Abstract
We present a case of a 40-yr-old woman diagnosed with a primary malignant struma ovarii. The patient was admitted with the complaint of pelvic pain and a large pelvic mass in the mid-portion of lower abdomen on gynecological examination. Pre-operative tumor markers and routine biochemistry were unremarkable. She was treated with total abdominal hysterectomy and right salpingo-oopherectomy. Post-operatively, she was diagnosed with a malignant struma ovarii through the usage of histopathological criteria similar to the guidelines for primary thyroid gland disease. The patient was subsequently performed left salpingo-oopherectomy and retroperitoneal pelvic lympadenectomy for re-staging. Although, left ovary and lymph nodes were histopathologically normal, she was offered thyroidectomy but she refused to accept the offer. Thyroglobulin level was monitored in the post-operative period. She is free of the disease for 18 months.
Germ cell tumors are derived from the primordial germ cells of the ovary and 20-25% of all benign and malignant ovarian neoplasms are of germ cell origin (1). Vast majority of these tumors are mature teratomas.
Teratomas are composed of various tissues including hair, skin, bone, teeth, as well as thyroid. Struma ovarii is a rare monodermal variant of ovarian teratoma accounting for only 2% of all mature teratomas. To be classified as a struma ovarii, teratoma must be composed predominantly of mature thyroid tissue (>50%) (2, 3). It is a benign condition, but occasionally malignant transformation is observed in about 5% of cases (4-6). Due to its rarity, there has been controversy about the diagnosis and treatment.
We report a case of malignant struma ovarii with a focus of papillary thyroid cancer.
A 40-yr-old, multiparous woman was admitted to the Department of Gynaecology at Kocaeli University with the complaint of pelvic pain for 2 yr. Her past medical history was uneventful.
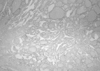
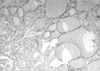
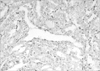
Her gynecological examination revealed a large pelvic mass in the mid-portion of lower abdomen. Abdominal ultrasonography showed a right hypoechoic mass measuring 15×20 cm suggestive of an ovarian neoplasm and a totally enlarged uteri measuring 10×6×5 cm with multiple small-sized myomas (0.5-2 cm) within the myometrium. Tumor markers and routine biochemistry were unremarkable. She was performed total abdominal hysterectomy and right salpingo-oopherectomy. Macroscopically, the right ovary was rubbery in consistency, and on a cut section it was homogeneous brown-tan in colour with areas of hemorrhage. An intraoperative frozen section was reported as a benign cystic teratoma containing thyroid tissue. Permanent sections of further sampled tumor revealed a focus of papillary thyroid carcinoma measuring 1 cm in diameter within the struma ovarii (Figs. 1, 2, 3). The tumor was totally confined within the teratomatous thyroid tissue. The diagnosis was rendered using the histopathological criteria similar to the guidelines for primary thyroid gland disease. Left salpingo-oopherectomy and retroperitoneal pelvic lympadenectomy were performed afterwards for re-staging operation. Left ovary and lymph nodes were histopathologically normal.
Her postoperative course was uneventful. After the operation, thyroid function tests and the thyroglobulin level were evaluated. Thyroid stimulating hormone was suppressed and free levels of thyroid hormones and thyroglobulin were in normal ranges. She was offered thyroidectomy for the purpose of confirmation of disease but the patient did not accept the offer.
The patient is free of disease for 18 months. Her thyroglobulin levels are within normal limits. Thyroid ultrasound scanning is normal.
Malignant struma ovarii is a rare gynecologic cancer. It may occur at any age, but the peak frequence is in the fifth and sixth decades of life. Rates of benign and malignant struma ovarii among teratomas are 2 and 0.3%, respectively (4, 8).
The most common presenting symptom is a pelvic mass. Eventhough it is a neoplasm consisting of thyroid tissue, only 8% of patients with struma ovarii present with clinical hyperthyroidism (7).
There are difficulties in the clinical diagnosis because of the absence of uniform diagnostic criteria with the rarity of the disease. The criterion for histopathological diagnosis has varied over years. Lately, Devaney et al. (9) have advocated that pathological diagnosis of malignant struma ovarii should follow the same guidelines as those for thyroid carcinomas.
Metastasis is rare in patients with malignant struma ovarii (5%). Routes of metastasis described in the literature are: 1) regional lymphatics to pelvic and paraaortic lymph nodes; 2) direct spread to the omentum and peritoneal cavity, the contralateral cavity; and 3) hematologic dissemination to the bone, lung, liver and brain (4, 10). These authors have also proposed that struma ovarii with metastatic behavior should be regarded as malignant (10, 11).
The optimum treatment of malignant struma ovarii is contraversial. Management alternatives include radical surgery with or without thyroidectomy, followed by adjuvant therapy (external radiotherapy, chemotherapy, thyroid supression) (12). Our opinion is that malignant struma ovarii should be treated with combination surgery including total abdominal hysterectomy, bilateral salpingo-oopherectomy, pelvicparaaortic lymph node sampling and thyroidectomy followed by I131 therapy. Thyroidectomy should be a part of radical surgery due to two reasons: firstly we can exclude the possibility of a primary thyroid carcinoma. Secondly after thyroidectomy, we can recognize the metastasis, recurrence and residual tissue with total body scanning with I131 and serum thyroglobulin levels. Management of the patients who desire fertility might be challenging. We agree with Dardik et al. (7) in the way that they should be performed fertility sparing surgery and definitive surgery after completion of childbearing. The patient was informed about the post-operative follow-up procedures, but we were unable to convince the patient about the thyroidectomy operation. Patients treated without thyroidectomy with survival rates of 12 to 180 months have been reported in the literature (11). As we are unable to determine the metastasis, recurrence or the residual tissue without thyroidectomy, we think that thyroidectomy should be a part of treatment.
Serum thyroglobulin levels and I131 scan could be used for the follow-up as a marker of recurrence (6, 13). If thyroidectomy is not performed, then I131 scan or thyroglobulin have no aid in the follow-up (14). But cases in the literature similar to ours, have been reported, where follow-up has been with thyroglobulin levels or no further examination or where after fine-needle aspiration of thyroid chemotherapy has been given (11, 12, 15).
As malignant struma ovarii cases are rare, there is no standart follow-up protocol for patients who have not been performed thyroidectomy.
In conclusion, more data is needed to determine an optimal diagnosis, management and follow-up protocols for this rare entity.
Figures and Tables
References
1. Berek JS, Hacker NF. Practical gynecologic oncology. 2003. 3rd ed. Philadelphia: Lippincott Williams & Wilkins;3–38.
2. Serov SF, Scully RE, Sobin LH. Histological typing of ovarian tumors. International histological classification of tumors. No.9. 1973. Geneva: World Health Organization.
3. Kabukcuoglu F, Baksu A, Yilmaz B, Aktumen A, Evren I. Malignant struma ovarii. Pathol Oncol Res. 2002. 8:145–147.

4. Rosenblum NG, LiVolsi VA, Edmonds PR, Mikuta JJ. Malignant struma ovarii. Gynecol Oncol. 1989. 32:224–227.

5. Zakhem A, Aftimos G, Kreidy R, Salem P. Malignant struma ovarii: report of two cases and selected review of the literature. J Surg Oncol. 1990. 43:61–65.

6. Volpi E, Ferrero A, Nasi PG, Sismondi P. Malignant struma ovarii: a case report of laparoscopic management. Gynecol Oncol. 2003. 90:191–194.

7. Dardik RB, Dardik M, Westra W, Montz FJ. Malignant struma ovarii; two case reports and review of the literature. Gynecol Oncol. 1999. 73:447–451.
8. Gould SF, Lopez RL, Speers WC. Malignant struma ovarii. A case report and literature review. J Reprod Med. 1983. 28:415–419.
9. Devaney K, Snyder R, Norris HJ, Tavassoli FA. Proliferative and histologically malignant struma ovarii: a clinicopathologic study of 54 cases. Int J Gynecol Pathol. 1993. 12:333–343.
10. Chan SW, Farrell KE. Metastatic thyroid carcinoma in the presence of struma ovarii. Med J Aust. 2001. 175:373–374.

11. DeSimone CP, Lele SM, Modesitt SC. Malignant struma ovarii: a case report and analysis of cases reported in the literature with focus on survival and I131 therapy. Gynecol Oncol. 2003. 89:543–548.

12. Makani S, Kim W, Gaba AR. Struma ovarii with a focus of papillary thyroid cancer: a case report and review of the literature. Gynecol Oncol. 2004. 94:835–839.

13. Zekri JM, Manifold IH, Wadsley JC. Metastatic struma ovarii: late presentation, unusual features and multiple radioactive iodine treatments. Clin Oncol (R Coll Radiol). 2006. 18:768–772.

14. Balci TA, Kabasakal L. Is the I-131 whole-body scanning proper for follow-up management of patients with malignant struma ovarii without performing the thyroidectomy? Gynecol Oncol. 2005. 99:520.
15. Roth LM, Karseladze AI. Highly differentiated follicular carcinoma arising from struma ovarii: a report of 3 cases, a review of the literature, and a reassessment of so-called peritoneal strumosis. Int J Gynecol Pathol. 2008. 27:213–222.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download