Abstract
This study was designed to examine the effects of recombinant human growth hormone replacement on somatic growth and cognitive function in hypophysectomized (HYPOX) female Sprague-Dawley rats. Rats (5 per group) were randomized by weight to 3 experimental groups: group 1, administered 200 µg/kg of GH once daily for 9 days; group 2, administered 200 µg/kg of GH twice daily; and group 3, administered saline daily. Somatic growth was evaluated by measurement of body weight daily and of the width of the proximal tibial growth plate of the HYPOX rats. Cognitive function was evaluated using the Morris water maze (MWM) test. The results indicated that GH replacement therapy in HYPOX rats promoted an increase in the body weight and the width of the tibial growth plate in a dose-dependent manner. On the third day of the MWM test, the escape latency in the GH-treated groups 1 and 2 was significantly shorter than that in the control rats (P<0.001 and P=0.032, respectively), suggesting that rhGH improved spatial memory acquisition in the MWM test. Therefore it is concluded that rhGH replacement therapy in HYPOX rats stimulates an increase in somatic growth in a dose-dependent manner and also has beneficial effects on cognitive functions.
Recombinant human growth hormone (rhGH) is used to treat growth hormone deficiency (GHD), statural disorders associated with Turner syndrome, chronic renal failure, Prader-Willi syndrome, intrauterine growth retardation, idiopathic short stature (ISS) and AIDS wasting in adults in the United States (1). GH replacement therapy has been shown to promote statural growth in children with GHD and with short stature due to other causes (2). The use of rhGH in adults having GHD has been shown to decrease their excess fat mass, increase lean mass (3), increase bone mineral density (BMD) (4), improve lipid profile (5, 6), and in some cases, improve the quality of life and physical performance (3, 7). GHD is known to be associated with cognitive impairment. Moreover, there is evidence that cognitive performance, particularly attention and memory, is improved by GH replacement therapy in GHD patients (8, 9). In addition, it has been suggested that the decline in GH and insulin-like growth factor I (IGF-I) that is observed with advancing age may contribute to impaired cognitive function associated with aging (10, 11). Further, it was proved that growth hormone-releasing hormone (GHRH) treatment, which brings about an increase in the GH and IGF-I levels, improved performance on a number of cognitive tasks in healthy normal elderly (12).
The association between GH and cognitive processes has also been supported by studies on rats. It was demonstrated that GH and GHRH facilitated long-term memory in young rats, and GH delayed the extinction response in both young and aged rats (13). Madeleine et al. (14) reported that GH replacement in hypophysectomized (HYPOX) rats, 1) improved spatial memory (a form of long-term memory) as observed in the Morris water maze (MWM) test and, 2) affected hippocampal levels of N-methyl-D-aspartate (NMDA) receptor subunit and post-synaptic density (PSD)-95 gene transcript levels in the glutamatergic neurotransmission system. HYPOX rat is a well-known animal model of GHD, and the hippocampus has an essential role in spatial memory in both humans and rats (15). The MWM test is a memory test based on the capacity of animals to rescue themselves by reaching a hidden goal platform in a pool of water (16). Rats with a damaged hippocampus display spatial navigation impairments and perform poorly in the MWM test (16).
We hypothesized that GH replacement in HYPOX female Sprague-Dawley rats would, 1) stimulate somatic growth as estimated from their body weights and the width of the proximal tibial growth plate and, 2) improve cognitive function as evaluated by the MWM test. We also evaluated the dose effects of GH on somatic growth and cognitive function.
HYPOX female Sprague-Dawley rats aged 51-58 days and weighing 90-100 g at study initiation were supplied by LG Life Science. They were hypophysectomized at 25-30 days of age. The rats were weighed at 37-44 days of age, and only healthy rats were retained. The retained rats were reweighed 7 days later, and only those rats that were in good health and had not gained or lost >10% of their body weight in the previous 7-day period were selected. We followed the USP30-NF25 procedure with somatotropin from the US Pharmacopeia (USP), which is a standard procedure used for GH bioassay using an animal model.
The rats were housed under standardized conditions (12-hr light and 12-hr dark cycle; lights switched on at 0800 hr; temperature, 21-26℃; humidity, 40-60%). Food and drinking water were available in the cages. Body weight was measured at 0800 hr everyday. Rats (5 per group) were randomized by weight to 3 experimental groups as follows. The rats in group 1 were administered daily subcutaneous injections of 200 µg/kg of rhGH (Growtropin®; Dong-A Pharm, Seoul, Korea) at 0800 hr. The rats in group 2 were administered subcutaneous injections of 200 µg/kg of rhGH twice daily at 0800 and 2000 hr. The rats in group 3 (control group) were administered daily subcutaneous injections of saline at 0800 hr. The animals were treated in this manner for 9 days.
All animal experiments were performed with the approval of the Institutional Animal Care and Use Committee, Laboratory Animal Research Center, Samsung Biomedical Research Institute, Seoul, Korea. All procedures conformed to the international guidelines for the ethical use of laboratory animals and efforts were made to minimize the number of animals used and to avoid any unnecessary suffering.
Spatial learning and memory were evaluated in the GH-treated and control rats by using a modification of the MWM performance test (16). The MWM test is a memory test based on the capacity of animals to rescue themselves by reaching a hidden goal platform in a pool of water (16). The MWM test was conducted in a circular water-maze tank (diameter, 157 cm and height, 60 cm) filled with water (temperature, 24±1℃) that had been opacified by adding powdered milk. A transparent Plexiglass platform (diameter, 10 cm and height, 47 cm) was submerged 3 cm below the water surface and placed in the one of the quadrants. The maze was located in the center of a well-lit room and was surrounded by black curtains (placed at 50 cm from the pool periphery) that contained 4 distinct visual cues. The swimming path of each rat was monitored by an overhead video camera connected to a personal computer and was analyzed by an automated tracking system (Smart v.20®; Panlab SL, Barcelona, Spain) (Fig. 1). During the training period, the animals were required to locate the hidden platform, which remained in the same position, in relation to the external visual cues. The training was carried out in 1 block of 3 trials per day. To begin each trial, the rats were placed in the water facing the maze wall in one of the 3 quadrants except the target quadrant containing the hidden platform. The daily order of entry into individual quadrants was randomized. Each trial ended once the animals found the platform; if the rats were unable to find the platform within 90 sec, they were guided toward it by an experimenter. After a period of 30 sec on the platform, the rats were immediately re-placed at a different start position for the next trial. At the end of a training session, the rats were dried and returned to their respective home cages. In each trial, the time taken for the rat to reach the platform (escape latency in seconds), the length of the swim path (distance in centimeters), and the swimming speed (centimeters/second) were measured. The water maze training procedure began on experimental day 5, and the test was given for 5 consecutive days. On day 9, probe trials were conducted at 2 hr after completion of training in order to evaluate spatial bias. The rats were released from the quadrant opposite to where the platform was located and were allowed to swim for 90 sec in the absence of the platform. We measured the percentage of time that the rats spent in the area corresponding exactly to the area occupied by the platform during the training period. Considering the fact that rats are nocturnal animals, all MWM tests were performed during the dark cycle.
On day 10, the animals were euthanized, and their tibias were harvested and fixed in 10% neutral buffered formalin. The fixed tibias were split along the frontal plane at the proximal end. The tibias were processed for paraffin embedding, sectioned, and stained with hematoxylin and eosin. The measurement of the width of the proximal tibial growth plate was performed on the left tibia. We obtained 3 measurements per section (medial, central, and lateral) at a magnification of ×100 under a light microscope. In all rats, the means of the 3 sections were averaged to obtain the width of the proximal tibial growth plate.
Due to its nature, the data were analyzed by non-parametric means and have been expressed as the median and interquartile ranges. The Kruskal-Wallis test was used for comparing the medians of several variables among the groups. When the differences between the medians were significant, a multiple comparison test using LSD was performed. Analysis of weight gain and improvement in the MWM test were evaluated using the Wilcoxon signed-ranks test. All statistical analyses were performed using SPSS version 11 (SPSS Inc., Chicago, IL, U.S.A.), and P<0.05 was considered as statistically significant.
We compared the relative dose-dependent effects of GH on stimulation of somatic growth in HYPOX rats. The growth parameters measured included weight gain and bone growth (the width of the tibial growth plate). Cumulative body weight gain and the width of the tibial growth plate in the experimental groups are presented in Table 1. The daily body weights of the groups are shown in Fig. 2.
There was no difference in the baseline body weight among the 3 groups. However, body weight development differed significantly, as observed by comparing the weight before GH treatment with that after GH treatment. Body weight remained constant in the control rats (P=1.000). It was expected because no weight gain was the postulation of adequate hypophysectomy. A significant weight gain was demonstrated when GH was administered by either once daily injection (median weight gain, 12 g; P=0.042) or twice daily injection (median weight gain, 22 g; P=0.043). The rats which received 200 µg/kg of GH twice daily gained significantly more weight than those who received 200 µg/kg of GH once daily (P=0.003) or saline (P=0.000). The cumulative weight gain of the rats which received 200 µg/kg of GH twice daily was approximately twice that of the rats who received 200 µg/kg of GH once daily, suggesting that GH stimulated an increase in the body weight of HYPOX rats in a dose-dependent manner.
Similar relative results among the 3 groups were obtained on measurement of the width of the tibial growth plate (Table 1). In rats which received 200 µg/kg of GH twice daily, the tibial growth plate was significantly wider as compared to the rats which received 200 µg/kg GH once daily (P=0.040) or saline (P=0.005). However, there was no significant difference in the width of the tibial growth plate between groups 1 and 3 (P=0.263). The representative stained sections of the tibial growth plate of HYPOX rats from the different experimental groups are shown in Fig. 3.
The MWM test was performed from experimental days 5 to 9. The escape latencies of the HYPOX rats on each day during the training period are shown in Fig. 4. On day 5, the median escape latency in groups 1, 2, and 3 was 61.5, 58.5, and 62.1 sec, respectively. On day 9, i.e., after training for 5 days, the median escape latency in groups 1, 2, and 3 was 35.6, 35.6, and 36.3 sec, respectively. In other words, during memory acquisition testing over the day, the escape latency was noted to have decreased in all groups. However, the decrease was statistically significant only in group 2 (P=0.043) and group 3 (P=0.043). There was no difference in the swimming speed among the groups or within the rats in a group on any day. Thus, the decrease in the escape latency indicated that the HYPOX rats in all the 3 groups were learning the behavioral task. On the last day of training, there was no difference in the escape latency among the 3 groups. However, the daily rate of decrease in escape latency was higher in the GH-treated groups 1 and 2 than in the control group. On day 7, the escape latency in the GH-treated groups 1 and 2 was significantly shorter than that in the control group (P=0.000 and P=0.032, respectively). This result indicated that GH-treated HYPOX rats had more rapid memory acquisition in the MWM test. On day 7, the rats which received 200 µg/kg of GH once daily had a shorter escape latency than those who received 200 µg/kg of GH twice daily (P=0.023); therefore, it could not be demonstrated that the higher the dose of GH, the better was the performance in the MWM test. On day 9, probe trials were conducted at 2 hr after completion of training in order to evaluate spatial bias. There was no difference between the percentage of time spent by the rats in the area corresponding exactly to the area occupied by the platform during the training period.
The purpose of the present study was to examine the effects of different GH doses on 1) somatic growth as estimated from body weight and width of the tibial growth plate and 2) spatial performance as examined by the MWM test in HYPOX female Sprague-Dawley rats. HYPOX rats have multiple pituitary hormone deficiencies, including GHD. We followed the USP procedure to obtain the HYPOX rats. According to this procedure, the HYPOX rats would not gain weight due to multiple pituitary hormone deficiencies, which, according to the USP is a standard method for GH bioassay using an animal model. HYPOX rats have been used to evaluate the effects of GH in several previous studies (14, 17, 18). Our results suggest that rhGH replacement therapy stimulates rapid weight gain accompanied with actual bone growth (measured by the width of the tibial growth plate) in HYPOX rats. Although there was no significant statistical difference in the width of the tibial growth plate between groups 1 and 3, median of the width of the tibial growth plate in group 1 was approximately 40 µm wider than group 3. Moreover, the same GH doses that stimulated growth also improved spatial performance as observed in the MWM test. Spatial memory is a form of long-term memory. In this study, rats which received GH had more rapid spatial memory acquisition than the control rats. On the third day of trial of the MWM test, the escape latency of the GH-treated rats was significantly shorter than that of the control rats. However, it was not obvious whether GH affects spatial memory in a dose-dependent manner because the escape latency in rats who received 200 µg/kg of GH once daily was shorter than that in rats who received 200 µg/kg of GH twice daily, indicating that a GH dose of 200 µg/kg/day is sufficient to improve spatial memory as observed in the MWM test. After training over five days, the spatial performance of all HYPOX rats had improved as observed in the MWM test, indicating that they were learning the behavioral task. And there was no difference in the escape latency among the groups on the fifth day of trial. It is considered that the effect of repeated trial exceeds the effect of GH after five days. And it is also possible that GH receptors in the brain are saturated in GH-treated rats.
Madeleine et al. (14) reported similar results. They administered 1 mg/kg of GH twice daily to HYPOX rats in order to evaluate spatial memory using the MWM test. The rhGH-treated rats had a significantly better spatial memory task performance than the control rats on the second and third days of the trial. Further training eliminated this difference between the groups, which was consistent with our results.
In the previous study (19), administration of twice daily GH injections (1 mg/kg) did not increase the serum IGF-I concentration in GH-deficient rats. In contrast, continuous infusions of GH (2.4 mg/kg/day) resulted in a 5-fold increase in the serum IGF-I concentration. Both modalities of GH administration caused significant weight gain, although the growth rate was significantly greater in rats which were administered GH by continuous infusion. The increase in the serum IGF-I concentration by continuous GH infusion may be due to up-regulation of the liver membrane GH receptors (20). Moreover, GH also has a short circulating half-life (21). Therefore, a more frequent dosing schedule would enhance IGF-I production and provide a pattern of circulating GH and IGF-I that more closely resembles the physiological state (22). Therefore, here, twice daily administration of GH was employed in order to increase the daily dose of GH. We did not measure the serum IGF-I concentration because, according to a previous study, twice daily injection of GH could not be expected to increase serum IGF-I concentrations (19).
It has been reported that GH acts directly on the growth plate, and the presence of GH receptors on rat chondrocytes (23) and human growth plates (24) has been demonstrated. Therefore, we selected body weight and the width of the tibial growth plate instead of serum IGF-I concentration as the measurement criteria to evaluate the effects of GH on somatic growth and to minimize stressful events such as blood sampling, which might have influenced the performance in the MWM test
Our results suggest that GH stimulates an increase in the body weight and the width of the tibial growth plate in HYPOX rats in a dose-dependent manner. Short-term GH treatment of adults with GHD increases body sodium and extracellular water resulting in an increase in the body weight (25); thus, the increase in the width of the tibial growth plate in this study reflects the fact that short-term GH replacement therapy stimulates actual organ growth. Further, the doses employed in our study (200 µg/kg of rhGH once or twice daily) have physiological effects on HYPOX rats. Generally, the dosage of GH used in children with GHD is 166-236 µg/kg/day.
It is well known that longitudinal bone growth is the result of chondrocyte proliferation and subsequent proliferation of the growth plate of a long bone (26). It has been reported that in cultured growth plate chondrocytes, GH acts on the resting zone chondrocytes and is responsible for local IGF-I production, which stimulates clonal expansion of proliferating chondrocytes in an autocrine/paracrine manner (27). Since IGF-I gene deletion causes dwarfism in mice (28) and extreme short stature in humans (29), IGF-I is believed to play an important role in longitudinal bone growth. Nevertheless, the finding that administration of GH increases the width of the growth plate germinal zone in IGF-I-deficient mice indicates a direct role of GH in proliferation of the growth plate (28).
A more interesting issue is the relationship between GH and cognitive function. Children with GHD have significant cognitive deficits, which may be moderated by GH treatment (30). Several studies have demonstrated that in adults with GHD, GH improves the quality of life and cognitive performance (8). There is also emerging evidence that the decline in GH and IGF-I observed with advancing age may contribute to the impairment in cognitive function associated with aging (10, 11, 31), and such age-related changes may be arrested or partially reversed by hormonal supplementation (12). In addition, it has been suggested that in the healthy elderly, a positive correlation exists between IGF-I levels and cognition (11).
The association between GH and cognitive processes has also been supported by studies on rats. GH was found to facilitate long-term memory in young but not in elderly rats; in contrast, the extinction response as recorded in a behavioral assay was affected in both young and aged rats (13).
The physiological and molecular mechanisms behind the improvement in cognitive functions after GH treatment are unclear. The existence of GH-binding sites in brain areas, such as the hippocampus, hypothalamus, putamen, and choroid plexus, has been reported (32). The action of GH in the central nervous system could be due to its direct effects on cells or due to GH-induced release of secondary mediators, such as IGF-I, that can cross the blood-brain barrier. It has been demonstrated that subcutaneous administration of physiological doses of GH to patients with GHD is associated with an increase in GH concentration in the cerebrospinal fluid (33). In addition, IGF-I receptors are widely distributed in the brain (34). Administration of IGF-I improved working memory and reference memory in the MWM test in male rats (35).
In a mouse model, it was suggested that the NMDA receptors in the hippocampus were an essential component of long-term potentiation, which plays an important role in spatial memory acquisition (36). The hippocampus is considered to be vital in multiple cognitive processes, particularly learning and memory. Lesions of the hippocampus and pharmacological blockade of the NMDA receptors disrupt spatial learning (16, 37). In comparison, increased expression of the NR2B subunit of the NMDA receptor in the forebrain enhances hippocampal learning (38). These findings have led to the view that normal spatial learning requires the integrity of NMDA receptor-mediated plasticity in the hippocampus (39).
The NMDA receptor complex consists of several types of subunits, including type 1 (NR1), type 2A (NR2A), and type 2B (NR2B) (40), and it is closely related to the PSD-95 protein family, which is thought to be responsible for the molecular organization of the synaptic NMDA receptors and subsequent signal transduction (41).
Madeleine et al. (42) demonstrated that GH may elicit an increase in the hippocampal GH receptor and NMDA receptor subunit NR2B gene transcript levels in young male Sprague-Dawley rats. They also confirmed a distinct interaction between GH and the NMDA receptor, which was supported by the positive correlation between the GH receptor and NR2B mRNA levels. However, the molecular mechanism by which GH influenced the expression of the NMDA receptor subunit gene transcripts in the hippocampus was unclear. Therefore, they examined the effect of daily subcutaneous injection of human IGF-I for 10 days in male rats belonging to different age groups (43). The result was that the NR2B subunit mRNA expression increased in the young (age, 11 weeks) but not in the elderly (age, 14-16 months) rats, and NR2A mRNA expression decreased in both groups. These results suggested that IGF-I might have mediated the actions of GH. Thereafter, they reported that rhGH treatment of HYPOX rats did not alter expression level of the NMDA receptor subunit NR2B gene, although there were improvements in the MWM test in the GH-treated HYPOX rats. However, the NR2A and PSD-95 gene transcript levels were elevated by rhGH treatment. Therefore, they suggested that the NR2A subunit was likely to play an important role in learning, and there was a link between the performance in the MWM test and PSD-95.
Another study demonstrated that hippocampus-dependent learning ability selectively correlates with the NR1 subunit immunofluorescence levels in the CA3 neurons of the hippocampus (39). From these results, it is obvious that hippocampal NMDA receptors may play an important role in learning and memory. However, most critical subunit of the NMDA receptor remains unclear.
In conclusion, this study demonstrates that 1) GH replacement therapy in HYPOX female Sprague-Dawley rats promotes increase in somatic growth as observed from measurement of the body weight and the width of the tibial growth plate and 2) GH-treated HYPOX rats show more rapid spatial memory acquisition in the MWM test. The dose of GH sufficient for improving the performance in the MWM test was suggested to be lower than that necessary for stimulating somatic growth in HYPOX rats. We were unable to demonstrate that rats who received 200 µg/kg of GH twice daily performed better in the MWM test than those who received 200 µg/kg of GH once daily.
We also suggest that parameters such as body weight, the width of the tibial epiphyseal plate, and spatial performance in the MWM test may be appropriate to evaluate the effects of GH administration by other routes, such as oral or intratracheal. Further studies are needed to explain the basic mechanisms of the effects of GH on cognitive function. And, the optimal doses of GH for improving cognitive function in human must be decided.
Figures and Tables
Fig. 1
The automated tracking system (Smart v.20®) used in the Morris water maze (MWM) test. There is a circular platform in the one of the quadrants. The 3 quadrants except the target quadrant that contained the hidden platform were used for entry during each trial on a particular day. The escape latency (the time taken to reach the hidden platform), the length of the swim path, and the swimming speed were measured automatically.

Fig. 2
Effects of rhGH on body weight of HYPOX rats. Body weights were measured over 10 days. GH was administered for 9 days. Weight gain in each group was analyzed using the Wilcoxon signed-ranks test. A significant weight gain was evident only in groups 1 and 2. Data are represented as the median and interquartile ranges (5 rats per group).
Group 1: rhGH 200 µg/kg subcutaneously once daily, P=0.042.
Group 2: rhGH 200 µg/kg subcutaneously twice daily, P=0.043.
Group 3: Saline subcutaneously once daily, P=1.000.

Fig. 3
Representative stained sections of the tibial growth plates of HYPOX rats. Arrows indicate the width of the proximal tibial growth plate. (A) Group 1: rhGH 200 µg/kg subcutaneously once daily, (B) Group 2: rhGH 200 µg/kg subcutaneously twice daily, (C) Group 3: Saline subcutaneously once daily. The width of the tibial growth plate is thicker in B than in A and C (P=0.040 and P=0.005, respectively) (H&E stained, ×100).
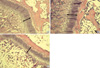
Fig. 4
Effects of rhGH treatment of HYPOX rats on performance in the MWM test from experimental days 5 to 9. The training was carried out in 1 block of 3 trials per day for 5 consecutive days. Improvements in the performance in the MWM test were evaluated using the Wilcoxon signed-ranks test (Group 1, P=0.080; Group 2, P=0.043; Group 3, P=0.043). *P=0.027, using Kruskal-Wallis test with Bonferroni's correction. The data obtained on day 7 were subjected to a multiple comparison test was performed using LSD. Data are represented as the median and interquartile ranges (5 rats per group).
Group 1: rhGH 200 µg/kg subcutaneously once daily.
Group 2: rhGH 200 µg/kg subcutaneously twice daily.
Group 3: Saline subcutaneously once daily.

Table 1
Effects of rhGH replacement therapy for 9 days on body weight gain and the width of the proximal tibial growth plate in hypophysectomized (HYPOX) rats (5 rats per group)

Data are expressed as the median and interquartile ranges.
Group 1, rhGH 200 µg/kg subcutaneously once daily; Group 2, rhGH 200 µg/kg subcutaneously twice daily; Group 3, Saline subcutaneously once daily.
*P=0.982; †P=0.002, Group 1 vs. Group 2: P=0.003, Group 1 vs. Group 3: P=0.001, Group 2 vs. Group 3: P=0.000; ‡P=0.028, Group 1 vs. Group 2: P=0.040, Group 1 vs. Group 3: P=0.263, Group 2 vs. Group 3: P=.005.
Statistical analysis: Kruskal-Wallis test with post hoc LSD.
References
1. Hindmarsh PC, Dattani MT. Use of growth hormone in children. Nat Clin Pract Endocrinol Metab. 2006. 2:260–268.

2. Underwood LE, Attie KM, Baptista J. Growth hormone (GH) dose-response in young adults with childhood-onset GH deficiency: a two-year, multicenter, multiple-dose, placebo-controlled study. J Clin Endocrinol Metab. 2003. 88:5273–5280.

3. Bengtsson BA, Eden S, Lonn L, Kvist H, Stokland A, Lindstedt G, Bosaeus I, Tolli J, Sjostrom L, Isaksson OG. Treatment of adults with growth hormone (GH) deficiency with recombinant human GH. J Clin Endocrinol Metab. 1993. 76:309–317.

4. O'Halloran DJ, Tsatsoulis A, Whitehouse RW, Holmes SJ, Adams JE, Shalet SM. Increased bone density after recombinant human growth hormone (GH) therapy in adults with isolated GH deficiency. J Clin Endocrinol Metab. 1993. 76:1344–1348.
5. Cuneo RC, Salomon F, Watts GF, Hesp R, Sonksen PH. Growth hormone treatment improves serum lipids and lipoproteins in adults with growth hormone deficiency. Metabolism. 1993. 42:1519–1523.

6. Salomon F, Cuneo RC, Hesp R, Sonksen PH. The effects of treatment with recombinant human growth hormone on body composition and metabolism in adults with growth hormone deficiency. N Engl J Med. 1989. 321:1797–1803.

7. Gibney J, Wallace JD, Spinks T, Schnorr L, Ranicar A, Cuneo RC, Lockhart S, Burnand KG, Salomon F, Sonksen PH, Russell-Jones D. The effects of 10 years of recombinant human growth hormone (GH) in adult GH-deficient patients. J Clin Endocrinol Metab. 1999. 84:2596–2602.

8. Deijen JB, de Boer H, van der Veen EA. Cognitive changes during growth hormone replacement in adult men. Psychoneuroendocrinology. 1998. 23:45–55.

9. Sathiavageeswaran M, Burman P, Lawrence D, Harris AG, Falleti MG, Maruff P, Wass J. Effects of GH on cognitive function in elderly patients with adult-onset GH deficiency: a placebo-controlled 12-month study. Eur J Endocrinol. 2007. 156:439–447.

10. van Dam PS, Aleman A, de Vries WR, Deijen JB, van der Veen EA, de Haan EH, Koppeschaar HP. Growth hormone, insulin-like growth factor I and cognitive function in adults. Growth Horm IGF Res. 2000. 10:Suppl B. S69–S73.
11. Aleman A, Verhaar HJ, De Haan EH, De Vries WR, Samson MM, Drent ML, Van der Veen EA, Koppeschaar HP. Insulin-like growth factor-I and cognitive function in healthy older men. J Clin Endocrinol Metab. 1999. 84:471–475.

12. Vitiello MV, Moe KE, Merriam GR, Mazzoni G, Buchner DH, Schwartz RS. Growth hormone releasing hormone improves the cognition of healthy older adults. Neurobiol Aging. 2006. 27:318–323.

13. Schneider-Rivas S, Rivas-Arancibia S, Vazquez-Pereyra F, Vazquez-Sandoval R, Borgonio-Perez G. Modulation of long-term memory and extinction responses induced by growth hormone (GH) and growth hormone releasing hormone (GHRH) in rats. Life Sci. 1995. 56:PL433–PL441.

14. Le Greves M, Zhou Q, Berg M, Le Greves P, Fholenhag K, Meyerson B, Nyberg F. Growth hormone replacement in hypophysectomized rats affects spatial performance and hippocampal levels of NMDA receptor subunit and PSD-95 gene transcript levels. Exp Brain Res. 2006. 173:267–273.

15. Scoville WB, Milner B. Loss of recent memory after bilateral hippocampal lesions. J Neuropsychiatry Clin Neurosci. 2000. 12:103–113.

16. Morris RG, Garrud P, Rawlins JN, O'Keefe J. Place navigation impaired in rats with hippocampal lesions. Nature. 1982. 297:681–683.

17. Cox GN, Rosendahl MS, Chlipala EA, Smith DJ, Carlson SJ, Doherty DH. A long-acting, mono-PEGylated human growth hormone analog is a potent stimulator of weight gain and bone growth in hypophysectomized rats. Endocrinology. 2007. 148:1590–1597.

18. Nagamine J, Nakagawa T, Taiji M. Recombinant human growth hormone (SMP-140) is effective for growth promotion in hypophysectomized rats. Biomed Res. 2006. 27:191–195.

19. Gargosky SE, Tapanainen P, Rosenfeld RG. Administration of growth hormone (GH), but not insulin-like growth factor-I (IGF-I), by continuous infusion can induce the formation of the 150-kilodalton IGF-binding protein-3 complex in GH-deficient rats. Endocrinology. 1994. 134:2267–2276.

20. Bick T, Amit T, Barkey RJ, Hertz P, Youdim MB, Hochberg Z. The interrelationship of growth hormone (GH), liver membrane GH receptor, serum GH-binding protein activity, and insulin-like growth factor I in the male rat. Endocrinology. 1990. 126:1914–1920.

21. Chapman IM, Helfgott A, Willoughby JO. Disappearance half-life times of exogenous and growth hormone-releasing factor-stimulated endogenous growth hormone in normal rats. J Endocrinol. 1991. 128:369–374.

22. MacGillivray MH, Baptista J, Johanson A. Outcome of a four-year randomized study of daily versus three times weekly somatropin treatment in prepubertal naive growth hormone-deficient children. Genentech Study Group. J Clin Endocrinol Metab. 1996. 81:1806–1809.

23. Gevers EF, van der Eerden BC, Karperien M, Raap AK, Robinson IC, Wit JM. Localization and regulation of the growth hormone receptor and growth hormone-binding protein in the rat growth plate. J Bone Miner Res. 2002. 17:1408–1419.

24. Werther GA, Haynes K, Edmonson S, Oakes S, Buchanan CJ, Herington AC, Waters MJ. Identification of growth hormone receptors on human growth plate chondrocytes. Acta Paediatr Suppl. 1993. 82:suppl 391. 50–53.

25. Hoffman DM, Crampton L, Sernia C, Nguyen TV, Ho KK. Short-term growth hormone (GH) treatment of GH-deficient adults increases body sodium and extracellular water, but not blood pressure. J Clin Endocrinol Metab. 1996. 81:1123–1128.

26. Stevens DA, Williams GR. Hormone regulation of chondrocyte differentiation and endochondral bone formation. Mol Cell Endocrinol. 1999. 151:195–204.

27. Isaksson OG, Lindahl A, Nilsson A, Isgaard J. Mechanism of the stimulatory effect of growth hormone on longitudinal bone growth. Endocr Rev. 1987. 8:426–438.

28. Wang J, Zhou J, Bondy CA. Igf1 promotes longitudinal bone growth by insulin-like actions augmenting chondrocyte hypertrophy. FASEB J. 1999. 13:1985–1990.

29. Woods KA, Camacho-Hubner C, Savage MO, Clark AJ. Intrauterine growth retardation and postnatal growth failure associated with deletion of the insulin-like growth factor I gene. N Engl J Med. 1996. 335:1363–1367.

30. van der Reijden-Lakeman IE, de Sonneville LM, Swaab-Barneveld HJ, Slijper FM, Verhulst FC. Evaluation of attention before and after 2 years of growth hormone treatment in intrauterine growth retarded children. J Clin Exp Neuropsychol. 1997. 19:101–118.
31. Rollero A, Murialdo G, Fonzi S, Garrone S, Gianelli MV, Gazzerro E, Barreca A, Polleri A. Relationship between cognitive function, growth hormone and insulin-like growth factor I plasma levels in aged subjects. Neuropsychobiology. 1998. 38:73–79.

32. Lai Z, Roos P, Zhai O, Olsson Y, Fholenhag K, Larsson C, Nyberg F. Age-related reduction of human growth hormone-binding sites in the human brain. Brain Res. 1993. 621:260–266.

33. Johansson JO, Larson G, Andersson M, Elmgren A, Hynsjo L, Lindahl A, Lundberg PA, Isaksson OG, Lindstedt S, Bengtsson BA. Treatment of growth hormone-deficient adults with recombinant human growth hormone increases the concentration of growth hormone in the cerebrospinal fluid and affects neurotransmitters. Neuroendocrinology. 1995. 61:57–66.

34. LeRoith D, Werner H, Beitner-Johnson D, Roberts CT Jr. Molecular and cellular aspects of the insulin-like growth factor I receptor. Endocr Rev. 1995. 16:143–163.

35. Markowska AL, Mooney M, Sonntag WE. Insulin-like growth factor-1 ameliorates age-related behavioral deficits. Neuroscience. 1998. 87:559–569.

36. Tsien JZ, Huerta PT, Tonegawa S. The essential role of hippocampal CA1 NMDA receptor-dependent synaptic plasticity in spatial memory. Cell. 1996. 87:1327–1338.

37. Steele RJ, Morris RG. Delay-dependent impairment of a matching-to-place task with chronic and intrahippocampal infusion of the NMD-Aantagonist D-AP5. Hippocampus. 1999. 9:118–136.

38. Tang YP, Shimizu E, Dube GR, Rampon C, Kerchner GA, Zhuo M, Liu G, Tsien JZ. Genetic enhancement of learning and memory in mice. Nature. 1999. 401:63–69.

39. Adams MM, Smith TD, Moga D, Gallagher M, Wang Y, Wolfe BB, Rapp PR, Morrison JH. Hippocampal dependent learning ability correlates with N-methyl-D-aspartate (NMDA) receptor levels in CA3 neurons of young and aged rats. J Comp Neurol. 2001. 432:230–243.

40. Paoletti P, Neyton J. NMDA receptor subunits: function and pharmacology. Curr Opin Pharmacol. 2007. 7:39–47.

41. Garner CC, Nash J, Huganir RL. PDZ domains in synapse assembly and signalling. Trends Cell Biol. 2000. 10:274–280.

42. Le Greves M, Steensland P, Le Greves P, Nyberg F. Growth hormone induces age-dependent alteration in the expression of hippocampal growth hormone receptor and N-methyl-D-aspartate receptor subunits gene transcripts in male rats. Proc Natl Acad Sci U S A. 2002. 99:7119–7123.

43. Le Greves M, Le Greves P, Nyberg F. Age-related effects of IGF-1 on the NMDA-, GH- and IGF-1-receptor mRNA transcripts in the rat hippocampus. Brain Res Bull. 2005. 65:369–374.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download