Abstract
Congenital central hypoventilation syndrome with Hirschsprung's disease, also known as Haddad syndrome, is an extremely rare disorder with variable symptoms. Recent studies described that congenital central hypoventilation syndrome had deep relation to the mutation of the PHOX2B gene in its diagnosis and phenotype. We report a newborn male infant with clinical manifestations of recurrent hypoventilation with hypercapnea and bowel obstruction. These clinical manifestations were compatible with congenital central hypoventilation syndrome and Hirschsprung's disease, and polyalanine 26 repeats in the PHOX2B gene supported the diagnosis of congenital central hypoventilation. We described a first case of Haddad syndrome in Korean and its clinical and genetic characteristics were discussed.
Congenital central hypoventilation syndrome (CCHS) is a life-threatening disorder characterized by an idiopathic failure of the automatic control of breathing, particularly during sleep. In CCHS, patients breathe with insufficient depth due to impaired sensitivity to hypercapnea and hypoxia. Hirschsprung's disease (HD), also known as congenital aganglionic megacolon, is characterized by the absence of ganglion cells in any part of the bowel wall. The absence of ganglion cells in Hirschsprung's disease inhibits normal peristalsis, resulting in a functional intestinal obstruction. The combination of CCHS and HD was reported first by Haddad in 1978 and it has been extremely rare with approximately 60 cases reported in the worldwide literature (1, 2).
Previous studies described that de novo mutation of the PHOX2B gene was involved in CCHS pathogenesis (3, 4). Many patients with CCHS are heterozygous for a polyalanine expansion mutation in the second polyalanine repeat residue of the PHOX2B gene, with one allele having 20 repeats and the affected allele having 25-33 repeats of the polyalanine sequence (3, 4). Some authors reported that a subset of patients had other mutations in the PHOX2B gene (5, 6). In Korean population, only one case has been reported, however, genetic analysis has not been performed (7). Here, we report a Haddad syndrome with the PHOX2B gene mutation for the first time in a Korean.
This 41-week-gestation male was born to a 30-yr-old mother with good health by normal spontaneous vaginal delivery in November 2009. His birth weight was 3,140 gm. There was no maternal complication during pregnancy. There was neither family history of congenital anomalies nor maternal exposure to drugs during pregnancy. The baby had APGAR scores of 7 and 5 at 1 and 5 min, respectively. He needed positive pressure ventilation with intubation because of progressive respiratory difficulties and hypoxia. The baby was immediately transferred to our hospital for further evaluation and management after stabilization.
The physical examination and laboratory findings for the baby were unremarkable on admission. He had the surfactant replacement therapy for acute respiratory failure. For the next few days, we tried extubation several times, but failed to succeed because the baby suffered from recurrent apnea and hypercarbia leading to the clinical suspicious of CCHS. On day 8 of life, a cranial ultrasound study was performed and it did not show evidence of hemorrhage or mass lesions.
During the second day of life, it was noted that the patient had distended abdomen and was unable to pass meconium without rectal stimulation. A gastrografin contrast enema study showed multiple meconium plugs in the colon suggesting meconium plug syndrome (Fig. 1A). Meconium was passed and abdominal distension was relieved after contrast study. On day 6 of life, he showed sustained abdominal distension with poor bowel movement. A barium contrast enema study was needed to rule out other causes of lower gastrointestinal obstruction. It showed a transition zone in rectosigmoid colon suggesting HD. The diagnosis was confirmed by rectal suction biopsy with acetylcholinesterase immunohistochemistry (Fig. 1B, C). The infant underwent an open laparotomy on day 12. He was noted to have long segment HD with a transition zone at mid descending colon. Seromuscular biopsies of the bowel taken 3cm proximal and distal to the transition zone revealed the presence of ganglion cells in the proximal biopsy specimen only. A descending loop colostomy was done at the 2 cm from proximal biopsy site. After the diagnosis, the patient was transferred to another hospital for the treatment. The patient had a tracheostomy to provide the necessary assisted ventilation. Laparoscopic Pull through Procedure was carried out for the treatment of HD. After the discharge, he has been followed up until now.
The cytogenetic study was performed clinically in the infant and his parents and their karyotypes were normal. Genomic DNA of the patient and his parents was isolated from peripheral blood using the Wizard Genomic DNA Purification Kit according to the manufacturer's instructions (Promega, Madison, WI, USA). Amplification of the coding sequence of the PHOX2B gene by polymerase chain reaction (PCR) was carried out as previously described with minor modifications (4). Polymerase chain reaction-single strand conformational polymorphism (PCR-SSCP) was performed as previously described (8). PCR products in the patient, his parents, and normal controls were electrophoresed through 8 and 10% polyacryamide gels (Fig. 2). In patient, a mutant band was found, however, his parents had no mutant band. To determine the sequences of the DNA sample showing the mutant band, direct sequencing was performed by Macrogen Inc, in Korea. However, the quality of the result was not clear. To get apparent result of sequencing, PCR products were cloned by TOPO TA cloning (Invitrogen Ltd., Paisley, United Kingdom) and 10 single colonies were collected for extracting plasmid DNA. Direct sequencing revealed expanded alleles containing polyalanine 26 repeats in exon 3 of the PHOX2B gene, further supporting the diagnosis CCHS (Fig. 3).
We report here a neonatal case of Haddad syndrome in Korean. Importantly, we were able to prove a PHOX2B gene mutation containing polyalanine 26 repeats in this patient. Hirschsprung's disease often produces intestinal obstruction in the neonate, characterized by abdominal distention, bilious vomiting, and failure to pass meconium within the first 48 hr. CCHS is characterized by relatively normal ventilation when the infant is awake and hypoventilation, even apnea, during sleep. Haddad syndrome is interpreted not so much as a combination of two diseases but rather as a complicated form of CCHS. When HD is associated with CCHS, it is different from classical HD, characterized by extensive intestinal aganglionosis (9). The incidence of CCHS is extremely low in HD cases, however, the incidence of HD in CCHS cases varies from 16% to 50% according to previous studies (3, 5, 6, 9). Additional symptoms are ophthalmic abnormalities, esophageal dysmotility, sensorineural hearing loss, neural crest tumors (ganglioneuromas, neuroblastomas), and dysmorphic facial features (3, 5, 9). However, additional symptoms were not found in our case. According to previous report in Korean, a Haddad syndrome patient also had no additional symptoms except congenital ptosis (7).
Genetic characteristics of CCHS have been widely investigated. The PHOX2B gene mutation in CCHS was first demonstrated by Amiel et al. (3), and its rate was over 90% among CCHS patients (4, 6). Polyalanine expansions are the most common mutations in patients with CCHS, though rare mutations have been reported such as frameshift mutation, nonsense mutation or missense mutation (3-5). In our case, his parents showed no PHOX2B gene mutation, suggesting de novo mutation of the PHOX2B gene in Haddad syndrome. Interestingly, genotype-phenotype correlation has been described in CCHS patients with the PHOX2B gene mutation (4). Increasing polyalanine repeat mutation size is associated with a more severe clinical phenotype (4). Trochet et al. (10) described that Haddad syndrome (CCHS + HD type) showed usually expansions of +6 and +7 alanines, and frameshift or missense PHOX2B mutations may predispose to neuroblastoma. Our patient had an expanded allele containing polyalanine 26 repeats in the PHOX2B gene. Clinical phenotype of our patient was severe, presenting long segment HD and no capacity to maintain sufficient oxygenation and ventilation for at least 30-60 min of sleep after removing mechanical ventilation support (4). However, our case with expansion of +6 alanines had no additional symptoms like neural crest tumors. Though the symptoms of CCHS and HD were severe, no additional symptom in the patient with this mutation type was in agreement with the hypothesis by Trochet et al. (10). This result supports that the assay for PHOX2B polyalanine repeat mutation represents a highly sensitive and specific technique for confirming the diagnosis of CCHS and estimating the prognosis.
In summary, this report presents a rare case of Haddad syndrome with de novo mutation of the PHOX2B gene in Korean for the first time. Considering the difficulty of diagnosis of this syndrome because of overlapped of symptoms and signs in an infant, the PHOX2B mutation test may assist the diagnosis under strong clinical suspicion. Further application of gene study is recommended for genetic counseling and prenatal diagnosis.
Figures and Tables
 | Fig. 1Diagnosis of Hirschsprung's disease. (A) Barium enema showing transitional zone (arrow) in the middle of the sigmoid colon. (B) Frozen section of the biopsy showing the absence of ganglion cells. (C) Enzyme histochemistry showing aberrant acetylcholine esterase. |
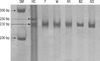 | Fig. 2The polyacrylamide gel electrophoresis of the PHOX2B gene SSCP profiles in Haddad syndrome and his family. A heterozygous mutation (232 bp and 250 bp) was found in the patient compared to the single strands in his family and normal samples (232 bp). SM, size marker; HS, Haddad syndrome; F, father; M, mother; N1, N2, and N3, normal controls. |
References
1. Haddad GG, Mazza NM, Defendini R, Blanc WA, Driscoll JM, Epstein MA, Epstein RA, Mellins RB. Congenital failure of automatic control of ventilation, gastrointestinal motility and heart rate. Medicine (Baltimore). 1978. 57:517–526.
2. Tomycz ND, Haynes RL, Schmidt EF, Ackerson K, Kinney HC. Novel neuropathologic findings in the Haddad syndrome. Acta Neuropathol. 2010. 119:261–269.
3. Amiel J, Laudier B, Attié-Bitach T, Trang H, de Pontual L, Gener B, Trochet D, Etchevers H, Ray P, Simonneau M, Vekemans M, Munnich A, Gaultier C, Lyonnet S. Polyalanine expansion and frameshift mutations of the paired-like homeobox gene PHOX2B in congenital central hypoventilation syndrome. Nat Genet. 2003. 33:459–461.
4. Matera I, Bachetti T, Puppo F, Di Duca M, Morandi F, Casiraghi GM, Cilio MR, Hennekam R, Hofstra R, Schöber JG, Ravazzolo R, Ottonello G, Ceccherini I. PHOX2B mutations and polyalanine expansions correlate with the severity of the respiratory phenotype and associated symptoms in both congenital and late onset Central Hypoventilation syndrome. J Med Genet. 2004. 41:373–380.
5. Sasaki A, Kanai M, Kijima K, Akaba K, Hashimoto M, Hasegawa H, Otaki S, Koizumi T, Kusuda S, Ogawa Y, Tuchiya K, Yamamoto W, Nakamura T, Hayasaka K. Molecular analysis of congenital central hypoventilation syndrome. Hum Genet. 2003. 114:22–26.
6. Weese-Mayer DE, Berry-Kravis EM, Zhou L, Maher BS, Silvestri JM, Curran ME, Marazita ML. Idiopathic congenital central hypoventilation syndrome: analysis of genes pertinent to early autonomic nervous system embryologic development and identification of mutations in PHOX2b. Am J Med Genet A. 2003. 123A:267–278.
7. Lee MK, Kim JS, Park SJ, Kim KS, Kim IK, Yoon CH, Kim KM. A case of Haddad syndrome. Korean J Pediatr Gastroenterol Nutr. 2005. 8:252–256.
8. Ha TW, Han KH, Son DG, Kim SP, Kim DK. Analysis of loss of heterozygosity in Korean patients with keratoacanthoma. J Korean Med Sci. 2005. 20:340–343.
9. Croaker GD, Shi E, Simpson E, Cartmill T, Cass DT. Congenital central hypoventilation syndrome and Hirschsprung's disease. Arch Dis Child. 1998. 78:316–322.
10. Trochet D, O'Brien LM, Gozal D, Trang H, Nordenskjöld A, Laudier B, Svensson PJ, Uhrig S, Cole T, Niemann S, Munnich A, Gaultier C, Lyonnet S, Amiel J. PHOX2B genotype allows for prediction of tumor risk in congenital central hypoventilation syndrome. Am J Hum Genet. 2005. 76:421–426.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download