Abstract
We investigated the role of fasting hormones and pro-inflammatory cytokines in cancer patients. Hormones (ghrelin, adiponectin, and leptin) and cytokines (TNF-α, IFN-γ, and IL-6) were measured by ELISA or RIA in lung cancer and colorectal cancer patients before the administration of cancer therapy, and measurements were repeated every 2 months for 6 months. From June 2006 to August 2008, 42 patients (19 with colorectal cancer and 23 with lung cancer) were enrolled. In total, 21 patients were included in the cachexia group and the others served as a comparison group. No significant difference in the initial adiponectin, ghrelin, TNF-α, IFN-γ, or IL-6 level was observed between groups, although leptin was significantly lower in cachectic patients than in the comparison group (15.3 ± 19.5 vs 80.9 ± 99.0 pg/mL, P = 0.007). During the follow-up, the patients who showed a > 5% weight gain had higher ghrelin levels after 6 months. Patients exhibiting elevated IL-6 levels typically showed a weight loss > 5% after 6 months. A blunted adiponectin or ghrelin response to weight loss may contribute to cancer cachexia and IL-6 may be responsible for inducing and maintaining cancer cachexia.
Anorexia is a condition in which a person has little or no appetite, which results in decreased consumption of food. It is often related to a more serious health problem such as cancer, AIDS or an emotional disorder. Cachexia is a syndrome characterized by weight loss, lipolysis, muscle wasting, anorexia, chronic nausea, and asthenia, with resultant changes in body image. The definition of cachexia varies, but it is generally accepted as a weight loss of 5% from pre-illness weight or a weight loss of 25% over 2 to 6 months (1). Cancer anorexia-cachexia syndrome (CACS) is the most common paraneoplastic syndrome and is regarded as an indicator of poor prognosis. Half of all cancer patients experience this syndrome to a mild degree, and it accounts for more than 20% of all cancer deaths (2). The mechanism of CACS is different from that of starvation; however, it is poorly understood and complicated by multiple cancer-associated processes, including the secretion of tumor-derived factors, the host inflammatory response, changes in cytokine levels, and treatment-induced anorexia.
Food intake and energy homeostasis are regulated by a complex network of peripheral mediators, such as hormones, neuropeptides, and cytokines. Ghrelin stimulates food intake and ghrelin levels are positively correlated with cachectic states (3-5). Adiponectin is inversely correlated with body weight (6), and leptin acts in the central nervous system (CNS) to suppress food intake and stimulate energy expenditure (7, 8). Many host-derived inflammatory mediators that participate in cancer cachexia have been identified; these include tumor necrosis factor-α (TNF-α), interleukin-1 (IL-1), interleukin-6 (IL-6), and interferon-γ (IFN-γ) (9). However, elevated levels of these cytokines are rarely found in the blood of cancer patients, and regulation by other mediators in these patients has not been demonstrated. The relationship between these hormones or cytokines and weight loss in cancer patients has not been clearly established. The objectives of our study were to investigate 1) the role of adiponectin, leptin, ghrelin, TNF-α, IL-6, and IFN-γ in cancer cachexia, 2) the association between hormones and cytokines in cachexia, and 3) the relationship between the clinical manifestations of cachexia and the levels of adiponectin, leptin, ghrelin, TNF-α, IL-6, and IFN-γ in patients with newly diagnosed colon or lung cancer.
The study population consisted of newly diagnosed colorectal or lung cancer patients from June 2006 to January 2008. The inclusion criteria were: 1) pathologically confirmed colorectal or lung cancer, 2) age over 18 yr, and 3) a consent form signed and dated before the study. Patients who had been treated with chemotherapy or radiotherapy or had undergone a major operation within 3 months, had a history of gastrectomy, an eating disorder, gastrointestinal obstruction, brain metastasis, secondary cancer in another organ, or another primary cachectic state (i.e., congestive heart failure, chronic obstructive pulmonary disease, or liver cirrhosis) were excluded. The patients were divided into cachexia and non-cachexia groups, with cachexia defined as a weight loss of ≥ 5% from pre-illness weight or over 6 months.
Blood samples were collected in the morning after overnight fasting for the measurement of adiponectin, ghrelin, leptin, TNF-α, IL-6, and IFN-γ, and for complete blood counts and serum chemistry, including lipid profiles. This sampling was not permitted when the infection was suspected by the physician. Quantikine® enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems, Minneapolis, MN, USA) were used to determine the serum levels of adiponectin, leptin, TNF-α, IL-6, and IFN-γ, and a radioimmunoassay (RIA) kit (Linco Research, St. Charles, MO, USA) was used to determine the serum levels of ghrelin. The lower limits of sensitivity for the assays for adiponectin, ghrelin, leptin, TNF-α, IL-6, and IFN-γ were 0.246 ng/mL, 93 pg/mL, 7.8 pg/mL, 1.6 pg/mL, 0.70 pg/mL, and 8.0 pg/mL, respectively. All laboratory assays were repeated at 2, 4, and 6 months after the initial measurement.
Continuous variables between the study groups were compared using the t-test, the Mann-Whitney U-test, or analysis of variance (ANOVA), and Pearson's chi-square or Fisher's exact test was used for categorical variables. Kaplan-Meier analysis with a log-rank test was used for survival analysis and to identify factors affecting survival. All significance tests were two-tailed.
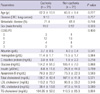
From June 2006 to January 2008, 42 patients were prospectively enrolled. Among these, 16 patients were fully evaluable for 6 months, whereas 20 patients had expired and six patients were transferred to another hospital within the follow-up period. Therefore, we analyzed 42 patients at baseline, 28 patients at 2 months, 24 patients at 4 months, and 16 patients at 6 months. The patients' demographic and clinical characteristics are summarized in Table 1. Of the 42 patients enrolled, 19 patients had colorectal cancer (CRC) and the rest were lung cancer patients. Half of the patients had cachexia, and the rest of the patients (n = 21) were non-cachectic. No significant differences were observed in age, type of cancer, rate of metastatic disease, performance status, lipid profile, or the levels of hemoglobin (Hb), C-reactive protein (CRP), glucose, insulin, or apoprotein B between the cachexia and non-cachexia groups. A higher percentage of patients in the cachexia group were male than in the non-cachexia group (90.5 vs 42.8%, P = 0.003), and albumin levels were significantly lower within the cachexia group compared to the non-cachexia group (3.7 ± 0.5 vs 4.0 ± 0.4 g/dL, respectively; P = 0.041).
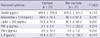
The mean levels of ghrelin and adiponectin were 669.9 ± 299.4 pg/mL and 98.6 ± 59.4 ng/mL in the cachexia group, and 628.6 ± 416.0 pg/mL and 88.4 ± 61.4 ng/mL in the non-cachexia group, respectively, and no significant difference was observed between the two groups. Leptin levels were significantly lower in the cachexia group compared to the non-cachexia group (15.3 ± 19.5 vs 80.9 ± 99.0 pg/mL, respectively; P = 0.007). The ranges of proinflammatory cytokines varied widely, and the mean levels of TNF-α, IFN-γ, and IL-6 were not significantly different between the cachexia and non-cachexia groups (Table 2).
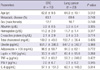
Clinical characteristics, such as age, rate of metastatic disease, sex, albumin, Hb, CRP, and cholesterol levels, did not differ between CRC and lung cancer patients. In addition, no significant difference in the level of ghrelin, adiponectin, leptin, TNF-α, IL-6, or IFN-γ was observed between patients with CRC and those with lung cancer (Table 3).
After 2 months, one patient experienced a weight gain of > 5%, and eight patients experienced weight losses > 5% compared to their state at enrollment. We compared hormone and cytokine levels between those who lost weight and those who showed no change, excluding the patient who gained weight. Between these two groups, no significant difference in ghrelin, adiponectin, leptin, TNF-α, IL-6, or IFN-γ levels was observed.
After 4 months, five patients experienced weight gains > 5%, and six patients experienced weight losses > 5% compared to their state at enrollment. The levels of ghrelin, adiponectin, and leptin at enrollment and 4 months later did not differ between those who gained weight, those who lost weight, and those whose weight was unchanged. The levels of TNF-α and IFN-γ did not differ significantly at baseline or 4 months later between the three groups. IL-6 levels showed an increase at 4 months in the weight loss group compared to the unchanged and weight gain groups, but the difference was not statistically significant (113.6 ± 261.5 vs 33.2 ± 80.6 vs 9.4 ± 9.6 pg/mL, respectively; P = 0.421; Table 4).
After 6 months, three patients experienced weight gains > 5%, and three patients experienced weight losses > 5%, compared to their state at enrollment. We found that ghrelin levels were significantly higher in patients showing weight gain compared to the unchanged and weight loss groups (1,223.1 ± 383.7 vs 594.2 ± 218.1 vs 654.3 ± 218.1 pg/mL, respectively; P = 0.007). No significant difference was detected in the levels of adiponectin, leptin, TNF-α, or IFN-γ at enrollment or 6 months later or from the time of enrollment to 6 months later among the three weight groups. The baseline IL-6 level was higher in the weight loss group than in the unchanged or weight gain group (44.6 ± 44.7 vs 12.1 ± 10.8 vs 4.1 ± 4.9 pg/mL, respectively; P = 0.048). After 6 months, the level of IL-6 showed an overall increase in the weight loss group compared to the unchanged and weight gain groups, but the difference was not statistically significant (217.2 ± 372.6 vs 56.7 ± 143.9 vs 32.2 ± 38.4 pg/mL, respectively; P = 0.410; Table 5).
No significant prognostic factor for survival was identified through our Kaplan-Meier survival analysis of ghrelin, adiponectin, leptin, TNF-α, IFN-γ, and IL-6 levels (Fig. 1).
CACS is associated with poor prognosis, including an increased risk of postoperative complications, impaired immune and pulmonary function, impaired tolerance to anti-neoplastic treatment, decreased quality of life, and shortened survival. Although the incidence of cancer cachexia varies among cancer types, 50% of all cancer patients experience this syndrome to a mild degree and 15% experience a loss of greater than 10% of baseline body weight. At the time of diagnosis, 80% of patients with upper gastrointestinal cancer and 60% of patients with lung cancer already demonstrate substantial weight loss (10). In addition, cancer treatment and medication for concurrent conditions may contribute to cancer cachexia. Cancer cachexia differs from malnutrition in starvation (11). The most common form of nutritional depletion in cancer cachexia is protein-calorie malnutrition, resulting in the loss of cell mass without depletion of vitamins or trace minerals (11). The existence of mediators in cancer cachexia that differ from those observed in starvation has been recognized. For example, the ventromedial hypothalamus (VMH), the serotonergic system, TNF-α, IL-6, IFN-γ, neuropeptide Y, nitric oxide, and leptin have been identified as central mediators of cachexia in animal studies (9). However, the mechanisms of cancer cachexia are not well defined, and no consensus has been reached regarding the role of mediators such as cytokines in cancer cachexia until now. Together with cytokines, hormones have a significant role in food intake and energy homeostasis; however, the pathophysiological role of these hormones has not been fully elucidated in cancer cachexia.
Ghrelin has growth hormone-releasing activity, stimulates food intake, induces adiposity, and inhibits leptin-induced reduction in feeding. In previous reports, ghrelin levels were positively correlated with decreased body mass index, and were higher in lung cancer patients than in healthy individuals (12). However, in the present study, no significant difference in ghrelin levels was observed between the cachexia and non-cachexia groups at baseline, although patients with weight gain at later time points showed significantly higher ghrelin levels than other groups. These results may indicate an impaired ghrelin response in cancer patients with cachexia. However, ghrelin may have potential as a treatment for cancer cachexia. Studies in a rat model demonstrated positive effects after ghrelin treatment, which may have involved orexigenic neuropeptides and anti-inflammatory (13). Thus, a clinical trial involving the administration of ghrelin in human cancer cachexia may be warranted.
Adiponectin levels are inversely correlated with body weight, and therefore low adiponectin levels are typically observed in obese individuals (14). However, low adiponectin levels have been reported in individuals who experienced weight loss with advanced lung cancer (15). We did not observe an inverse correlation between body weight change and adiponectin levels; adiponectin levels did not differ significantly between the cachexia and non-cachexia groups. We observed no significant difference or change in adiponectin levels with weight change during follow-up. Thus, the impaired regulation of adiponectin with weight change may contribute to cancer cachexia, and further study is required to determine the pathophysiological role of adiponectin in cancer cachexia.
Leptin is known to act in the CNS to suppress food intake and stimulate energy expenditure; however, no consistent relationship has been demonstrated between leptin and cancer cachexia. Existing data on the association between leptin levels and cancer are contradictory. Leptin levels were reportedly low in gastrointestinal cancer patients, but high in those with breast cancer (16-18). In our study, leptin was significantly suppressed in the cachexia group compared to the non-cachexia group; therefore, we suggest that leptin may be regulated normally in cancer cachexia, unlike adiponectin and ghrelin. We also found no significant difference in leptin levels between patients with CRC and those with lung cancer. Leptin and adiponectin were reported to be down-regulated by TNF-α in cachexia (19, 20), and ghrelin suppresses production of cytokines such as IL-6 and TNF-α (21, 22), but we did not find any reverse correlation between TNF-α and leptin or ghrelin. We found no significant difference in cytokines between the cachexia and non-cachexia groups at baseline measurement; however, the wide range of values observed may have resulted in statistical bias. Importantly, our results indicated that high IL-6 levels at baseline were maintained and increased in patients who later experienced weight loss. We suggest that IL-6 may be responsible for inducing and maintaining cancer cachexia. These results are supported by the finding of Baltgalvis et al. (23) that IL-6 overexpression in ApcMin/+/IL-6-/- mice led to a decrease in muscle and fat mass and increased the intestinal polyp burden.
We also used Kaplan-Meier analysis to investigate the role of hormones and cytokines as predictive and prognostic markers. According to this analysis, ghrelin, adiponectin, leptin, TNF-α, IFN-γ, and IL-6 levels are not significant predictive factors of survival. However, patients with high ghrelin, low adiponectin, high leptin, high TNF-α, or low IL-6 had a tendency toward prolonged survival, although it did not cross the threshold for statistical significance in the survival curve. Therefore, further studies are needed to determine the roles of these hormones and cytokines as prognostic markers.
The limitations of this study include a small number of patients, which may have resulted in statistical errors, particularly in the follow-up testing. Nevertheless, the results of this prospective study may contribute to the understanding of the pathogenesis and treatment of cancer cachexia.
In summary, we found that 1) leptin levels decreased significantly in patients with cachexia compared to those without, but ghrelin and adiponectin levels were not significantly altered; 2) ghrelin levels were significantly higher in patients with weight gain, but leptin and adiponectin did not change significantly with weight change; 3) patients who experienced weight loss showed increased IL-6 levels at baseline and throughout the follow-up compared to the weight gain and no change groups; and 4) no significant prognostic factor for survival was identified among the hormones and cytokines examined.
In conclusion, these results suggest that impaired responsiveness of adiponectin and ghrelin may contribute to cancer cachexia and that IL-6 may be responsible for inducing and maintaining cancer cachexia. Furthermore, our data suggest that a clinical trial for the administration of ghrelin in cancer cachexia treatment may be warranted.
Figures and Tables
References
1. Abeloff MD, Armitage JO, Niederhuber JE, Kastan MB, McKenna WG. Clinical oncology. 2004. 3rd ed. Philadelphia: Elsevier;749–757.
2. Tisdale MJ. Cancer cachexia. Langenbecks Arch Surg. 2004. 389:299–305.
3. Tschöp M, Smiley DL, Heiman ML. Ghrelin induces adiposity in rodents. Nature. 2000. 407:908–913.
4. Nagaya N, Uematsu M, Kojima M, Date Y, Nakazato M, Okumura H, Hosoda H, Shimizu W, Yamagishi M, Oya H, Koh H, Yutani C, Kangawa K. Elevated circulating level of ghrelin in cachexia associated with chronic heart failure: relationships between ghrelin and anabolic/catabolic factors. Circulation. 2001. 104:2034–2038.
5. Cummings DE, Weigle DS, Frayo RS, Breen PA, Ma MK, Dellinger EP, Purnell JQ. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med. 2002. 346:1623–1630.
6. Staiger H, Tschritter O, Machann J, Thamer C, Fritsche A, Maerker E, Schick F, Häring HU, Stumvoll M. Relationship of serum adiponectin and leptin concentrations with body fat distribution in humans. Obes Res. 2003. 11:368–372.
7. Meier U, Gressner AM. Endocrine regulation of energy metabolism: review of pathobiochemical and clinical chemical aspects of leptin, ghrelin, adiponectin, and resistin. Clin Chem. 2004. 50:1511–1525.
8. Havel PJ. Peripheral signals conveying metabolic information to the brain: short-term and long-term regulation of food intake and energy homeostasis. Exp Biol Med (Maywood). 2001. 226:963–977.
9. Argilés JM, Busquets S, López-Soriano FJ. Cytokines as mediators and targets for cancer cachexia. Cancer Treat Res. 2006. 130:199–217.
10. Bruera E. ABC of palliative care. Anorexia, cachexia, and nutrition. BMJ. 1997. 315:1219–1222.
11. DeVita VT, Lawrence TS, Rosenberg SA. Cancer principles and practice of oncology. 2008. 8th ed. Philadelphia: Lippincott Williams & Wilkins;2649–2661.
12. Karapanagiotou EM, Polyzos A, Dilana KD, Gratsias I, Boura P, Gkiozos I, Syrigos KN. Increased serum levels of ghrelin at diagnosis mediate body weight loss in non-small cell lung cancer (NSCLC) patients. Lung Cancer. 2009. 66:393–398.
13. DeBoer MD, Zhu XX, Levasseur P, Meguid MM, Suzuki S, Inui A, Taylor JE, Halem HA, Dong JZ, Datta R, Culler MD, Marks DL. Ghrelin treatment causes increased food intake and retention of lean body mass in a rat model of cancer cachexia. Endocrinology. 2007. 148:3004–3012.
14. Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, Miyagawa J, Hotta K, Shimomura I, Nakamura T, Miyaoka K, Kuriyama H, Nishida M, Yamashita S, Okubo K, Matsubara K, Muraguchi M, Ohmoto Y, Funahashi T, Matsuzawa Y. Paradoxical decrease of an adipose specific-protein, adiponectin, in obesity. Biochem Biophys Res Commun. 1999. 257:79–83.
15. Jamieson NB, Brown DJ, Michzel Wallace A, McMillan DC. Adiponectin and the systemic inflammatory response in weight-losing patients with non-small cell lung cancer. Cytokine. 2004. 27:90–92.
16. Wallace AM, Kelly A, Sattar N, McArdle CS, McMillan DC. Circulating concentrations of "free" leptin in relation to fat mass and appetite in gastrointestinal cancer patients. Nutr Cancer. 2002. 44:157–160.
17. Bolukbas FF, Kilic H, Bolukbas C, Gumus M, Horoz M, Turhal NS, Kavakli B. Serum leptin concentration and advanced gastrointestinal cancers: a case controlled study. BMC Cancer. 2004. 4:29.
18. Wu MH, Chou YC, Chou WY, Hsu GC, Chu CH, Yu CP, Yu JC, Sun CA. Circulating levels of leptin, adiposity and breast cancer risk. Br J Cancer. 2009. 100:578–582.
19. Wang B, Jenkins JR, Trayhurn P. Expression and secretion of inflammation-related adipokines by human adipocytes differentiated in culture: integrated response to TNF-alpha. Am J Physiol Endocrinol Metab. 2005. 288:E731–E740.
20. Machado AP, Costa Rosa LF, Seelaender MC. Adipose tissue in Walker 256 tumour-induced cachexia: possible association between decreased leptin concentration and mononuclear cell infiltration. Cell Tissue Res. 2004. 318:503–514.
21. Neary NM, Small CJ, Wren AM, Lee JL, Druce MR, Palmieri C, Frost GS, Ghatei MA, Coombes RC, Bloom SR. Ghrelin increases energy intake in cancer patients with impaired appetite: acute, randomized, placebo-controlled trial. J Clin Endocrinol Metab. 2004. 89:2832–2836.
22. Hanada T, Toshinai K, Kajimura N, Nara-Ashizawa N, Tsukada T, Hayashi Y, Osuye K, Kangawa K, Matsukura S, Nakazato M. Anti-cachectic effect of ghrelin in nude mice bearing human melanoma cells. Biochem Biophys Res Commun. 2003. 301:275–279.
23. Baltgalvis KA, Berger FG, Pena MM, Davis JM, Muga SJ, Carson JA. Interleukin-6 and cachexia in ApcMin/+ mice. Am J Physiol Regul Integr Comp Physiol. 2008. 294:R393–R401.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download