Abstract
It has been suggested that Helicobacter pylori eradication may influence production of some peptides in the stomach, which can affect appetite. This hypothesis is controversial. To verify the hypothesis, we conducted this randomized controlled trial using H. pylori infected subjects without any gastrointestinal symptoms. The treatment group received triple H. pylori eradication therapy for 7 days and the control group received no medication. We measured ghrelin, obestatin and the tumor necrosis factor-α (TNF-α) mRNA levels in endoscopic biopsy specimens and the changes from baseline to follow-up. The plasma active n-octanoyl ghrelin and obestatin levels were measured in both groups. The ghrelin/obestatin ratios in plasma and gastric mRNA expression were calculated at baseline and follow-up. Ghrelin mRNA expression in the fundic mucosa after H. pylori eradication increased significantly compared to the control group (4.47±2.14 vs. 1.79±0.96, P=0.009), independent of inflammatory changes. However, obestatin mRNA expression decreased in the antral mucosa (-0.57±1.06 vs. 0.41±0.72, P=0.028). The treatment group showed a marginal increase (P=0.060) in plasma ghrelin/obestatin ratio. The TNF-α mRNA expression also decreased significantly with treatment. This randomized controlled trial demonstrates that H. pylori eradication increases ghrelin mRNA expression, independent of inflammatory cell changes.
Helicobacter pylori infection is an emerging global health problem and is regarded as a major cause of chronic gastritis, peptic ulcer and gastric adenocarcinoma (1, 2). It not only is the major pathogen of the gastrointestinal tract but also may be responsible for dyspeptic symptoms and reduced appetite (3, 4). Recent studies have shown that the eradication of H. pylori may be associated with improved appetite and weight gain (5, 6). However, the underlying mechanism is not well understood.
Ghrelin is a 28-amino-acid peptide that stimulates appetite and is secreted mainly in the stomach (7-9). Some studies have demonstrated that H. pylori infection causes a marked reduction in gastric and plasma ghrelin (10). Ghrelin has been suggested as a possible mediator between H. pylori infection and appetite. Other studies have supported this hypothesis by demonstrating that eradicating H. pylori increases ghrelin production (11, 12). However, these studies were limited in that they were designed as observational studies, and the enrolled subjects had inflammations of varying severity, ranging from gastritis to more serious inflammatory conditions, such as peptic ulcers and occasionally gastric cancer. Because inflammation influences ghrelin production (12), the relationship between H. pylori eradication and ghrelin production might have been biased by these inflammatory changes. Furthermore, these studies did not control for gastrointestinal symptoms such as pain and indigestion, which may also affect appetite and ghrelin production (13). To elucidate the effects of H. pylori eradication on ghrelin production, more sophisticated investigations controlling these biases are needed.
Zhang et al. (14) reported that the ghrelin gene also encodes the obestatin peptide. It was initially reported that obestatin functioned against ghrelin; for example, obestatin reduced refeeding, whereas ghrelin stimulated food intake and gastric transit. It is possible that ghrelin and obestatin work together to regulate homeostasis and body weight (15, 16), although their mutual action is still disputed. In addition, obestatin tends to be lower in gastrectomy subjects (16). These data suggest that eradication therapy for H. pylori infection could modify the production of both obestatin and ghrelin. However, there is no data regarding an association between H. pyloriinfection and obestatin, and no clinical trials verifying the relationship. Therefore, we evaluated ghrelin and obestatin production after treating H. pylori infections, through a randomised, controlled trial using healthy volunteers without gastrointestinal symptoms or peptic ulcers.
We recruited 65 volunteers with no gastrointestinal symptoms. Because ulcer healing is another important factor affecting appetite and ghrelin production, as suggested by our previous study (17) and can act as a bias in this study, we recruited only healthy volunteers not having peptic ulcers. The study was approved by the Institutional Review Board of Inje University, Ilsan-Paik Hospital in Korea. Written informed consent was obtained from the patients before their participation. Initially, all the participants underwent upper gastrointestinal endoscopy. The exclusion criteria were as follows: age <20 yr; age >70 yr; pregnancy; abnormal gastric lesions, including ulcers or cancer; duodenal ulcers; liver disease; renal impairment; previous gastrointestinal surgery; history of H. pylori eradication; and drug abuse (10, 18). After an initial endoscopy, seven patients were excluded because of peptic ulcers, 26 were excluded because they were H. pylori-negative and two refused to participate. The remaining 30 subjects with H. pylori were randomly assigned to treatment and control groups. The appetites of all subjects were estimated using a Visual Analog Scale (VAS) of appetite that has been shown to be reliable and valid for Koreans (19).
To avoid the effects of diurnal hormone variation, endoscopy was performed after an overnight fast between 08:00 and 10:00. Two biopsy specimens were taken from the midportion of the fundus along the greater curvature, and two were taken from intact mucosa in the gastric antrum, 2 cm proximal to the pylorus. The four samples were transferred to tubes containing TRIzol® (Gibco, Long Island, NY, USA) and stored immediately at -70℃ until assayed. Two additional endoscopic biopsies were taken. One sample was fixed in 10% formalin and embedded in paraffin for histological assessment and the other sample was used for the rapid urease test (CLO test: Ballard Medical Products, Draper, UT, USA) to detect H. pylori infection. All of the procedures were repeated at the time of the second endoscopy.
The treatment group received triple therapy consisting of a twice-daily regimen of 20 mg of esomeprazole, 1,000 mg of amoxicillin and 500 mg of clarithromycin for 7 days. The control group did not receive any prescription and was directed not to take any medication. Both groups underwent a second endoscopy 5 weeks later. All the examiner including pathologist or endoscopist were blind to subject group identity.
Blood samples were taken between 08:00 and 10:00, after an overnight fast. The sample was transferred into a chilled tube containing EDTA-2Na, centrifuged immediately and stored at -70℃ until assayed. The sample designated for ghrelin analysis was acidified with 50 µL of 1 NHCl and 10 µL of phenylmethylsulfonyl fluoride (PMSF) were added at 1 mL of plasma. These samples were measured in duplicate. The plasma active octanoylated form of ghrelin concentration was measured using a radioimmunoassay kit (Linco Research, St. Charles, MO, USA). The plasma obestatin was measured using a radioimmunoassay kit (Phoenix Pharmaceuticals, Belmont, CA, USA). The inter- and intra-assay coefficients of variation were 13.7 and 9.5% for ghrelin and less than 12% and less than 5% for obestatin, respectively.
The RNA was recovered using a standard reported procedure. The RNA was precipitated with isopropanol, and the pellet was washed with 70% ethanol, air-dried and dissolved in sterile diethylpyrocarbonate-treated water. The concentration and purity of the RNA were determined using spectrophotometry (Ultrospec® 1100 Pro; Amersham Pharmacia Biotech, Buck, UK) to measure the optical density ratio at 260 and 280 nm.
One microgram of total RNA was used as a template to generate cDNA by using M-MLV reverse transcriptase (Super Bio, Suwon, Korea) with random hexamer priming. The resultant cDNA was amplified using an Exicycler (Bioneer, Seoul, Korea). Real-time PCR analysis was carried out with SYBR® Premix Ex Taq™ (TaKaRa Bio, Tokyo, Japan) and specific primers. The sequences of primers were as follows: Ghrelin: 5'-ATG CTC TGG CTG GAC TTG-3'(sense) and 5'-TCT GCT TGA CCT CCA TCT T-3'(antisense; ACC No. NM_016362; product size, 155 bp); Obestatin: 5'-CAG AGG ATG AAC TGG AAG TC-3'(sense) and 5'-CAG AGG ATG TCC TGA AGA AA-3'(antisense; ACC No. NM_016362; product size, 118 bp); and TNF-a: 5'-CTT CTG GCT CAA AAA GAG AA-3'(sense) and 5'-GTC AGG GAT CAA AGC TGT AG-3'(antisense; ACC No. BC 028148; product size, 189 bp). The gene mRNA levels were normalized using β-actin.
The biopsy samples were treated in a standard manner, and neutrophil and mononuclear cell infiltration were assessed using an updated Sydney system (20). Neutrophil infiltration into the lamina propria was scored on a scale of 0 to 3, as described by the Sydney system. Mononuclear cell infiltration was determined as described above. The presence of H. pylori in the biopsy material was determined histologically using a Giemsa stain. After 5 weeks, the same histological assessments were repeated. Histological evaluations were performed by a well-trained pathologist who was blind to the treatment, endoscopic diagnosis and H. pylori infection status.
The data are expressed as mean±SD. A Student's t-test was used to compare the baseline characteristics and their changes between the treatment and control groups. Analysis of variance (ANOVA) was used to compare the expression of ghrelin mRNA and TNF-α mRNA according to the changes of neutrophil infiltration, and a Student's t-test was used according to the changes of mononuclear cell infiltration. A paired t-test was used to compare the ghrelin/obestatin ratio in the plasma and gastric mRNA expression measured before and after treatment. Multiple regression analysis was used to assess the effects of H. pylori eradication on ghrelin production by the gastric mucosa, adjusted by the weight change and change in neutrophil or mononuclear cell infiltration. A value of P<0.05 was considered statistically significant.
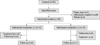
No initial differences between the treatment and control groups were observed with regard to the baseline characteristics including age, body mass index (BMI), appetite, plasma levels of ghrelin and obestatin, ghrelin, obestatin and TNF-α mRNA expression levels in the gastric mucosa, and neutrophil and mononuclear cell infiltration (Table 1). At the 5-week follow-up, three subjects from the treatment group and two from the control group withdrew. Three more people from the treatment group were excluded from the final assessment because they were still H. pylori-positive after treatment. Two from the control group were excluded for taking a drug that affects appetite (Fig. 1).
Ghrelin mRNA expression in the gastric fundic mucosa increased significantly in the treatment group compared with the control group (4.47±2.14 vs. 1.79±0.96, P<0.01). As shown in Fig. 2, gastric ghrelin mRNA expression increased substantially with treatment for all subjects in the treatment group but showed inconsistent or minor changes in the control group (Fig. 3). The plasma active ghrelin level in patients who received eradication drugs did not show a significant change compared with the control group (2.63±6.23 vs. -0.70±9.37, P=0.374). Similarly, a comparison of the VAS scores of hunger (P=0.767) and BMI (P=0.198) between the treatment and control groups failed to demonstrate statistically significant differences. Obestatin mRNA expression in the gastric antral mucosa decreased after H. pylori eradication in the treatment group but not in the control group (-0.57±1.06 vs. 0.41±0.72, P=0.028). Obestatin mRNA expression in the gastric fundic mucosa did not change in either group (Table 2).
TNF-α mRNA expression in the gastric mucosa decreased after H. pylori eradication (P=0.005, Table 2). Neutrophil and mononuclear cell infiltration decreased significantly in the treatment group after H. pylori eradication (P<0.001 and P<0.001, respectively), whereas the values did not change in the control group.
We assessed the effect of inflammatory changes on ghrelin production by the gastric mucosa. Ghrelin mRNA expression increased with a decrease in mononuclear cell infiltration (P=0.046, Table 3), but not with the changes in infiltrating neutrophils. According to the multiple regression analysis, which excluded possible confounding factors such as inflammations and weight change, the eradication of H. pylori was an important factor related to increased ghrelin mRNA expression, regardless of inflammation (P=0.004, not shown in the Tables).
There was no significant difference in the ratio of ghrelin expression to obestatin expression in the plasma or gastric mucosa between before and after H. pylori eradication (Table 4).
This study showed a significant increase in ghrelin mRNA expression in the gastric mucosa after H. pylori eradication. The obestatin mRNA expression level in the gastric antrum decreased after H. pylori eradication. The changes of plasma active ghrelin and obestatin in the treatment group were similar to the control group. The plasma ghrelin/obestatin ratio of the treatment group changed insignificantly from 2.31±0.44 to 3.01±1.00 (P=0.06).
Many researchers reported that H. pylori infection causes a marked reduction in plasma ghrelin levels (21). The alteration of plasma gastric originated appetite-controlling hormones may contribute to the changes of appetite or dyspeptic symptoms in people with H. pylori infections. Therefore, H. pylori infection is considered as an important factor in reduced appetite. Additionally, there is some evidence that an improvement in dyspeptic symptoms occurred among patients with non-ulcer dyspepsia when H. pylori was eradicated (21). Interestingly enough, the increased appetite as a results of H. pylori eradication may support the theory that the cure from H. pylori infections is associated with weight gain (6). However, the results of other studies have either been inconsistent or failed to show that ghrelin mediates the association between increased appetite and H. pylori eradication (18, 22). We conducted this randomised controlled trial to better control a possible confounders and demonstrate a clearer association between appetite and H. pylori eradication.
Some authors suggest that inflammation of the gastric mucosa is the one of the important mechanisms of H. pylori induced change of ghrelin production. Isomoto et al. showed that greater inflammation in an H. pylori-infected gastric mucosa resulted in less ghrelin production (18). Previous studies on this issue recruited subjects with peptic ulcers or stomach cancer. But peptic ulcers and cancer are conditions with severely inflamed mucosal layers. They may be caused by other factors than H. pylori infection. So the change in ghrelin production by H. pylori eradication in these previous studies might be biased by the severe inflammation of these diseases. We also found that ulcer healing was a more important factor in ghrelin production than H. pylori eradication (17). Therefore, we recruited healthy subjects without peptic ulcers in order to minimize the confounding factors and found that ghrelin production increased after H. pylori eradication, independent of inflammation.
We found that plasma ghrelin in the treatment group increased after H. pylori eradications were not statistically significant. If there is an H. pylori eradication effect in appetite control, it could be caused by the activation of the ghrelin receptor in gastric mucosa. However, the function of ghrelin is not well understood. This result is similar to that of a Japanese study in which Isomoto et al. (11) showed that the plasma ghrelin level did not change after treating H. pyloriinfections (18, 23). In contrast, Nwokolo et al. showed a clear increase of plasma ghrelin levels after H. pylori treatment. Those results led to some confusion. Cumming (24) suggested that the results were inconsistent because the subjects in the study by Nwokolo et al. were younger and probably had a shorter duration of H. pylori infection, which might have produced a different treatment response than that in the study by Isomoto et al. Our subjects were middle-aged, much older than those in Nwokolo's study, and our results are similar to those of the Japanese study.
We recruited healthy subjects with gastritis in order to eliminate the effect of severe inflammation, such as gastric ulcers. However, we observed a reduction of inflammation in the gastric mucosa after H. pylori eradication. Although neutrophil infiltration clears rapidly after H. pylori eradication, mononuclear cell invasion persists for 6 months to 1 yr, and recovery from glandular atrophy takes even longer, if it occurs at all (25). In addition, mononuclear infiltration, which is associated with glandular atrophy, changed slightly on follow up examination (12, 18). Thus, we could expect the change of ghrelin mRNA expression to continue to increase with the reduction of inflammation after treatment. However, a long-term follow-up study of H. pylori eradication is necessary to test this hypothesis.
A previous study reported that body weight and BMI increased significantly 12 months after the eradication of H. pylori (5). Our study was designed to evaluate the change in ghrelin production at 4 weeks after H. pylori eradication and therefore was too short to detect a change in BMI.
Obestatin, although derived from the same peptide precursor as ghrelin, was initially reported to antagonize the action of ghrelin by activating orphan G protein-coupled receptor 39 (14). Since the original study, two studies have partially confirmed the effects of obestatin on food intake and gastric emptying (26, 27), but two other studies have failed to replicate the findings (28, 29). A recent study has linked obestatin to obesity in humans, showing that fasting plasma obestatin was significantly suppressed in obese subjects (15, 16) and increased as weight was lost. This was paralleled by increases in circulating ghrelin concentrations. Guo et al. (15) suggested that the circulating preprandial ghrelin-to-obestatin ratio is elevated in obese humans. Vicennati et al. (30) study showed that in the presence of obesity, women had a decreased ghrelin/obestatin ratio. Based on available data, it is difficult to explain these disparate findings. Differences in the obese status could partially explain such disparate obestatin values. However, these findings may support the hypothesis that obese individuals with an imbalance of ghrelin and obestatin levels even though the results are inconsistent.
In the present study, the post-treatment plasma ghrelin/obestatin ratio increased, although insignificantly (from 2.31±0.44 to 3.01±1.00, P=0.06). There was a small difference in the obestatin mRNA expression level of gastric antrum after H. pylori eradication, which didn't influence the plasma level of obestatin. The stomach is considered an important organ for obestatin secretion because obestatin can be purified from the stomach and tends to be lower in gastrectomy patients (16). It is not surprising that H. pylori infection or its treatment can affect gastric obestatin production. However, our results did not provide evidence suggesting that obestatin production is associated with H. pylori eradication.
In summary, we demonstrated that ghrelin mRNA expression in the gastric mucosa increased after H. pylori eradication. We also suggest a hypothesis that H. pylori eradication is associated with a change in ghrelin in the gastric mucosa, regardless of inflammation. However, we could not demonstrate a clear association with obestatin. Our study was too short in duration to evaluate changes in BMI and long term changes of the ghrelin in gastric mucosa after H. pylori eradication. A follow-up study of longer duration is needed.
Figures and Tables
Fig. 2
The ghrelin mRNA expression level of gastric mucosa between baseline and 5 weeks later in the group with or without H. pylori eradication. *P<0.001 by paired t test; †P<0.05 by t-test. The changes of ghrelin mRNA expression levels in the gastric mucosa from baseline to 5 weeks later were compared in each group.

Fig. 3
The changes of gastric ghrelin mRNA expression levels in the treatment group before and after H. pylori eradication therapy (A) and in the control group between baseline and 5 weeks later (B).

Table 2
The change in ghrelin, obestatin, TNF-α, and inflammatory cells of the treatment group after intervention or 5 months later*

*Data are the mean±SD; †The changes of variables indicate the changed values measured from baseline to 5 weeks. The treatment group received H. pylori eradication therapy during the 1st week. However, the control group did not; ‡The P value was determined using the Student's t-test.
TNF-α, tumor necrosis factor-α; BMI, body mass index; VAS, visual analogue scale.
References
1. Marshall BJ, Warren JR. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet. 1984. 1:1311–1315.

2. Correa P, Houghton J. Carcinogenesis of Helicobacter pylori. Gastroenterology. 2007. 133:659–672.

3. Bravo LE, Mera R, Reina JC, Pradilla A, Alzate A, Fontham E, Correa P. Impact of Helicobacter pylori infection on growth of children: a prospective cohort study. J Pediatr Gastroenterol Nutr. 2003. 37:614–619.

4. Cho I, Blaser MJ, Francois F, Mathew JP, Ye XY, Goldberg JD, Bini EJ. Helicobacter pylori and overweight status in the United States: data from the Third National Health and Nutrition Examination Survey. Am J Epidemiol. 2005. 162:579–584.

5. Azuma T, Suto H, Ito Y, Muramatsu A, Ohtani M, Dojo M, Yamazaki Y, Kuriyama M, Kato T. Eradication of Helicobacter pylori infection induces an increase in body mass index. Aliment Pharmacol Ther. 2002. 16:Suppl 2. 240–244.

6. Furuta T, Shirai N, Xiao F, Takashima M, Hanai H. Effect of Helicobacter pylori infection and its eradication on nutrition. Aliment Pharmacol Ther. 2002. 16:799–806.

8. St-Pierre DH, Wang L, Tache Y. Ghrelin: a novel player in the gut brain regulation of growth hormone and energy balance. News Physiol Sci. 2003. 18:242–246.
9. Wren AM, Seal LJ, Cohen MA, Brynes AE, Frost GS, Murphy KG, Dhillo WS, Ghatei MA, Bloom SR. Ghrelin enhances appetite and increases food intake in humans. J Clin Endocrinol Metab. 2001. 86:5992.

10. Osawa H, Nakazato M, Date Y, Kita H, Ohnishi H, Ueno H, Shiiya T, Satoh K, Ishino Y, Sugano K. Impaired production of gastric ghrelin in chronic gastritis associated with Helicobacter pylori. J Clin Endocrinol Metab. 2005. 90:10–16.
11. Nwokolo CU, Freshwater DA, O'Hare P, Randeva HS. Plasma ghrelin following cure of Helicobacter pylori. Gut. 2003. 52:637–640.

12. Tatsuguchi A, Miyake K, Gudis K, Futagami S, Tsukui T, Wada K, Kishida T, Fukuda Y, Sugisaki Y, Sakamoto C. Effect of Helicobacter pylori infection on ghrelin expression in human gastric mucosa. Am J Gastroenterol. 2004. 99:2121–2127.

13. Shinomiya T, Fukunaga M, Akamizu T, Irako T, Yokode M, Kangawa K, Nakai Y, Nakai Y. Plasma acylated ghrelin levels correlate with subjective symptoms of functional dyspepsia in female patients. Scand J Gastroenterol. 2005. 40:648–653.

14. Zhang JV, Ren PG, Avsian-Kretchmer O, Luo CW, Rauch R, Klein C, Hsueh AJ. Obestatin, a peptide encoded by the ghrelin gene, opposes ghrelin's effects on food intake. Science. 2005. 310:996–999.

15. Guo ZF, Zheng X, Qin YW, Hu JQ, Chen SP, Zhang Z. Circulating preprandial ghrelin to obestatin ratio is increased in human obesity. J Clin Endocrinol Metab. 2007. 92:1875–1880.

16. Huda MS, Durham BH, Wong SP, Deepak D, Kerrigan D, McCulloch P, Ranganath L, Pinkney J, Wilding JP. Plasma obestatin levels are lower in obese and post-gastrectomy subjects, but do not change in response to a meal. Int J Obes (Lond). 2008. 32:129–135.

17. Jang EJ, Park SW, Park JS, Park SJ, Hahm KB, Paik SY, Sin MK, Lee ES, Oh SW, Park CY, Baik HW. The influence of the eradication of Helicobacter pylori on gastric ghrelin, appetite, and body mass index in patients with peptic ulcer disease. J Gastroenterol Hepatol. 2008. 23:Suppl 2. S278–S285.
18. Isomoto H, Ueno H, Nishi Y, Wen CY, Nakazato M, Kohno S. Impact of Helicobacter pylori infection on ghrelin and various neuroendocrine hormones in plasma. World J Gastroenterol. 2005. 11:1644–1648.
19. Kim HB, Lee ES, Oh SW, Kim YH, Lee DE, Hwang CK, Lee EY, Yoon YS, Yang YJ. Validity, reproducibility of visual analogue scales in assessment of appetite sensations. J Korean Acad Fam Med. 2008. 29:736–745.
20. Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996. 20:1161–1181.
21. Konturek PC, Cześnikiewicz-Guzik M, Bielanski W, Konturek SJ. Involvement of Helicobacter pylori infection in neuro-hormonal control of food intake. J Physiol Pharmacol. 2006. 57:Suppl 5. 67–81.
22. Moayyedi P, Soo S, Deeks J, Delaney B, Harris A, Innes M, Oakes R, Wilson S, Roalfe A, Bennett C, Forman D. Eradication of Helicobacter pylori for non-ulcer dyspepsia. Cochrane Database Syst Rev. 2006. 2:CD002096.

23. Isomoto H, Ueno H, Saenko VA, Mondal MS, Nishi Y, Kawano N, Ohnita K, Mizuta Y, Ohtsuru A, Yamashita S, Nakazato M, Kohno S. Impact of Helicobacter pylori infection on gastric and plasma ghrelin dynamics in humans. Am J Gastroenterol. 2005. 100:1711–1720.

24. Cummings DE. Helicobacter pylori and ghrelin: interrelated players in body-weight regulation? Am J Med. 2004. 117:436–439.

25. Blaser MJ, Atherton JC. Helicobacter pylori persistence: biology and disease. J Clin Invest. 2004. 113:321–333.

26. Bresciani E, Rapetti D, Donà F, Bulgarelli I, Tamiazzo L, Locatelli V, Torsello A. Obestatin inhibits feeding but does not modulate GH and corticosterone secretion in the rat. J Endocrinol Invest. 2006. 29:RC16–RC18.

27. Zizzari P, Longchamps R, Epelbaum J, Bluet-Pajot MT. Obestatin partially affects ghrelin stimulation of food intake and growth hormone secretion in rodents. Endocrinology. 2007. 148:1648–1653.

28. Nogueiras R, Pfluger P, Tovar S, Arnold M, Mitchell S, Morris A, Perez-Tilve D, Vázquez MJ, Wiedmer P, Castañeda TR, DiMarchi R, Tschöp M, Schurmann A, Joost HG, Williams LM, Langhans W, Diéguez C. Effects of obestatin on energy balance and growth hormone secretion in rodents. Endocrinology. 2007. 148:21–26.





 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download