Abstract
This study aimed to determine the incidence and characteristics of HER-2 gene heterogeneity in invasive breast cancer in a single institution. Included were 971 cases of primary invasive breast cancer diagnosed between 2008 and 2010. Fluorescence in situ hybridization (FISH) image files were retrospectively reviewed and HER-2 gene heterogeneity was defined as more than 5% but less than 50% of analyzed invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2, according to the College of American Pathologists guidelines. HER-2 gene heterogeneity was identified in 24 (2.5%) cases. The mean proportion of invasive tumor cells with a HER-2/chromosome 17 ratio higher than 2.2 was 11.6% (range: 5%-25%). Of 24 cases, HER-2 gene status was not amplified in 8, showed borderline amplification in 2, and amplification in 14. All HER-2 amplification cases were low-grade. In conclusion, HER-2 gene heterogeneity of invasive breast cancer is identified in routine FISH examination. This may affect the results of HER-2 gene amplification status in FISH studies.
HER-2 is an oncogene that is overexpressed and/or amplified in about one-third of breast cancer cases (1, 2). It is associated with tumor aggressiveness such as high tumor grade, lymph node metastasis, and brain metastasis (3-7). More importantly, HER-2 is a biomarker for targeted immunotherapy such as with trastuzumab (8-10). Therefore, the proper evaluation of HER-2 gene status is important for breast cancer therapy. In clinical practice, the standard method to examine HER-2 gene status is immunohistochemistry (IHC) or fluorescence in situ hybridization (FISH) (11-13). However, breast cancer is typically a heterogeneous tumor with intratumoral heterogeneity in not only histologic features, but also genetic features (14, 15). The incidence of HER-2 gene heterogeneity in invasive breast cancer is 1.3%-54% (16-21). However, the incidence of HER-2 gene heterogeneity in invasive breast cancer in Korea has not been studied. The purpose of this study was to investigate the incidence and characteristics of HER-2 gene heterogeneity in invasive breast cancer in routine FISH examination.
This study included 972 cases of primary invasive breast cancer diagnosed between January 2008 and December 2010. We retrospectively reviewed image files from FISH studies for the HER-2 gene that had at least three images (range 3-10) per case. Clinicopathologic parameters such as age at initial diagnosis, histologic type, histologic grade, estrogen receptor status, progesterone receptor status, and HER-2 IHC results were obtained from pathology reports. The histological grade was assessed using the Nottingham grading system (22). HER-2 staining was analyzed according to the American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) guidelines using the following categories: 0 = no immunostaining; 1+ = weak incomplete membranous staining, less than 10% of tumor cells; 2+ = complete membranous staining, either uniform or weak in at least 10% of tumor cells; and 3+ = uniform intense membranous staining in at least 30% of tumor cells (23).
Before FISH analysis, invasive tumors were examined on hematoxylin-eosin stained slides. FISH was subsequently performed on the tested tumor. FISH was performed using a PathVysion HER-2 DNA Probe Kit (Vysis, Downers Grove, IL, USA) according to the manufacturer's instructions. HER-2 gene copy number on the slides was evaluated using an epifluorescence microscope (Olympus, Tokyo, Japan). At least 60 tumor cell nuclei in three separate regions were investigated for HER-2 and chromosome 17 signals. HER-2 gene amplification was determined according to the ASCO/CAP guidelines. An absolute HER-2 gene copy number lower than 4 or a HER-2 gene/chromosome 17 copy number ratio (HER-2/Chr17 ratio, or amplification index [AI]) less than 1.8 was considered HER-2 negative. An absolute HER-2 copy number between 4 and 6 or a HER-2/Chr17 ratio between 1.8 and 2.2 was considered HER-2 equivocal. An absolute HER-2 copy number greater than 6 or a HER-2/Chr17 ratio higher than 2.2 was considered HER-2 positive (23). Chromosome 17 polysomy was defined as a centromeric chromosome 17 spot count of 3.0 or more in at least 80% of tumor cells (24). High-grade amplification was defined as a HER-2/Chr17 ratio higher than 5.0, and low-grade amplification was defined as a HER-2/Chr17 ratio greater than 2.2 but less than 5.0 (25, 26). HER-2 gene heterogeneity (GH) was defined as more than 5% but less than 50% of the analyzed invasive tumor cells having a HER-2/Chr17 ratio higher than 2.2, according to CAP guidelines (27).
Data were processed using SPSS for Windows, version 12.0 (SPSS Inc., Chicago, IL, USA). Student's unpaired t-test and Fisher's exact test were used to examine differences in continuous and categorical variables, respectively. Correlation analysis was performed by Pearson's method. Significance was assumed when P < 0.05.
Table 1 shows the clinicopathologic characteristics of the patients. Of 971 cases, HER-2 GH was observed in 24 (2.5%) cases, all of which were invasive ductal carcinoma (IDC) and showed a higher proportion of borderline or amplified HER-2 gene status than breast cancer without HER-2 GH (P < 0.001). Of 24 cases with HER-2 GH, all 14 cases with HER-2 amplification showed low-grade amplification (P < 0.001). The incidence of chromosome 17 polysomy was higher in cases with HER-2 GH than cases without (P = 0.001).
Fig. 1 shows histology, IHC, and FISH results for breast cancer with HER-2 GH. Histology was IDC, not otherwise specified (NOS) (Fig. 1A-C). Only some invasive tumor cells (arrow) showed 3+ overexpression of HER-2 (Fig. 1D-F), which was compatible with the FISH results that showed only a proportion of invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2 (Fig. 1G-I). Detailed characteristics of the 24 cases with HER-2 GH are in Table 2. HER-2 gene AI rang-ed from 1.0 to 4.6, and the HER-2 GH pattern was single cell in 22 cases and cluster pattern in 2 cases. The mean proportion of invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2 was 11.6% (range: 5%-25%), with significant correlation between the proportion of invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2 and HER-2 AI (Fig. 2; r = 0.358 and P = 0.002). The proportion of invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2 increased from the no-amplification group to the borderline group to the amplification group, but the difference was not significant (Fig. 3, P = 0.085).
This study evaluated the intratumoral heterogeneity for HER-2 in invasive breast cancer using FISH. The incidence of HER-2 GH was 2.4%, similar to the incidence of 1.3%-54% seen in previous studies (16-21). However, a simple comparison is not reasonable because study group characteristics, the number of study groups, the area evaluated by FISH, and the method of counting HER-2 signals is likely to differ among studies.
HER-2 GH can be classified into geographic heterogeneity and intercellular heterogeneity according to the method used to determine the tumor area evaluated by FISH. Geographic heterogeneity is defined as one core showing HER-2 amplification, while another shows no HER-2 amplification, when multiple tumor areas are sampled by tissue core using tissue microarray. Previous studies using this tissue microarray method showed a HER-2 GH incidences of 13%-16% (19, 21). Intercellular heterogeneity is indicated when a different HER-2 gene status is noted among invasive tumor cells evaluated in one field or focus, specifically when one microscopic field has both tumor cells with a HER-2/Chr17 ratio that is higher than 2.2 and tumor cells have a lower ratio.
In this study, image files obtained from FISH study were retrospectively reviewed, and intercellular heterogeneity rather than geographic heterogeneity was identified. A possible explanation for the lower incidence of HER-2 GH in this study could be the study method. However, we did identify HER-2 GH, even though a retrospective study design was used to examine image data from daily clinical practice. In general, breast cancer GH results from clonal diversification and differences in genetic composition from growing genetic instability in some part of the tumor (14, 28, 29). Genetic differences in a single tumor were shown by laser microdissection and a comparative genomic hybridization study, and these differences were confirmed by FISH and microsatellite instability analysis (15). Szollosi et al. (30) reported the cell-to-cell heterogeneity of HER-2 gene amplification in a FISH study of breast cancer, showing prominent heterogeneity in both the copy number of HER-2/cell and the p185HER-2 protein level, consistent with results of this study. HER-2 GH could be explained by acquisition or loss of the HER-2 gene, but HER-2 gene amplification is more plausible, considering that HER-2 is associated with tumor aggressiveness (20).
Clinically, HER-2 GH is important in the interpretation of HER-2 gene status according to FISH results and in the selection of patients for trastuzumab treatment. In this study, when HER-2 gene status was interpreted by ASCO/CAP guidelines, HER-2 GH was noted in all groups: no amplification, borderline amplification, and amplification. In addition, all cases with both HER-2 GH and HER-2 amplification showed low-grade amplification. Therefore, HER-2 GH may affect the interpretation of HER-2 amplification status results by FISH. To prevent this bias, ASCO/CAP guidelines suggest investigating at least two (and up to four) representative fields, which should be determined by scanning the entire slide (23). However, differences in the area evaluated by FISH and HER-2 signal counting method could give rise to inconsistencies in interpreting HER-2 gene status results. A previous study reported that the results of routine assessment of the HER-2 gene might be affected by breast cancer GH with low-grade amplification due to subclones of tumor cells with chromosome 17 polysomy without gene amplification (21). Another important issue in HER-2 GH is trastuzumab treatment, but whether trastuzumab treatment is potentially beneficial to patients with HER-2 GH is not known, so clinical studies are necessary. Although the clinical implications of HER-2 GH for trastuzumab treatment are uncertain, when HER-2 GH is identified, the phrase "HER-2 genetic heterogeneity is present" should be inserted in the pathology report with: 1) percentage of invasive tumor cells with HER-2 amplification; 2) pattern of amplified cells, for example scattered cells or specific clusters; and 3) pattern of specific clusters, ratio and number of chromosome enumeration probe 17 signals/cell or area and HER-2 signals/cell or area (27). In conclusion, the phenomenon of HER-2 GH in invasive breast cancer is identified in routine FISH examination, and HER-2 GH may affect the interpretation of HER-2 gene status by FISH.
Figures and Tables
Fig. 1
Breast cancer with HER-2 gene heterogeneity. The histology of breast cancer with HER-2 gene heterogeneity is invasive ductal carcinoma (H&E, × 100, A-C). Immunohistochemical stain for HER-2 shows heterogeneous 3+ overexpression (arrows, HER-2, × 200, D-F). Fluorescence in situ hybridization study shows heterogeneous invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2 (arrow, × 1,000, G-I).
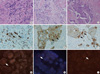
Fig. 2
Correlation between proportion of invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2 and HER-2 amplification index.

Fig. 3
Proportion of invasive tumor cells with a HER-2/Chr17 ratio higher than 2.2 according to HER-2 gene status.

AUTHOR SUMMARY
Evaluation of Intratumoral HER-2 Heterogeneity by Fluorescence In Situ Hybridization in Invasive Breast Cancer: A Single Institution Study
Sarah Lee, Woohee Jung, SoonWon Hong and Ja Seung Koo
HER-2 gene is the important biomarker for targeted immunotherapy, and the interpretation of HER-2 gene status could be affected by intratumoral HER-2 gene heterogeneity. In this study, we investigated intratumoral HER-2 gene heterogeneity in invasive breast cancer. The phenomenon of HER-2 gene heterogeneity was identified in routine FISH examination of invasive breast cancer. Further clinical studies are requested to evaluate the effectiveness of targeted immunotherapy for the patients with HER-2 gene heterogeneity.
References
1. Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987. 235:177–182.
2. Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE, Levin WJ, Stuart SG, Udove J, Ullrich A, Press MF. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989. 244:707–712.
3. Borg A, Baldetorp B, Fernö M, Killander D, Olsson H, Sigurdsson H. ERBB2 amplification in breast cancer with a high rate of proliferation. Oncogene. 1991. 6:137–143.
4. Kallioniemi OP, Holli K, Visakorpi T, Koivula T, Helin HH, Isola JJ. Association of c-erbB-2 protein over-expression with high rate of cell proliferation, increased risk of visceral metastasis and poor long-term survival in breast cancer. Int J Cancer. 1991. 49:650–655.
5. Lovekin C, Ellis IO, Locker A, Robertson JF, Bell J, Nicholson R, Gullick WJ, Elston CW, Blamey RW. c-erbB-2 oncoprotein expression in primary and advanced breast cancer. Br J Cancer. 1991. 63:439–443.
6. McCann AH, Dervan PA, O'Regan M, Codd MB, Gullick WJ, Tobin BM, Carney DN. Prognostic significance of c-erbB-2 and estrogen receptor status in human breast cancer. Cancer Res. 1991. 51:3296–3303.
7. Gusterson BA, Gelber RD, Goldhirsch A, Price KN, Säve-Söderborgh J, Anbazhagan R, Styles J, Rudenstam CM, Golouh R, Reed R. International (Ludwig) Breast Cancer Study Group. Prognostic importance of c-erbB-2 expression in breast cancer. J Clin Oncol. 1992. 10:1049–1056.
8. Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001. 344:783–792.
9. Piccart-Gebhart MJ, Procter M, Leyland-Jones B, Goldhirsch A, Untch M, Smith I, Gianni L, Baselga J, Bell R, Jackisch C, Cameron D, Dowsett M, Barrios CH, Steger G, Huang CS, Andersson M, Inbar M, Lichinitser M, Láng I, Nitz U, Iwata H, Thomssen C, Lohrisch C, Suter TM, Rüschoff J, Suto T, Greatorex V, Ward C, Straehle C, McFadden E, Dolci MS, Gelber RD. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005. 353:1659–1672.
10. Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE Jr, Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG, Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas EP, Lingle WL, Klein PM, Ingle JN, Wolmark N. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005. 353:1673–1684.
11. Pauletti G, Godolphin W, Press MF, Slamon DJ. Detection and quantitation of HER-2/neu gene amplification in human breast cancer archival material using fluorescence in situ hybridization. Oncogene. 1996. 13:63–72.
12. Bartlett JM, Going JJ, Mallon EA, Watters AD, Reeves JR, Stanton P, Richmond J, Donald B, Ferrier R, Cooke TG. Evaluating HER2 amplification and overexpression in breast cancer. J Pathol. 2001. 195:422–428.
13. Bilous M, Dowsett M, Hanna W, Isola J, Lebeau A, Moreno A, Penault-Llorca F, Rüschoff J, Tomasic G, van de Vijver M. Current perspectives on HER2 testing: a review of national testing guidelines. Mod Pathol. 2003. 16:173–182.
14. Kuukasjärvi T, Karhu R, Tanner M, Kahkönen M, Schäffer A, Nupponen N, Pennanen S, Kallioniemi A, Kallioniemi OP, Isola J. Genetic heterogeneity and clonal evolution underlying development of asynchronous metastasis in human breast cancer. Cancer Res. 1997. 57:1597–1604.
15. Aubele M, Mattis A, Zitzelsberger H, Walch A, Kremer M, Hutzler P, Höfler H, Werner M. Intratumoral heterogeneity in breast carcinoma revealed by laser-microdissection and comparative genomic hybridization. Cancer Genet Cytogenet. 1999. 110:94–102.
16. Pertschuk LP, Axiotis CA, Feldman JG, Kim YD, Karavattayhayyil SJ, Braithwaite L. Marked intratumoral heterogeneity of the proto-oncogene Her-2/neu determined by three different detection systems. Breast J. 1999. 5:369–374.
17. Glöckner S, Buurman H, Kleeberger W, Lehmann U, Kreipe H. Marked intratumoral heterogeneity of c-myc and cyclinD1 but not of c-erbB2 amplification in breast cancer. Lab Invest. 2002. 82:1419–1426.
18. Andersson J, Linderholm B, Bergh J, Elmberger G. HER-2/neu (c-erbB-2) evaluation in primary breast carcinoma by fluorescent in situ hybridization and immunohistochemistry with special focus on intratumor heterogeneity and comparison of invasive and in situ components. Appl Immunohistochem Mol Morphol. 2004. 12:14–20.
19. Shin SJ, Hyjek E, Early E, Knowles DM. Intratumoral heterogeneity of her-2/neu in invasive mammary carcinomas using fluorescence in-situ hybridization and tissue microarray. Int J Surg Pathol. 2006. 14:279–284.
20. Hanna W, Nofech-Mozes S, Kahn HJ. Intratumoral heterogeneity of HER2/neu in breast cancer: a rare event. Breast J. 2007. 13:122–129.
21. Brunelli M, Manfrin E, Martignoni G, Miller K, Remo A, Reghellin D, Bersani S, Gobbo S, Eccher A, Chilosi M, Bonetti F. Genotypic intratumoral heterogeneity in breast carcinoma with HER2/neu amplification: evaluation according to ASCO/CAP criteria. Am J Clin Pathol. 2009. 131:678–682.
22. Elston CW, Ellis IO. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology. 1991. 19:403–410.
23. Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, Allred DC, Cote RJ, Dowsett M, Fitzgibbons PL, Hanna WM, Langer A, McShane LM, Paik S, Pegram MD, Perez EA, Press MF, Rhodes A, Sturgeon C, Taube SE, Tubbs R, Vance GH, van de Vijver M, Wheeler TM, Hayes DF. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol. 2007. 25:118–145.
24. Tubbs RR, Hicks DG, Cook J, Downs-Kelly E, Pettay J, Hartke MB, Hood L, Neelon R, Myles J, Budd GT, Moore HC, Andresen S, Crowe JP. Fluorescence in situ hybridization (FISH) as primary methodology for the assessment of HER2 status in adenocarcinoma of the breast: a single institution experience. Diagn Mol Pathol. 2007. 16:207–210.
25. Ross JS, Fletcher JA. HER-2/neu (c-erb-B2) gene and protein in breast cancer. Am J Clin Pathol. 1999. 112:1 Suppl 1. S53–S67.
26. Tanner M, Gancberg D, Di Leo A, Larsimont D, Rouas G, Piccart MJ, Isola J. Chromogenic in situ hybridization: a practical alternative for fluorescence in situ hybridization to detect HER-2/neu oncogene amplification in archival breast cancer samples. Am J Pathol. 2000. 157:1467–1472.
27. Vance GH, Barry TS, Bloom KJ, Fitzgibbons PL, Hicks DG, Jenkins RB, Persons DL, Tubbs RR, Hammond ME. Genetic heterogeneity in HER2 testing in breast cancer: panel summary and guidelines. Arch Pathol Lab Med. 2009. 133:611–612.
28. Nowell PC. The clonal evolution of tumor cell populations. Science. 1976. 194:23–28.
29. Shen CY, Yu JC, Lo YL, Kuo CH, Yue CT, Jou YS, Huang CS, Lung JC, Wu CW. Genome-wide search for loss of heterozygosity using laser capture microdissected tissue of breast carcinoma: an implication for mutator phenotype and breast cancer pathogenesis. Cancer Res. 2000. 60:3884–3892.
30. Szöllösi J, Balázs M, Feuerstein BG, Benz CC, Waldman FM. ERBB-2 (HER2/neu) gene copy number, p185HER-2 overexpression, and intratumor heterogeneity in human breast cancer. Cancer Res. 1995. 55:5400–5407.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download