Abstract
This study was undertaken to evaluate the therapeutic effects of topical ozonated olive oil on acute cutaneous wound healing in a guinea pig model and also to elucidate its therapeutic mechanism. After creating full-thickness skin wounds on the backs of guinea pigs by using a 6 mm punch biopsy, we examined the wound healing effect of topically applied ozonated olive oil (ozone group), as compared to the pure olive oil (oil group) and non-treatment (control group). The ozone group of guinea pig had a significantly smaller wound size and a residual wound area than the oil group, on days 5 (P<0.05) and 7 (P<0.01 and P<0.05) after wound surgery, respectively. Both hematoxylin-eosin staining and Masson-trichrome staining revealed an increased intensity of collagen fibers and a greater number of fibroblasts in the ozone group than that in the oil group on day 7. Immunohistochemical staining demonstrated upregulation of platelet derived growth factor (PDGF), transforming growth factor-β (TGF-β) and vascular endothelial growth factor (VEGF) expressions, but not fibroblast growth factor expression in the ozone group on day 7, as compared with the oil group. In conclusion, these results demonstrate that topical application of ozonated olive oil can accelerate acute cutaneous wound repair in a guinea pig in association with the increased expression of PDGF, TGF-β, and VEGF.
The closure of cutaneous wounds involves complex tissue movements such as hemorrhage, inflammation, re-epithelization, granulation tissue formation, and the late remodeling phase of repair (1). These events involve coordination of dozens of types of cells and matrix proteins, which are all important to control stages of the repair process. Previous studies have demonstrated that endogenous growth factors, such as fibroblast growth factors (FGF) (2), platelet derived growth factors (PDGF) (3), transforming growth factor-β (TGF-β) (1) and vascular endothelial growth factors (VEGF) (4) are the important regulatory polypeptides for coordinating the healing process. They are released from macrophages, fibroblasts, and keratinocytes at the site of injury and they participate in the regulation of re-epithelization, granulation tissue formation, collagen synthesis and neovascularization.
Ozone (O3) has been widely recognized as one of the best bactericidal, antiviral and antifungal agents (5, 6) and it has been used empirically as a clinical therapeutic agent for chronic wounds, such as trophic ulcers, ischemic ulcers and diabetic wounds (7-9). The beneficial effects of O3 on wound healing might be assumed to be due to decreased bacterial infection, ameliorated impaired dermal wound healing or increased oxygen tension by O3 exposure in the wound area (10, 11).
It was reported that O3 exposure is associated with activation of transcription factor NF-κB; this is important to regulate inflammatory responses and eventually the entire process of wound healing (5, 12, 13). It was shown that huge amounts of PDGF and TGF-β1 were released from platelets in the heparinized plasma of a limb ischemia patient after ozonation (14, 15). It has been revealed that there were substantial increases of steady-state mRNA levels of TGF-β1 in the fibroblasts that were co-cultured with bronchoepithelial cells after O3 exposure (16). A recent study has shown that hydrogen peroxide (H2O2) potently induced the VEGF expression in human keratinocytes which can stimulate wound healing (17). From these previous studies of O3, we hypothesized that O3 might enhance acute cutaneous wound healing, and this could be associated with growth factors such as FGF, PDGF, TGF-β and VEGF.
Nowadays, O3 is profitably and practically employed as ozonated olive oil; this contains the O3 molecule stabilized as an ozonide between the double bonds of a monounsaturated fatty acid such as oleic acid, which is ideal for the topical use of O3 to treat chronically infected cutaneous and mucosal areas of the body (5). Ozonated materials referred to as ozonides are formed by the reaction of olefins with ozone. Any olefin can be treated with gaseous ozone to form an ozonide. The ozonide compositions have the capacity to deliver nascent oxygen deep within the lesion without causing primary skin irritation.
Ozonated oil has been used topically for the treatment of chronic wounds, but there have been few studies concerning with the therapeutic effects of ozonated olive oil on acute cutaneous wound healing in animal models. The present study was designed to evaluate the therapeutic effect of topical ozonated olive oil on acute cutaneous wound healing in a guinea pig model, and to elucidate its therapeutic mechanisms that are associated with such growth factors as FGF, PDGF, TGF-β, and VEGF.
Sixteen female guinea pigs (400-450 g), aged 8-9 weeks, were placed in a room that was under a 12-hr light and 12-hr dark cycle. The animals were placed individually in separate cages with ad libitum access to food and water. The animal care, handling, and experimental procedures were carried out in accordance with a protocol approved by the Animal Care and Use Committee of the Catholic University of Korea.
After 7 days of acclimation, the guinea pigs were anesthesized with ketamine and their backs were shaved and then sterilized with normal saline. The skin was then pinched and folded, and a sterile biopsy punch (6 mm in diameter, Stiefel Co., Offenbach, Germany) was used to make a full-thickness hole in the skin. Two wounds were created on both sides of the back for a total 4 circular wounds per animal.
Two drops (about 0.1 mL) of ozonated olive oil (OZOO®, Aurora, Inc., Miami, FL, U.S.A.) were applied everyday to two sites of the four wounds (Ozone group). Olive oil (OLO) as a pure base was applied to a third wound (Oil group). As a control group, nothing was applied on the fourth wound. The wounds were then dressed with Opsite® (Smith&Nephew, Hull, U.K.) to cover them without dryness. An elastic bandage was wrapped around the area to prevent further injury.
After each guinea pig was wounded as described above, the wounds were photographed at indicated times by using a digital camera (Canon 350D, Canon Inc., Tokyo, Japan) that was placed at a 20 cm distance to help eliminate error in measuring the size of the wounds. The photographic images were analyzed by Canvas X10.0 software (ACDSEE Inc., Miami, FL, U.S.A.) and the area of wound closure was measured by using the same program. The parameters of the wounds, such as wound size and the residual wound area, were measured daily from the photographic images taken.
* Measured parameters
1) Wound size (mm2)
Analysis of clinical wound closure in each group of wounds was performed through digital processing on days 0, 3, 5, 7, and 11.
On days 3 (n=4), 7 (n=4), and 11 (n=8) after wounding, guinea pigs were euthanized using ketamine and the four wounded tissues from each guinea pig were excised. Then the tissues were cut into halves; one half was placed in formalin (10% formaldehyde in phosphate-buffered saline) for hematoxylin-eosin and Masson-trichrome staining, and the second half was quickly kept as a frozen state (-80℃) for immunohistochemistry.
The specimens for histological examination were collected from each group by the full-thickness excision. Intensity of the collagen fibers and the fibroblast proliferation were examined under microscope in hematoxylin-eosin and Masson-trichrome staining. The staining intensity of the collagen fibers was graded under ×200 magnification as follows: -, completely negative staining intensity; ±, lower staining intensity; +, moderate staining intensity; ++, slightly higher staining intensity; +++, considerably higher staining intensity. The number of fibroblasts was counted in the 5 randomized fields per specimen under ×400 magnification. Three dermatologists, who were "blinded" to which groups the specimens were in, independently analyzed all the specimens and then the mean numbers and standard deviations of the fibroblasts in all groups of wound were calculated, respectively.
All the cryostat sections (5 µm) of the healing wounds of guinea pigs were placed on coated micro slides (Muto-glass, Tokyo, Japan) for immunohistochemical staining procedures. The samples for immunohistochemistry were washed several times with 0.1 M phosphate buffered saline (PBS) and then reacted for 15 min with 0.3% H2O2 solution diluted with PBS to inactivate the endogenous peroxidase in the tissues. After washing three times for 10 min with PBS, the nonspecific antigen-antibody reactions were inhibited by a 1 hr treatment with 2.5% normal horse serum (R.T.U. Vectastatin® Universal ABC Elite kit, Vector Laboratories, Inc., Burlingame, CA, U.S.A.). The samples were reacted in 1:100 diluted solutions of each of the primary antibodies such as rabbit anti-FGF serum (Santa Cruz Biotechnology Inc., Delaware, CA, U.S.A.), anti-PDGF serum (Santa Cruz Biotechnology Inc.), anti-TGF-β serum (Santa Cruz Biotechnology Inc.), and anti-VEGF serum (Santa Cruz Biotechnology Inc.) at 4℃ for 24 hr. After the primary antibody reaction, each sample was washed three times for 10 min with PBS, and then they were reacted at room temperature for 1 hr with treatment of the secondary antibody solution, biotinylated horse anti-IgG (R.T.U. Vectastatin® Universal ABC Elite kit). After washing three times for 10 min each with PBS, the samples were reacted at room temperature for 1 hr with avidin-biotin-peroxidase complex solution (R.T.U. Vectastatin® Universal ABC Elite kit). For the chromogen reactions, the Vector® Nova-RED™ substrate kit (Vector Laboratories, Inc., Burlingame, CA, U.S.A.) was used with reagents 1, 2, and 3 and H2O2 at room temperature for 3-5 min. After the chromogen reaction, the sample slides were washed three times for 10 min each with PBS. The washed sample tissues were dried out on gelatin-coated slides for 2 hr at room temperature. They are made transparent with xylene treatment and next covered with polymount. Three "blinded" dermatologists then independently analyzed the relative intensity of the immunohistochemical staining of the slides for FGF, PDGF, TGF-β, and VEGF on 5 randomized fields per specimen under ×200 magnification. The intensity was graded as follows: 0, no positive reaction; +, 1-25% of the cells were positively stained; ++, 26-50% of the cells were positively stained; +++, 51-75% of the cells were positively stained; ++++, 76-100% of the cells were positively stained. The mean values of the scores were analyzed for interpreting the histochemical reactions.
All statistical analyses were performed using the ANOVA followed by Turkey-Kramer's multiple-comparison test, or Kruskal-Wallis test of variance with Bonferroni's correction. SPSS v10.0 (SPSS Inc., Chicago, IL, U.S.A.) was used for all calculations. All values were expressed as means and standard deviations. Differences were considered significant if P<0.05.
As shown in Fig. 1, there was an enhanced wound closure in the ozone group of wounds, the left two of the four wounds, as compared to the oil group as well as the control group. On day 11, all of the wounds completely re-epithelized irrespective of treatments. The ozone group showed a significantly smaller wound size than the oil group on days 5 (P<0.05) and 7 (P<0.01). The ozone group showed 58%, 46.3%, and 16.4% in residual wound area on days 3, 5 and 7, respectively. On the other hand, the oil group revealed 61.8%, 53.7% and 25.6% in residual wound area, respectively (Table 1). Thus, there was a significant difference in the residual wound area between the ozone group and the oil group on days 5 (P<0.05) and 7 (P<0.01). On repeated-measures of ANOVA, the ozone group showed a significantly decreased residual wound area as compared to the oil group, as well as the control group (P<0.05).
On the hematoxylin-eosin staining, epithelization, infiltration of inflammatory cells and vascular proliferations were commonly seen on all of the wounds; but there were proliferation of fibroblasts and collagen fibers noted in the ozone group as well (data not shown). We performed the Massontrichrome staining in order to determine whether ozonated OLO's ability to accelerate wound closure was associated with collagen synthesis and fibroblast proliferation at the wound bed and at the edge of the injury site. The staining intensity of collagen fibers and the number of fibroblasts were evaluated on days 3 and 7. On day 3, the ozone group did not show a significant difference in the staining intensity of collagen fibers and the number of fibroblasts as compared to the oil group. In contrast, on day 7, the ozone group revealed an increased staining intensity of collagen fibers at the wound bed and at the edge of the entire dermis in comparison to the oil and control groups (Fig. 2). On day 7, the numbers of fibroblasts in the ozone group were 62.3 and 35.6 at the wound bed and edge, respectively; but, the numbers of fibroblasts in the oil group were 48.5 and 22.7, respectively (Table 2). Thus, there was a significant difference in the staining intensity of collagen fibers and the number of fibroblasts between the ozone group and the oil group on day 7 (P<0.05), but not on day 3.
In order to determine which growth factors play an important role for the accelerating wound closure associated with the proliferation of fibroblasts and collagen fibers by the ozonated OLO, we evaluated the immunohistochemical staining intensity of FGF, PDGF, TGF-β, and VEGF on day 7 (Fig. 3). FGF expressions were identified in dermal fibroblasts and collagen fibers, but this was barely detected in the epidermis of the control group. There were little differences in the FGF expression between the ozone group and the oil group (Table 3). PDGF was expressed in dermal inflammatory cells, fibroblasts, epidermal cells and keratinocytes of hair follicles of the control group. The ozone group revealed a relatively higher PDGF expression, as compared to the oil group. There was a relatively distinct difference in the dermis between the former and the latter. TGF-β expressions were detected in the dermal fibroblasts, epidermal cells and keratinocytes of the hair follicles of the control group. Likewise, the ozone group showed a relatively increased expression of TGF-β, as compared to the oil group. VEGF expression was identified in dermal fibroblasts, endothelial cells and collagen fibers, but it was barely detected in the epidermis of the control group. The ozone group revealed a relatively increased VEGF expression in both the dermis and the epidermis, as compared to the oil group. These findings demonstrated that the ozone group revealed relatively higher expressions of PDGF, TGF-β, and VEGF, but not FGF than the oil group on day 7.
Two different ozonized solutions, consisting of sunflower ozonized oil and ozonated OLO, have been usually used as a topical form of O3. Because of their antibacterial and antimycotic properties, these ozonated oils have been empirically used for treating superficial bacterial and fungal infections (5, 18). Previous studies showed that O3 could affect the expression of pro-inflammatory cytokines, such as interleukin-1 (IL-1) and tumor necrosis factor-α (TNF-α), and the adaptive inflammatory responses including cyclooxygenase-2 (COX-2) gene activation in keratinocyte via activation of NF-κB (19, 20). It was also demonstrated that O3 exposure increased the expression of proliferating cell nuclear antigen (PCNA) protein and K10, a keratin expressed in well differentiated suprabasal keratinocytes in skin tissues (20, 21). These findings suggested that O3 could induce the keratinocyte proliferation and differentiation and it could affect skin biology.
OLO will react with O3, forming ozonide at room temperature in a mildly exothermic reaction. As a sample of OLO is ozonized, OLO will gain weight as the reaction proceeds. OLO, high in essential fatty acids, is the only media that will allow O3 to remain in an active or nascent state for a long length of time. Two drops of ozonated OLO, about 0.1 mL, were used in our experiment because it was thought that about 0.1 mL was sufficient to cover the 6 mm-sized punch wounds.
Although there are no reports about the toxicological study of ozonated OLO, it was reported that sunflower ozonized oil had no side effects in human-patients (18), except for slight irritation being noted at the epithelium of mice (22) from the previous toxicological studies. Because the O3 molecule can be stabilized as an ozonide between the double bonds of a monounsaturated fatty acid such as oleic acid (5, 23), ozonated OLO is an ideal preparation for the topical form of O3 and it remains stable for 2 yr when stored at 4℃. Furthermore, ozonated OLO is easy to access as a commercialized form and to use for applying to cutaneous wound because of its liquid form. Thus, in this study, we used ozonated OLO as a topical form of O3 to treat cutaneous wound.
Full-thickness punch wounds used in this study, not including panniculectomy, are useful model to evaluate re-epithelization after topical application. Although pigs are often useful models since their cutaneous architecture is most similar to that of human skin, guinea pigs are cheaper to obtain than pigs and the care of those is cheaper and easier as well (24).
Based on the results from the wound closure between the ozone group and the oil group on days 0, 3, 5, 7, and 11, the ozone group showed a significantly smaller wound size and residual wound area than the oil group in the guinea pig model on days 5 and 7. Thus, these results demonstrated that O3 could enhance acute cutaneous wound healing. Especially, on day 7, both the wound size and residual wound area of the ozone group were much more significantly decreased. This implies that topical exposure of O3 may affect granulation tissue formation of the wound healing process rather than affecting immediate formation of blood clot and recruitment of inflammatory cells during the inflammation phase.
On Masson-trichrome staining on day 3, there was not much difference in the staining intensity of collagen fibers and fibroblast proliferation between the ozone group and the oil group. However, on day 7, the ozone group revealed about one and half times increased staining intensity of collagen fibers and significantly increased fibroblasts at the wound edge as well as at the wound bed, compared to the oil group. These findings indicated that O3 may act on acute wound healing directly or indirectly via collagen synthesis and fibroblast proliferation during granulation tissue formation and the early tissue remodeling phase of wound healing.
Fibroblasts have been known to play important roles in reepithelization, collagen fiber synthesis, extracellular matrix regeneration, remodeling of wounds, and for the release of such endogenous growth factors as FGF, PDGF, TGF-β, and VEGF (1, 25). In this study, increased expressions of PDGF and TGF-β were seen in the ozone group on day 7, which is correlated with increased staining intensity of collagen fibers and fibroblast proliferation on the same day. There were also increased expressions of PDGF and TGF-β in the epidermal keratinocytes and the hair follicular cells adjacent to the injury. These findings suggest that O3 might induce expressions of PDGF and TGF-β from epidermal keratinocyte as well as from the dermal fibroblast at the injury site. In the present study, there was a relatively increased expression of VEGF in the epidermal keratinocytes of the ozone group on day 7; this was consistent with the previous study showing that VEGF was gradually increased from the 1st day to the 7th day of normal wound healing process (26). On hematoxylin-eosin staining, the relatively increased vascularity in the ozone group might be due to the increased expression of VEGF by ozonation. These findings may be associated with the generation of H2O2 through ozonation, which can directly induce a VEGF expression and/or indirectly induce it by the induction of heme oxygenase-1 (27). Because VEGF is main cytokine of vascularization in the late phase of wound healing (28), further study will be needed to clarify the effect of O3 on the neovascularization of cutaneous wound healing. In contrast to the increased expressions of PDGF, TGF-β, and VEGF in the ozone group, there was little difference in the FGF expressions between the ozone group and the oil group on day 7. One of the possible explanations for this result is that FGF had already been up-regulated within 24 hr after wounding (29).
In conclusion, these results demonstrate that application of ozonated OLO, the topical form of O3, can accelerate acute cutaneous wound repair in the guinea pig model by promoting collagen synthesis and fibroblast proliferation at the injury site and by increasing the expression of growth factors such as PDGF, TGF-β, and VEGF. Taken together, we can infer that topical O3 may be regarded as an alternative therapeutic modality to enhance cutaneous wound healing.
Figures and Tables
Fig. 1
The effects of ozonated olive oil on clinical wound closure. The photomicrographs demonstrate the enhanced wound closure, in the ozone group, on the left two wounds (a and b) on the back of the guinea pig, as compared to the oil group (c) as well as the control group (d). (Bar=5 mm).

Fig. 2
Masson-trichrome staining of the wound bed and the edge of the injury site on days 3 and 7. The ozone group revealed the increased staining intensity of collagen fibers and the number of fibroblasts at the wound bed and edge, in comparison to the oil group and the control group on day 7, but not on day 3 (original magnification ×400, Bar=50 µm).
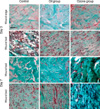
Fig. 3
Immunohistochemical staining for FGF, PDGF, TGF-β, and VEGF on day 7. The ozone group revealed the relatively increased expressions of PDGF (B, F, J), TGF-β (C, G, K), and VEGF (D, H, L), but not FGF (A, E, I) as compared to the oil group on day 7 (×100, original magnification, inlet; ×400, Bar=500 µm).
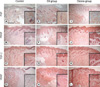
Table 1
Comparison of the average wound size and residual wound area on post-operation days 0, 3, 5, 7, and 11

References
1. Werner S, Grose R. Regulation of wound healing by growth factors and cytokines. Physiol Rev. 2003. 83:835–870.

2. Ornitz DM, Itoh N. Fibroblast growth factors. Genome Biol. 2001. 2:REVIEWS3005.
3. Heldin CH, Westermark B. Mechanism of action and in vivo role of platelet-derived growth factor. Physiol Rev. 1999. 79:1283–1316.

4. Lauer G, Sollberg S, Cole M, Flamme I, Sturzebecher J, Mann K, Krieg T, Eming SA. Expression and proteolysis of vascular endothelial growth factor is increased in chronic wounds. J Invest Dermatol. 2000. 115:12–18.

5. Valacchi G, Fortino V, Bocci V. The dual action of ozone on the skin. Br J Dermatol. 2005. 153:1096–1100.

6. Al-Dalain SM, Martinez G, Candelario-Jalil E, Menendez S, Re L, Giuliani A, Leon OS. Ozone treatment reduces markers of oxidative and endothelial damage in an experimental diabetes model in rats. Pharmacol Res. 2001. 44:391–396.
7. Martinez-Sanchez G, Al-Dalain SM, Menendez S, Re L, Giuliani A, Candelario-Jalil E, Alvarez H, Fernandez-Montequin JI, Leon OS. Therapeutic efficacy of ozone in patients with diabetic foot. Eur J Pharmacol. 2005. 523:151–161.
8. Bocci V. Ozone: a new medical drug. 2005. Dordrecht: Springer.
9. de Monte A, van der Zee H, Bocci V. Major ozonated autohemotherapy in chronic limb ischemia with ulcerations. J Altern Complement Med. 2005. 11:363–367.
10. Lim Y, Phung AD, Corbacho AM, Aung HH, Maioli E, Reznick AZ, Cross CE, Davis PA, Valacchi G. Modulation of cutaneous wound healing by ozone:differences between young and aged mice. Toxicol Lett. 2006. 160:127–134.
11. Gajendrareddy PK, Sen CK, Horan MP, Marucha PT. Hyperbaric oxygen therapy ameliorates stress-impaired dermal wound healing. Brain Behav Immun. 2005. 19:217–222.

12. Janic B, Umstead TM, Phelps DS, Floros J. Modulatory effects of ozone on THP-1 cells in response to SP-A stimulation. Am J Physiol Lung Cell Mol Physiol. 2005. 288:L317–L325.

13. Valacchi G, van der Vliet A, Schock BC, Okamoto T, Obermuller-Jevic U, Cross CE, Packer L. Ozone exposure activates oxidative stress responses in murine skin. Toxicology. 2002. 179:163–170.

14. Bocci V. Biological and clinical effects of ozone. Has ozone therapy a future in medicine? Br J Biomed Sci. 1999. 56:270–279.
15. Valacchi G, Bocci V. Studies on the biological effects of ozone: 10. Release of factors from ozonated human platelets. Mediators Inflamm. 1999. 8:205–209.

16. Lang DS, Jorres RA, Mucke M, Siegfried W, Magnussen H. Interactions between human bronchoepithelial cells and lung fibroblasts after ozone exposure in vitro. Toxicol Lett. 1998. 96-97:13–24.

17. Sen CK, Khanna S, Babior BM, Hunt TK, Ellison EC, Roy S. Oxidant-induced vascular endothelial growth factor expression in human keratinocytes and cutaneous wound healing. J Biol Chem. 2002. 277:33284–33290.

18. Sechi LA, Lezcano I, Nunez N, Espim M, Dupre I, Pinna A, Molicotti P, Fadda G, Zanetti S. Antibacterial activity of ozonized sunflower oil (Oleozon). J Appl Microbiol. 2001. 90:279–284.

19. Fischer SM. Is cyclooxygenase-2 important in skin carcinogenesis? J Environ Pathol Toxicol Oncol. 2002. 21:183–191.

20. Valacchi G, Pagnin E, Corbacho AM, Olano E, Davis PA, Packer L, Cross CE. In vivo ozone exposure induces antioxidant/stress-related responses in murine lung and skin. Free Radic Biol Med. 2004. 36:673–681.

21. Paramio JM, Casanova ML, Segrelles C, Mittnacht S, Lane EB, Jorcano JL. Modulation of cell proliferation by cytokeratins K10 and K16. Mol Cell Biol. 1999. 19:3086–3094.

22. Menendez S, Falcon L, Simon DR, Landa N. Efficacy of ozonized sunflower oil in the treatment of tinea pedis. Mycoses. 2002. 45:329–332.
23. Bocci V. Oxygen-ozone therapy: a critical evaluation. 2002. Dordrecht: Kluwer Academic Publisher.
24. FDA Wound Healing Clinical Focus Group. Guidance for industry: chronic cutaneous ulcer and burn wounds-developing products for treatment. Wound Repair Regen. 2001. 9:258–268.
25. Vincent F. Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, editors. Mechanisms of cutaneous wound repair. Fitzpatrick's dermatology in general medicine. 2003. 6th ed. New York: McGraw Hill;236–246.
26. Detmar M, Brown LF, Berse B, Jackman RW, Elicker BM, Dvorak HF, Claffey KP. Hypoxia regulates the expression of vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) and its receptors in human skin. J Invest Dermatol. 1997. 108:263–268.

27. Jazwa A, Loboda A, Golda S, Cisowski J, Szelag M, Zagorska A, Sroczynska P, Drukala J, Jozkowicz A, Dulak J. Effect of heme and heme oxygenase-1 on vascular endothelial growth factor synthesis and angiogenic potency of human keratinocytes. Free Radic Biol Med. 2006. 40:1250–1263.

28. Nissen NN, Polverini PJ, Koch AE, Volin MV, Gamelli RL, DiPietro LA. Vascular endothelial growth factor mediates angiogenic activity during the proliferative phase of wound healing. Am J Pathol. 1998. 152:1445–1452.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download