and clinical spectrum in 25 patients with autosomal dominant hypocalcaemia - a German survey. Clin Endocrinol (Oxf). 2011; 75:760–765.
8. Arnold A, Horst SA, Gardella TJ, Baba H, Levine MA, Kronenberg HM. Mutation of the signal peptide-encoding region of the preproparathyroid hormone gene in familial isolated hypoparathyroidism. J Clin Invest. 1990; 86:1084–1087.
9. Thakker RV. Genetic developments in hypoparathyroidism. Lancet. 2001; 357:974–976.
10. Bilezikian JP, Khan A, Potts JT Jr, Brandi ML, Clarke BL, Shoback D, Jüppner H, D'Amour P, Fox J, Rejnmark L, et al. Hypoparathyroidism in the adult: epidemiology, diagnosis, pathophysiology, target-organ involvement, treatment, and challenges for future research. J Bone Miner Res. 2011; 26:2317–2337.
11. Yi HS, Eom YS, Park IB, Lee S, Hong S, Jüppner H, Mannstadt M, Lee S. Identification and characterization of C106R, a novel mutation in the DNA-binding domain of GCMB, in a family with autosomal-dominant hypoparathyroidism. Clin Endocrinol (Oxf). 2012; 76:625–633.
12. Ku CS, Cooper DN, Polychronakos C, Naidoo N, Wu M, Soong R. Exome sequencing: dual role as a discovery and diagnostic tool. Ann Neurol. 2012; 71:5–14.
13. Park SY, Mun HC, Eom YS, Baek HL, Jung TS, Kim CH, Hong S, Lee S. Identification and characterization of D410E, a novel mutation in the loop 3 domain of CASR, in autosomal dominant hypocalcemia and a therapeutic approach using a novel calcilytic, AXT914. Clin Endocrinol (Oxf). 2013; 78:687–693.
14. Kim MY, Tan AH, Ki CS, Lee JI, Jang HW, Shin HW, Kim SW, Min YK, Lee MS, Lee MK, et al. Autosomal dominant hypocalcemia caused by an activating mutation of the calcium-sensing receptor gene: the first case report in Korea. J Korean Med Sci. 2010; 25:317–320.
15. Maret A, Ding C, Kornfield SL, Levine MA. Analysis of the GCM2 gene in isolated hypoparathyroidism: a molecular and biochemical study. J Clin Endocrinol Metab. 2008; 93:1426–1432.
16. Bamshad MJ, Ng SB, Bigham AW, Tabor HK, Emond MJ, Nickerson DA, Shendure J. Exome sequencing as a tool for Mendelian disease gene discovery. Nat Rev Genet. 2011; 12:745–755.
17. Goswami R, Brown EM, Kochupillai N, Gupta N, Rani R, Kifor O, Chattopadhyay N. Prevalence of calcium sensing receptor autoantibodies in patients with sporadic idiopathic hypoparathyroidism. Eur J Endocrinol. 2004; 150:9–18.
18. Tomar N, Bora H, Singh R, Gupta N, Kaur P, Chauhan SS, Sharma YD, Goswami R. Presence and significance of a R110W mutation in the DNA-binding domain of GCM2 gene in patients with isolated hypoparathyroidism and their family members. Eur J Endocrinol. 2010; 162:407–421.
19. Yun FH, Wong BY, Chase M, Shuen AY, Canaff L, Thongthai K, Siminovitch K, Hendy GN, Cole DE. Genetic variation at the calcium-sensing receptor (CASR) locus: implications for clinical molecular diagnostics. Clin Biochem. 2007; 40:551–561.
20. Cole DE, Peltekova VD, Rubin LA, Hawker GA, Vieth R, Liew CC, Hwang DM, Evrovski J, Hendy GN. A986S polymorphism of the calcium-sensing receptor and circulating calcium concentrations. Lancet. 1999; 353:112–115.
21. Cole DE, Vieth R, Trang HM, Wong BY, Hendy GN, Rubin LA. Association between total serum calcium and the A986S polymorphism of the calcium-sensing receptor gene. Mol Genet Metab. 2001; 72:168–174.
22. Bollerslev J, Wilson SG, Dick IM, Devine A, Dhaliwal SS, Prince RL. Calcium-sensing receptor gene polymorphism A986S does not predict serum calcium level, bone mineral density, calcaneal ultrasound indices, or fracture rate in a large cohort of elderly women. Calcif Tissue Int. 2004; 74:12–17.
23. Scillitani A, Guarnieri V, De Geronimo S, Muscarella LA, Battista C, D'Agruma L, Bertoldo F, Florio C, Minisola S, Hendy GN, et al. Blood ionized calcium is associated with clustered polymorphisms in the carboxyl-terminal tail of the calcium-sensing receptor. J Clin Endocrinol Metab. 2004; 89:5634–5638.
24. Kapur K, Johnson T, Beckmann ND, Sehmi J, Tanaka T, Kutalik Z, Styrkarsdottir U, Zhang W, Marek D, Gudbjartsson DF, et al. Genome-wide meta-analysis for serum calcium identifies significantly associated SNPs near the calcium-sensing receptor (CASR) gene. PLoS Genet. 2010; 6:e1001035.
25. Egbuna OI, Brown EM. Hypercalcaemic and hypocalcaemic conditions due to calcium-sensing receptor mutations. Best Pract Res Clin Rheumatol. 2008; 22:129–148.
26. Choi HS, Oh HJ, Choi H, Choi WH, Kim JG, Kim KM, Kim KJ, Rhee Y, Lim SK. Vitamin D insufficiency in Korea--a greater threat to younger generation: the Korea National Health and Nutrition Examination Survey (KNHANES) 2008. J Clin Endocrinol Metab. 2011; 96:643–651.
27. Breslau NA. Normal and abnormal regulation of 1,25-(OH)2D synthesis. Am J Med Sci. 1988; 296:417–425.
28. Nakajima K, Yamazaki K, Kimura H, Takano K, Miyoshi H, Sato K. Novel gain of function mutations of the calcium-sensing receptor in two patients with PTH-deficient hypocalcemia. Intern Med. 2009; 48:1951–1956.
29. Nakamura Y, Matsumoto T, Tamakoshi A, Kawamura T, Seino Y, Kasuga M, Yanagawa H, Ohno Y. Prevalence of idiopathic hypoparathyroidism and pseudohypoparathyroidism in Japan. J Epidemiol. 2000; 10:29–33.
30. Mitchell DM, Regan S, Cooley MR, Lauter KB, Vrla MC, Becker CB, Burnett-Bowie SA, Mannstadt M. Long-term follow-up of patients with hypoparathyroidism. J Clin Endocrinol Metab. 2012; 97:4507–4514.
31. Li Y, Song YH, Rais N, Connor E, Schatz D, Muir A, Maclaren N. Autoantibodies to the extracellular domain of the calcium sensing receptor in patients with acquired hypoparathyroidism. J Clin Invest. 1996; 97:910–914.






 PDF
PDF ePub
ePub Citation
Citation Print
Print


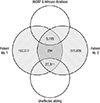

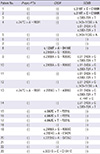
 XML Download
XML Download