Abstract
CpG-Oligodeoxynucleotide (ODN) has two backbones. Phosphorothioate backbone (PS) shows a strong immunostimulating effect while phosphodiester (PE) shows little in vivo. 3' hexameric deoxyriboguanosine-run (3' dG6-run) conjugation to PE CpG-ODN has been reported to enhance immunostimulation and to protect against asthma when injected at the time of sensitization in mice. We evaluated the treatment effects of PE and PS CpG-ODN with or without 3' dG6-run on asthma in presensitized mice. BALB/c mice sensitized with ovalbumin and alum were challenged with 1% ovalbumin on three days. CpG-ODNs (100 µg) or PBS were injected 4 times; 27 hr before challenge and 3 hr before each challenge (CpG-dG6: CpG-ODN with 3' dG6-run, PE*-CpG-dG6: PE-CpG-dG6 with two PS backbones at the 5' terminus). PE-CpG showed no treatment effect. PE-CpG-dG6 only increased ovalbumin-specific IgG2a. PE*-CpG-dG6 increased ovalbumin-specific IgG2a but also reduced BAL fluid eosinophils and airway hyperresponsiveness. PS-CpG increased ovalbumin-specific IgG2a, reduced airway inflammation and airway hyperresponsiveness. PS-CpG-dG6 was less effective than PS-CpG on airway inflammation and airway hyperresponsiveness. In pre-sensitized mice, PE-CpG required not only 3' dG6-run but also the modification of two PS linkages at 5' terminus to inhibit features of asthma. PS-CpG was strong enough to inhibit asthma but PS-CpG-dG6
was less effective.
CpG-Oligodeoxynucleotides (ODN) is an oligodeoxynucleotides that contain CpG motifs, 5'-purine-purine-unmethylated deoxycytosine-deoxyguanosine-pyrimidine-pyrimidine-3' (1). CpG-ODN, a well-known TLR9 agonist, stimulates antigen-presenting cells (APCs) like plasmacytoid dendritic cells to produce Th1-promoting cytokines, such as type I IFN and IL-12. CpG-ODN is also involved in the maturation of plasmacytoid dendritic cells to potent antigen-presenting cells, the transition of monocytes into functional dendritic cells, NK cell stimulation, B cell activation, and in the production of Th1 type antibodies (2).
CpG-ODN is considered to be a potential new therapeutic modality for allergic diseases, cancer, and infectious diseases (2). Recently, a clinical trial of CpG-ODN conjugated with ragweed allergen Amb a 1 was conducted in allergic rhinitis (3). Patients received only six weekly injections of Amb a 1-CpG-ODN conjugate or placebo vaccine before the first ragweed season and were monitored during the next two ragweed seasons, and this 6-week regimen of allergen-CpG ODN vaccinations appeared to offer long-term clinical efficacy for the treatment of ragweed induced allergic rhinitis.
CpG-ODN can be classified into two forms of different backbone structures according to its chemical properties (4, 5). One of these has a phosphodiester backbone (PE) whereas the other has a phosphorothioate backbone (PS). The PE is the natural form of bacterial DNA which contains many CpG-motifs (20 times more than vertebrates) (1). This form serves as a danger signal, which is detected by TLR9, a pattern-recognition receptor, when bacteria invade the host (2). However, the PE form has no or little immunostimulatory effect in vivo because it is easily attacked by exonucleases. On the other hand the PS backbone is a synthetic form that is resistant to exonucleases, and which has a strong immunostimulatory effect even in in vivo (4). This is why most experiments on CpG-ODN have been performed with PS CpG-ODN in vivo. Moreover, the immunostimulatory effects of CpG-ODNs depend on flanking sequences and the optimal sequences are species-specific (2). Concerns about the possible side effects of PS CpG-ODN, such as, granuloma formation at high dosages and splenomegaly, drove the developments of new strategies to enhance the immunostimulatory potential of PE CpG-ODN (4-6). Some strategies now available; the conjugation of CpG-ODN with a 3' hexameric deoxyriboguanosine run (3' dG6 run) (4, 5) and packing CpG-ODN into virus-like particles (7). We have previously demonstrated that the conjugation of CpG-ODN with 3' dG6 run augmented the immunostimulatory effect of PE CpG-ODN and inhibited the development of asthma when it was delivered with allergen at the time of sensitization in mice (4). Moreover, the 3' dG6 run was observed to have an enhancing effect in both PE and PS CpG-ODNs (4). Interestingly, PE CpG-ODN with a 3' dG6 run induced less splenomegaly but had an inhibitory effect on allergen-specific IgE production similar to that of PS CpG-ODN (5). In this study, we evaluated the effects of PE or PS CpG-ODN with or without a 3' dG6 run conjugate treatments at the time of allergen challenge.
All CpG ODNs were purchased from GenoTech (Daejon, Korea). Their sequences and modifications are presented in Fig. 1A. PS-CpG is used to designate PS CpG-ODN, and its PE form is represented by PE-CpG. PS-CpG-dG6 is used to designate PS-CpG with six deoxyriboguanosine residues at the 3' terminus, and its PE form is designed as PE-CpG-dG6. PE*-CpG-dG6 represents PE-CpG-dG6 with two PS backbones at the 5' terminus.
Six-week old female BALB/c mice were purchased from DBL (Daejon, Korea). BALB/c mice were maintained in the clinical research institute at Seoul National University Hospital. All animal experiments were approved by the committee on animal experimentation at our institution.
As Fig. 1B illustrates, six mice in each group were sensitized by injection of 20 µg ovalbumin (OVA) (Grade V; Sigma, St. Louis, MO, U.S.A.) emulsified in 2 mg aluminum hydroxide intraperitoneally (i.p.). Animals received an identical booster immunization 14 days later. On days 21, 22, and 23 after the initial sensitization, mice were challenged for 30 min with an aerosol of 1% (w/v) OVA in PBS in a Plexiglas chamber using an ultrasonic nebulizer (NE-U12, Omron, Japan). Mice were given four injections of 100 µg of CpG ODNs or PBS i.p. in total volume of 100 µL PBS, at 24 hr intervals from 27 hr before the first OVA inhalation challenge, i.e., 3 hr before each challenge.
Twenty-four hours after the final OVA challenge, airway hyperresponsiveness (AHR) was assessed by determining methacholine-induced airflow obstruction (Penh) using one chamber whole body plethysmography (Allmedicus, Anyang, Korea), as previously described (4, 8-10). Increasing doses of methacholine (ranging from 2.5-50 mg/mL; Sigma) were administered by nebulization for 3 min, and Penh values were calculated over the subsequent 3 min. Results were presented as PC200 values, which are defined as the concentration of methacholine required to increase baseline Penh by 200%.
Forty-eight hours after the last OVA challenge, tracheae were cannulated and lungs were lavaged with five 0.4 mL aliquots of pyrogen-free saline. After Diff-quickR staining lung lavage cells in cytospin preparations, two investigators counted blindly more than 300 inflammatory cells under a light microscope and classified these as macrophages, lymphocytes, neutrophils, or eosinophils (4, 8).
Following BAL, lungs were infused with 10% formalin and embedded in paraffin. Lung sections were stained with hematoxylin and eosin and assessed by light microscopy. Inflammation scores were graded as previously described (4, 8). Briefly, degree of peribronchial and perivascular inflammation was evaluated using a subjective scale of 0-3. A value of 0 was assigned when no inflammation was detectable, a value of 1 for occasional cuffing by inflammatory cells, a value of 2 when most bronchi or vessels were surrounded by a thin layer (one to five cells) of inflammatory cells, and a value of 3 when most bronchi or vessels were surrounded by a thick layer (more than five cells) of inflammatory cells. The total lung inflammation was defined as the average of peribronchial
and perivascular inflammation score.
Forty-eight hours after the last OVA challenge, blood samples were obtained by cardiac puncture. Antibody titers were measured as previously described (4, 8). Briefly, microtiter plates (Dynex Technologies, Chantilly, VA, U.S.A.) were coated overnight with 2 µg/mL of OVA in a 50 mM carbonate buffer (pH 9.6) at 4℃. Nonspecific binding was blocked with 2% bovine serum albumin for 1 hr at 20℃. After incubating with test sera for 2 hr, plates were incubated with horse radish peroxidase-labeled goat anti-mouse IgE or IgG2a (PharMingen, San Diego, CA, U.S.A.) for 1 hr at 20℃. The reaction was developed using a tetramethylbenzidine (Sigma, St. Louis, MO, U.S.A.) substrate and then stopped by adding 2 N H2SO4. Subsequently, optical density was measured at 450 nm. A high titer of anti-OVA IgE or IgG2a was used as a standard, and linear standard curves were obtained by serially diluting standard serum. The results are expressed in arbitrary units (A.U.) according to measured O.D. values.
Mouse and spleen weights, the latter of which were cautiously separated without bleeding, were measured as previously reported (5). Spleen to body weight ratio were compared.
Cytokine production by splenocytes was evaluated as previously described (5). Briefly, spleens were homogenized using a 94-µm screen (Bellco Glass Inc. Vineland, NJ, U.S.A.) to obtain single cell suspensions. Splenocytes (2×106) were then cultured with OVA (100 µg/mL) or PBS control in 12-well plates. After 2 days, IL-4, IL-5, IFN-γ, and IL-12 production levels were quantified in culture supernatants by sandwich ELISA using specific monoclonal antibody pairs.
To investigate the treatment effects of CpG ODNs with different backbones and with or without a 3' dG6-run in presensitized mice against the development of allergic asthma, we measured AHR to inhaled methacholine after OVA challenge (Fig. 2A). As was expected, the administration of PS-CpG, but not of PE-CpG, effectively inhibited AHR. However, PE-CpG-dG6, which had shown a significant inhibitory effect when delivered at the time of sensitization (4), did not inhibit AHR as well as PE-CpG.
PE*-CpG-dG6 reduced AHR and this effect was comparable to that of PS-CpG (P>0.05). Interestingly, the inhibitory effect of PS-CpG on airway hyperresponsiveness reduced when it was conjugated with a dG6-run at its 3' terminus (PS-CpG-dG6).
Eosinophil proportions in BAL fluid were similar in PBS (70.7±2.8%), PE-CpG (67.0±3.6%), and PE-CpG-dG6 (62.9±10.8%) treated mice (P>0.05). However, PE*-Cp-GdG6 significantly decreased BAL eosinophil counts (33.1±3.8%) vs. PE-CpG treated mice (P<0.05), as shown in Fig. 2B. The administration of PS-CpG more significantly inhibited airway eosinophilia (4.2±1.3%) than PE*-CpG-dG6 (P<0.05). The administration of PS-CpG-dG6 also significantly inhibited airway eosinophilia (by 20.1±10.5%) but this was less than that achieved by PS-CpG (P<0.05). No significant difference was observed between PS-CpG-dG6 and PE*-CpG-dG6 treated mice in terms of BAL eosinophilia (P>0.05).
Concerning lung histology, PE-CpG, PE-CpG-dG6, PE*-CpG-dG6, and PS-CpG-dG6 treated mice showed peribronchial and perivascular inflammation at the same level as that observed in PBS treated mice (peribronchial/total inflammation scores: 2.3±0.2/2.6±0.1, 2.3±0.2/2.6±0.2, 2.2±0.3/2.5±0.2, 2.1±0.1/2.3±0.1 vs. 2.2±0.2/2.5±0.1, respectively, P>0.05). Only PS-CpG treated mice showed significant inhibitions of peribronchial, perivascular, and total lung inflammation vs. PE-CpG treated mice (peribronchial/total inflammation scores: 1.4±0.4/1.6±0.6 vs. 2.3±0.2/2.6±0.1, P<0.05).
Interestingly, serum OVA specific IgE levels, which depend on IL-4 and IL-13, were not changed by any form of CpG backbone or by the presence of the 3' dG6-run, as shown in Fig. 3. Serum OVA specific IgG1 levels were not changed either.
IgG2a levels that depend on IL-12 and may reflect Th1 immune response were not changed by PE-CpG only, but increased after 3' dG6-run (PE-CpG-dG6) conjugation. PE*-CpG-dG6 also showed higher IgG2a levels that were comparable to those of PE-CpG-dG6. PS-CpG and PS-CpG-dG6 treated mice also showed higher IgG2a levels than PE-CpG or PBS treated mice. However, IgG2a production was significantly lower for PS-CpG with a 3' dG6-run than for PS-CpG (P<0.05).
The ratio of spleen to body weight was highest in the group treated with PS-CpG: 17.88±0.49 (P<0.05), followed by PS-CpG-dG6 (10.37±0.74) and PE*-CpG-dG6 (7.77±0.78). This ratio was higher for PS-CpG-dG6 than for PE*-CpG-dG6, and these two ratios were higher than those of PBS (5.10±0.18), PE-CpG (5.53±0.23), and PE-CpG-dG6 (5.37±0.38) (P<0.05) (Fig. 4).
To understand the mechanisms underlying the prevention of allergic lung inflammation by the 3' dG6-run containing CpG ODNs, we examined cytokine production from splenocytes stimulated with OVA (Fig. 5). IL-4 production was significantly decreased by PS-CpG and PS-CpG-dG6 (156.5±36.0, 115±17.8 vs. PE-CpG 372±41.7 pg/mL, P<0.05). No significant change in IL-4 production was observed in PBS, PE-CpG, PE-CpG-dG6 and PE*-CpG-dG6 treated mice (511.2±111.6, 372±41.7, 370.8±34.4, 453.7±55.3 pg/mL, P>0.05).
IL-5 production was significantly decreased by PE*-CpG-dG6, PS-CpG and PS-CpG-dG6 (1,587.8±296.9, 363.9±116.7, 681.3±172.5 vs. PE-CpG 2,440.5±63.9 pg/mL, P<0.05). No significant change in IL-5 production was observed in PBS, PE-CpG, and PE-CpG-dG6 treated mice (2,189.1±432, 2,440.5±63.9, 1,798.3±319.9 pg/mL, P>0.05).
IL-12 production was significantly increased by PE-CpG-dG6, PE*-CpG-dG6, PS-CpG, and PS-CpG-dG6 (45.2±2.3, 77.7±1.7, 173.9±29.9, 114.3±15.6 vs. PE-CpG 29.6±3.4 pg/mL, P<0.05). No change in IL-12 production was observed for PBS, and PE-CpG treated mice (28.9±4.7, 29.6±3.4 pg/mL P>0.05).
IFN-γ production was significantly increased by PE*-CpG-dG6, PS-CpG, and PS-CpG-dG6 (1,233.7±154.6, 7,581.5±5,446.1, 13,019.4±8,009.9 vs. PE-CpG 151.6±76.8 pg/mL, P<0.05). No significant change in IFN-γ production was observed in PBS, PE-CpG, and PE-CpG-dG6 treated mice (280.7±44.9, 151.6±76.8, 222.5±80.4 pg/mL, P>0.05).
PE-CpG-dG6 produced more amount of IL-12 than PE-CpG (P<0.05) which was correlated with increased production of serum OVA specific IgG2a. PE*-CpG-dG6 produced more amount of IL-12 than PE-CpG-dG6 (P<0.05). PE*-CpG-dG6 produced less amount of IL-12 and more amount of IL-4 and IL-5 than PS-CpG and PS-CpG-dG6 (P<0.05). IFN-γ production by PE*-CpG-dG6, PS-CpG, and PS-CpG-dG6 was not different (P>0.05). The levels of IL-4, IL-5, IL-12, and IFN-γ produced by PS-CpG and PS-CpG-dG6 were not different (P>0.05).
Treating CpG-ODNs with or without a 3' dG6-run at the time of allergen challenge produced results that differed from those obtained by treating CpG-ODNs at the time of sensitization (4). 3' dG6-run conjugation to CpG ODNs changed their asthma inhibiting effects in a backbone type dependent manner. PS-CpG administered at the time of challenge was strong enough to inhibit asthma phenotypes as delivered at the time of sensitization, while PE-CpG required not only 3' dG6-run but also the modification of two PE linkages at its 5' terminus to PS linkages to produce an inhibitory effect. PS-CpG-dG6 was less effective and could not inhibit airway inflammation and airway hyperresponsiveness as PS-CpG.
PE-CpG has little immunostimulatory effect in vivo because its phosphodiester backbone is easily degraded by exonucleases (4, 5). We have shown that the failure of PE-CpG to inhibit asthmatic responses could be overcome by 3' dG6-run conjugation when PE-CpG-dG6 is injected at the time of sensitization (4). Runs of dG residues can form a four stranded conformation, which is called the G-quartet, which confer nuclease resistance and facilitate binding to the scavenger receptors of macrophages (11, 12). Accordingly, 3' dG6-run may extend the half-life of PE-CpG and allow the efficient targeting of PE-CpG by APCs, presumably via a scavenger receptor (4). In this study, however, PE-CpG-dG6 had little immunostimulatory effect when it was delivered at the time of allergen challenges in pre-sensitized mice; in fact, it only increased IgG2a and mild IL-12 production in the present study.
PE*-CpG-dG6, which has two PS linkages at the 5' terminus of PE-CpG-dG6, had a better immunostimulatory effect than PE-CpG or PE-CpG-dG6. PE*-CpG-dG6 inhibited airway hyperresponsiveness like PS-CpG, decreased BAL fluid eosinophilia, increased serum allergen specific IgG2a and Th1 cytokine production by splenocytes. Two PS bonds located at the 5' terminus can contribute to resistance against exonuclease and do not affect the G-quartet structure (12). PE*-CpG-dG6 was also more effective at preventing murine allergic asthma than PE-CpG-dG6 in previous report (4). Thus, two 5' terminal PS linkages may synergize with 3' dG6-run in PE backbone CpG-ODN, and thus, further enhance immunomodulatory properties.
PS-CpG was more potent, and alone was found to decrease airway inflammation as determined by lung histology. This study suggests that a stronger immunomodulatory effect is required to reverse Th2 response after sensitization than at the time of sensitization. However, there is concern about the possible side effects of PS-CpG such as long-lasting effects of lymphadenopathy, splenomegaly, as well as sustained local IFN-γ and IL-12 production after the administration (5, 6). Splenomegaly, a possible side effect of CpG-ODNs-extramedullary hematopoiesis, seemed to be more related to the presence of the phosphorothioate backbone, as we have previously reported (5). PS-CpG showed splenomegaly and splenomegaly was reduced by PS-CpG-dG6, which failed to inhibit airway hyperresponsiveness. PE*-CpG-dG6 inhibited airway hyperresponsiveness with less splenomegaly than PS-CpG or PS-CpG-dG6. It may be a possible advantage of PE*-CpG-dG6 that it could deliver immunomodulatory effects without increasing risk of splenomegaly.
PS-CpG inhibited both airway inflammation and airway hyperresponsiveness. PE*-CpG-dG6, which inhibited airway hyperresponsiveness, inhibited eosinophilic airway inflammation in BAL fluid less than PS-CpG but could not inhibit airway inflammation in lung tissue. Dissociation of airway inflammation and airway hyperresponsiveness has been reported, and L-selectin could be one of possible factors (8, 13). PS-CpG may inhibit airway hyperresponsiveness by the increased production of IL-12 and IFN-γ with decreased IL-4 and IL-5 production. For PE*-CpG-dG6, increased production of IL-12 with decreased IL-5, which can decrease eosinophilia in BAL fluid, may be important mediators on the inhibition of airway hyperresponsiveness. IL-4, IL-5, IL-13, IL-10, and other factors could be involved in the complex mechanism of airway hyperresponsivenss (8, 14, 15). CpG-ODN is known to produce IL-10 which can show immunomodulatory effect with suboptimal concentrations of Th1-type cytokines for IL-5 suppression (16). CpG-ODN can induce a much more vigorous Th1 in the absence of IL-10 (16). CpG-ODN can also regulate activity of indoleamine 2,3-dioxygenase, the rate-limiting enzyme in catalysis of tryphtophan to kynureinines; kynurenines are potent immunomodulatory molecules that regulate T cell function (16). We failed to detect the regulatory function of CpG-ODN, which is the limitation in this study.
Discrepancy affected the immunomodulatory potential and the inhibition of airway hyperresponsiveness, possibly due to the combined effects of multiple mechanisms of airway hyperresponsiveness. PS-CpG-dG6 inhibited eosinophilic airway inflammation in BAL fluid less than PS-CpG (but as much as that of PE*-CpG-dG6) and could not inhibit airway hyperresponsiveness while cytokine levels measured showed no difference to those of PS-CpG. This finding contradicts that of our previous in vivo study, which found that 3' dG6-run conjugation showed an enhanced immunostimulatory effect for both PE and PS CpG when delivered at the time of sensitization (4). However, it concurs with those findings of previous studies (6, 11). In in vitro studies, it has been reported that the conjugation of poly-G to PS-CpG may act like an neutralizing motif (CpG-N) and the conjugation of a 3' dG6-run to PS-CpG was not found to potentiate TNF-α and IL-12 production by splenic dendritic cells; rather it had an inhibitory effect (6, 11, 17). Recently, characterization of suppressive or inhibitory oligonucleotides has been reported that inhibit TLR9 mediated activation of innate immunity (18). The inhibitory motif is a synthetic form of PS but not PE backbone, which is especially short, 11-15 base long oligonucleotides with pyrimidin-rich triplets followed by a GGG sequence, e.g. CCTN3-5GGG sequence (18-20). There are two forms; trans and cis-form. It was reported that trans-form showed stronger inhibitory effect than cis-form (18). In this study, the sequence of PS-CpG-dG6 contained that of cis-form inhibitory sequence (cctgacgttgggggg) and that could be why PS 3' dG6 showed less effect on PS-CpG in this study. However, the levels of cytokines such as IL-4, IL-5, IL-12 and IFN-γ produced by PS-CpG and PS-CpG-dG6 were not significantly different in this study. The exact mechanism involved remains to be elucidated. Measurement of suppressive cytokines such as IL-10 or TGF-β might have given some clue for this discrepancy and it would be the limitation of this study.
Treating CpG-ODNs with or without a 3' dG6-run at the time of allergen challenge produced results that differed from those obtained by treating CpG-ODNs at the time of sensitization. PS-CpG was found to be the most effective form of CpG-ODN but induced splenomeglay. PE-CpG required not only 3' dG6-run but also the additional modification of two 5' linkages to PS to be some effective one with less splenomegaly. Conjugation of a 3' dG6 run into PS-CpG, which contained cis-form of inhibitory motif, showed a negative effect in this setting.
Figures and Tables
 | Fig. 1Experimental protocols of sensitization and challenge. (A) Sequences and modifications of CpG ODNs used in this study. Immunostimulatory CpG motifs are underlined. Capital letters represent phosphodiester backbones and small letters phosphorothioate backbones. (B) BALB/c mice were sensitized by injecting OVA/alum intraperitoneally (i.p.) and later challenged with OVA on the indicated days (six mice per group). Mice were administered four injections of 100 µg of CpG ODNs or PBS i.p. in a total volume of 100 µL PBS at 24 hr intervals from 27 hr before the first OVA inhalation challenge. |
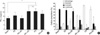 | Fig. 2Effects of the 3' dG6 run conjugated to CpG ODNs on the development of airway hyperresponsiveness (AHR) (A) (*: P<0.05, ns: no significant difference), and cellular proportions in bronchoalveolar lavage (BAL) fluid (B) (P<0.05, eosinophil proportions, *: vs. PBS, †: vs. PE-CpG-dG6, ‡: vs. PE*-CpG-dG6, #: vs. PS-CpG). AHR is represented by PC200, and cellular proportions are expressed as relative percentages of cell counts. Representative data of two independent experiments are shown. Bars indicate mean values±SEM of six mice. |
 | Fig. 3Antibody responses in the sera of OVA-challenged BALB/c mice. Representative data of two independent experiments are shown. Bars indicate the mean values±SEM of six mice. (P<0.05, *: vs. PBS, †: vs. PE-CpG-dG6, ‡: vs. PE*-CpG-dG6, #: vs. PS-CpG). |
References
1. Krieg AM, Wagner H. Causing a commotion in the blood: immunotherapy progresses from bacteria to bacterial DNA. Immunol Today. 2000; 21:521–526.

2. Vollmer J. Progress in drug development of immunostimulatory CpG oligodeoxynucleotide ligands for TLR9. Expert Opin Biol Ther. 2005; 5:673–682.

3. Creticos PS, Schroeder JT, Hamilton RG, Balcer-Whaley SL, Khattignavong AP, Lindblad R, Li H, Coffman R, Seyfert V, Eiden JJ, Broide D. Immune Tolerance Network Group. Immunotherapy with a ragweed-toll-like receptor 9 agonist vaccine for allergic rhinitis. N Engl J Med. 2006; 355:1445–1455.
4. Park Y, Chang YS, Lee SW, Cho SY, Kim YK, Min KU, Kim YY, Cho SH, Sung YC. The enhanced effect of a hexameric deoxyriboguanosine run conjugation to CpG oligodeoxynucleotides on protection against allergic asthma. J Allergy Clin Immunol. 2001; 108:570–576.

5. Chang YS, Kim YK, Min KU, Kim YY, Seong YC, Cho SH. Conjugation of 3' hexameric deoxyriboguanosine run to phosphodiester CpG oligodeoxynucleotides can inhibit allergen-specific IgE synthesis with less risk of splenomegaly. J Allergy Clin Immunol. 2005; 116:1388–1390.

6. Dalpke AH, Zimmermann S, Albrecht I, Heeg K. Phosphodiester CpG oligonucleotides as adjuvants: polyguanosine runs enhance cellular uptake and improve immunostimulative activity of phosphodiester CpG oligonucleotides in vitro and in vivo. Immunology. 2002; 106:102–112.

7. Storni T, Ruedl C, Schwarz K, Schwendener RA, Renner WA, BachmannMF . Nonmethylated CG motifs packaged into virus-like particles induce protective cytotoxic T cell responses in the absence of systemic side effects. J Immunol. 2004; 172:1777–1785.

8. Chang YS, Kim YK, Kim TB, Kang HR, Kim SS, Bahn JW, Min KU, Kim YY, Cho SH. Airway inflammation and allergen specific IgE production may persist longer than airway hyperresponsiveness in mice. J Korean Med Sci. 2004; 19:69–73.

9. Chang YS, Kim YK, Bahn JW, Kim SH, Park HW, Kim TB, Cho SH, Min KU, Kim YY. Comparison of asthma phenotypes using different sensitizing protocols in mice. Korean J Intern Med. 2005; 20:152–158.

10. Jeon SG, Lee CG, Oh MH, Chun EY, Gho YS, Cho SH, Kim JH, Min KU, Kim YY, Kim YK, Elias JA. Recombinant basic fibroblast growth factor inhibits the airway hyperresponsiveness, mucus production, and lung inflammation induced by an allergen challenge. J Allergy Clin Immunol. 2007; 119:831–837.

11. Lee SW, Song MK, Baek KH, Park Y, Kim JK, Lee CH, Cheong HK, Cheong C, Sung YC. Effects of a hexameric deoxyriboguanosine run conjugation into CpG oligodeoxynucleotides on their immunostimulatory potentials. J Immunol. 2000; 165:3631–3639.

12. Bishop JS, Guy-Caffey JK, Ojwang JO, Smith SR, Hogan ME, Cossum PA, Rando RF, Chaudhary N. Intramolecular G-quartet motifs confer nuclease resistance to a potent anti-HIV oligonucleotide. J Biol Chem. 1996; 271:5698–5703.

13. Fiscus LC, Van Herpen J, Steeber DA, Tedder TF, Tang ML. L-Selectin is required for the development of airway hyperresponsiveness but not airway inflammation in a murine model of asthma. J Allergy Clin Immunol. 2001; 107:1019–1024.
14. Leong KP, Huston DP. Understanding the pathogenesis of allergic asthma using mouse models. Ann Allergy Asthma Immunol. 2001; 87:96–109.

15. Cockcroft DW, Davis BE. Mechanisms of airway hyperresponsiveness. J Allergy Clin Immunol. 2006; 118:551–559.

16. Kline JN. Eat dirt: CpG DNA and immunomodulation of asthma. Proc Am Thorac Soc. 2007; 4:283–288.

17. Ballas ZK, Rasmussen WL, Krieg AM. Induction of NK activity in murine and human cells by CpG motifs in oligodeoxynucleotides and bacterial DNA. J Immunol. 1996; 157:1840–1845.
18. Peter M, Bode K, Lipford GB, Eberle F, Heeg K, Dalpke AH. Characterization of suppressive oligodeoxynucleotides that inhibit Toll-like receptor-9 mediated activation of innate immunity. Immunology. 2008; 123:118–128.
19. Lenert P, Rasmussen W, Ashman RF, Ballas ZK. Structural characterization of the inhibitory DNA motif for the type A (D)-CpG-induced cytokine secretion and NK-cell lytic activity in mouse spleen cells. DNA Cell Biol. 2003; 22:621–631.

20. Lenert P. Inhibitory oligodeoxynucleotides-therapeutic promise for systemic autoimmune diseases? Clin Exp Immunol. 2005; 140:1–10.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download