Abstract
Preterm labor after 34 weeks of gestation has shown no great difference from full-term labor in terms of neonatal morbidity and mortality when proper antepartum management (antibiotics or steroids treatment) is done. However, various studies have discussed different views on the risks and safety of preterm delivery at 32+0-33+6 weeks of gestation. We evaluated the complications of different preterm groups that included the neonates born at 32+0-33+6 weeks of gestation (142), stratified randomly selected neonates born at 34+0-36+6 weeks of gestation (267) and neonates born after 37+0 weeks of gestation (356) at our hospital between December 1999 and April 2006. As a result, it was found that neonates born at 34+0-36+6 weeks of gestation showed no great difference from infants born at full term. However, neonates born at 32+0-33+6 weeks were more likely to be admitted to neonatal intensive care unit or develop neonatal complications significantly than the neonates born at 34+0-36+6 weeks and at full term. Therefore, it is suggested that neonates born at 32+0-33+6 weeks have higher risk of neonatal complications following their preterm labor than those born at later than 34+0 weeks. Thus, it would be difficult to accept the idea that preterm labor at 32+0-33+6 weeks is safe.
Preterm labor and preterm delivery are critical factors regarding neonatal morbidity and mortality (1, 2) Much efforts have been made on prediction, prevention, diagnosis and treatment of preterm labor, and several previous studies including the one reported by our university hospital (3) have shown that there is no great difference regarding neonatal morbidity and mortality between the group with preterm labor after 34 weeks of gestation and the full-term labor group when proper ante partum management (antibiotics, steroids treatment) was provided (4-15). Also, it has been demonstrated that the rates of neonatal mortality and complications are significant in preterm babies born prior to 32 weeks of gestation and with body weight less than 1,500 g (16, 17). However, relevant studies have expressed diverse opinions about the risks and safety of preterm infants born at 32+0-33+6 weeks of gestation (18-20). According to recent studies, the upper limit of the usage of the uterine contraction inhibitor for pregnant women with preterm labor has not been clearly decided (21); yet, some studies even argue that there is no significant difference in perinatal morbidity and mortality between pregnant women who stopped taking uterine contraction inhibitor after 32 weeks of gestation and those who continued taking it (22). Therefore, this study aimed to examine the perinatal outcomes of different groups on the neonates' conditions at delivery and the neonates' morbidity and mortality in order to see if preterm labor at 32+0-33+6 weeks is more dangerous than at other periods; and when they are safe, if uterine contraction inhibitor can be used as early as at 32+0 weeks.
This study identifies safe gestational age of neonates when mothers go through preterm labor in order to provide an index to determine whether or not to maintain pregnancy when there is a risk of preterm delivery due to preterm labor or premature rupture of membranes (PROM).
A pregnant woman with preterm labor experiences physical pain daily. Therefore, it would be a considerable help for pregnant women as well as doctors if it is possible to bring gestational age with no post-delivery complications significantly different full-term delivery sooner.
Our research subjects consisted of neonates born at 32+0-33+6 weeks (184 babies), 34+0-36+6 weeks (472 babies), and later than 37+0 weeks (2,353 babies) at Inje University Ilsan Paik hospital from December 1999 to April 2006. The data were analyzed on the following categories: the conditions of the mother and the fetus prior to delivery, the condition of the neonate at birth, and neonatal morbidity and mortality. We excluded the cases of twins, fetal death in utero, and improper records (inaccurate gestational week or incorrect records of the mother and the neonates). Consequently, 142, 267, and 356 cases were examined for the 32+0-33+6 week group, the 34+0-36+6 weeks group and the more than 37+0 weeks group, respectively, and the last two groups were selected by using stratified random sampling.
For mothers, age, obstetrics history (term or preterm delivery), experience of preterm labor, chorioamnionitis, PROM, hypertensive disorders of pregnancy, placenta previa, gestational diabetes mellitus (GDM), and anemia were considered, and fetal distress, prolapse of the umbilical cord, breech presentation, hydramnios, and oligohydramnios were included in antepartum evaluation of the fetal condition to find out the correlation with the preterm delivery. Evaluation of antepartum management included the proper use of antibiotics and steroids treatment (when a mother had 2 or more maternal complications, "2 or more complications" was recorded). A neonate's body weight, sex, Apgar score (1' and 5'), and the presence of meconium staining were examined right after birth. Also, the categories including admission history of neonatal intensive care unit (NICU) and the period of stay in NICU, neonatal convulsion, intraventricular hemorrhage (IVH), neonatal retinopathy, sepsis, respiratory distress syndrome (RDS), ventilator care and the rates of fetal death were used as indicators for neonatal morbidity and mortality.
Statistical analyses of the data were conducted by using chi-square examination, Fisher's exact test, analysis of variance (ANOVA), and multiple logistic regression analysis, using the SAS program. We recognized the values with a p value less than 0.05 as significant.
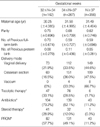
The results on the condition and treatment of pregnant women before delivery in the three groups -32+0-33+6 weeks, 34+0-36+6 weeks, and later than 37+0 weeks of gestation-(Table 1) found no significant difference by maternal age and history of pregnancy that could be considered to affect preterm delivery. PROM frequencies were significantly high in the 32+0-33+6 weeks group and the 34+0-36+6 weeks group (57.7% and 49.1%), which might be related to the increased use of the antibiotics as a preventive treatment in these groups (73.2% and 52.1%). The rate of uterine contraction inhibitor use to prevent preterm labor was significantly high in the 32+0-33+6 weeks group. Also, the rate of steroid use for RDS was high up to 28.9%; and as expected, the high rate of tocolysis and steroid use was found in the groups before 34 weeks. However, using steroid after 34 weeks included mothers treated with steroid before 34 weeks and delivered babies after 34 weeks.
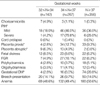
When maternal complications were compared (Table 2), it was found that of maternal complications in the 32+0-33+6 weeks group, the occurrence rates of preeclampsia, placenta previa, placenta abruptio, and GDM were significantly high. Eight factors that were found to be significant in Table 1, 2 were considered as confounding variables in analysis of neonatal morbidity.
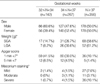
When the neonatal conditions at birth were compared by gestational weeks (Table 3), the Apgar score less than 7 points at 1 min was significant among the neonates born at 32+0-33+6 weeks of gestation (41.5%), and large for gestational age (LGA) baby weight and meconium staining were also found to be significant.
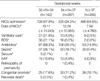
Table 4 shows the results of comparing the neonatal diseases that affect neonatal morbidity. The neonates born after 32+0-33+6 weeks of gestation had significantly high probability of hospitalization to NICU (97.9%) and the average period of stay was longer by 19.11 days. Neonatal complications, including ventilator care, RDS, use of antibiotics, sepsis, seizure, IVH, neonatal death, and congenital anomaly were significant.
Preeclampsia, placenta previa, placenta abruptio, GDM, use of antibiotics, steroid, tocolysis, and PROM were also found to be significantly high in the 32+0-33+6 weeks group. We conducted multiple logistic regression analysis to exclude the effect of confounding factors on the occurrence of neonatal morbidity; and, the result showed that administration of NICU, ventilator care, using of antibiotics, and neonatal complications like RDS seizure, and IVH were statistically significant (Table 5).
Because the 32+0-33+6 weeks group had significantly high occurrence of neonatal complications, we conducted further analysis regarding the effect of steroid on neonatal complications (Table 6). However, this study's statistical findings did not support the idea that steroid treatment for the neonates born at 32+0-33+6 weeks of gestation has a significant effect on the occurrence of neonatal complications.
Preterm delivery is one of the most important factors of neonatal mortality and morbidity not only in South Korea (22) but also in the United States (23). It has been reported that excluding congenital anomaly, 85% of neonatal mortality is caused by preterm delivery, and in many countries, a tremendous amount of cost is demanded to treat perinatal diseases.
Until now, the expenditure allotted for the treatment of preterm infants was not efficiently used because the factors that affect perinatal morbidity and mortality and delivery time of preterm labor have not been clearly identified.
Recently, the incidence of perinatal death has been considerably decreased as the neonatology has been making its advance, but it is still a critical issue to manage morbidity and mortality after preterm delivery. Gestational age can be one of the most important predictors of morbidity and mortality of neonates. It is clear that the rates of infant survival and of being born as normal infants are high as gestational age approaches close to the full term.
In this study, the incidence of PROM, preeclampsia, placenta previa, placenta abruptio and GDM were significantly high in the 32+0-33+6 weeks group, which once again demonstrated that PROM is one of the most important decision factors of preterm delivery.
The high incidence of using tocolysis, antibiotics, steroids at 32 gestational weeks is thus inevitable. And maternal complications were analyzed as shown in Table 1, 2 to conduct accurate statistical processing with significant maternal complications considered as confounding variables.
According to Helen et al. (21), the rate of admission to NICU was high for the preterm infant born at 32+0-33+6 weeks of gestation, but our study found that 97.9% of the preterm infant born at 32+0-33+6 weeks and 84.3% of the preterm infant born at 34+0-36+6 weeks were admitted to NICU. These figures can be attributed to the fact that the admission rate of preterm infants to NICU is somewhat higher in Korea as preterm infants, even those with no severe problems, are tended to be observed in NICU for some period of time. However, even excluding these cases, the rate of admission to NICU is still high.
One minute Apgar score included in neonatal conditions at birth was significantly low in a previous study by Inje University Ilsan Paik hospital (15). In this study with a higher number of samples, only one minute Apgar score was significantly low. This reaffirms that in the case of preterm infants born at 32+0-33+6 weeks, the rate of the necessity of immediate cardiopulmonary resuscitation is significantly higher than the 34+0-36+6 weeks group.
In terms of prenatal complications, the neonates born before 32+0-33+6 weeks of gestation was overall weak, excluding the cases of sepsis, neonatal death, retinopathy of prematurity, and congenital anomaly. Jones et al. (24) reported that the incidence of admission to NICU and the rate of RDS occurrence markedly decreased as the gestational age advances after 34 weeks. However, the rate of neonatal morbidity did not significantly decline after 34 weeks of gestation. Our study confirmed that the incidence of admission to NICU and the rate of RDS occurrence had little difference between the 32+0-33+6 weeks group and the 34+0-36+6 weeks group.
According to Grgić et al. (25) and Crowther et al. (26), the complications related to neonatal respiratory system have noticeably decreased when steroids were used to the neonates born prior to 32+0-33+6 weeks. In order to examine the rate of complications in the respiratory system, the 32+0-33+6 weeks group was divided into the group with steroidal treatment and the group without it were compared, but it was not found that steroid treatment had significantly reduced neonatal complications. Despite the extensive samples, the results showed no significant efficiency of steroid on respiratory maturity. This might be explained that only the use of ventilator care and the presence of RDS were used to evaluate the efficiency of steroidal treatment; we expect to have more precise results if the period of time using ventilator care and the grade of RDS were included. Our university is planning to conduct further research with more samples and more detailed analysis of the preterm labor at 32+0-33+6 weeks and 34+0-36+6 weeks of gestation.
Although it is known that the survival rates of full-term infants and preterm infants after 34 weeks of gestation are of no difference, there existed a debate regarding ante partum management of preterm infants born at 32+0-33+6 weeks. Throughout this study we expected earlier preterm delivery by several days from 34+0 weeks having appropriate antepartum management. However, the neonatal complications compared between preterm infants born at 32+0-33+6 weeks and at full term showed very significant differences from which we concluded that active management is necessary in order to keep up gestational age as close to full term as possible.
This study aimed at clarifying the claim that there was no significant difference in preterm labor between 32+0-33+6 weeks and 34+0-36+6 weeks based on the data generated from the patients of our hospital. However, neonatal complications of the preterm labor at 32+0-33+6 weeks are significantly high that it is difficult to fully accept the notion that preterm labor at 32+0-33+6 weeks is safe.
Figures and Tables
References
2. Lewit EM, Baker LS, Corman H, Shiono PH. The direct cost of low birth weight. Future Child. 1995. 5:35–56.

3. Ahn JH, Choi HM, Hwang YS, Hong SH, Jung BJ, Jeon MK, Lee ES. Preterm delivery between 34-36 weeks of gestation; is it danger? Korean J Obstet Gynecol. 2002. 45:84–88.
4. Liggins GC, Howie RN. A controlled trial of antepartum glucocorticoid treatment for prevention of the respiratory distress syndrome in premature infants. Pediatrics. 1972. 50:515–525.
5. Rush RW, Keirse MJ, Howat P, Baum JD, Anderson AB, Turnbull AC. Contribution of preterm delivery to perinatal mortality. Br Med J. 1976. 2:965–968.

6. Wesselius-de Casparis A, Thiery M, Yo le Sian A, Baumgarten K, Brosens I, Gamisans O, Stolk JG, Vivier W. Results of double-blind, multicentre study with ritodrine in premature labour. Br Med J. 1971. 3:144–147.
7. Ingemarsson I. Effect of terbutaline on premature labor. A double-blind placebo-controlled study. Am J Obstet Gynecol. 1976. 125:520–524.
8. Spellacy WN, Cruz AC, Birk SA, Buhi WC. Treatment of premature labor with ritodrine: a randomized controlled study. Obstet Gynecol. 1979. 54:220–223.
9. Larsen JF, Hansen MK, Hesseldahl H, Kristoffersen K, Larsen PK, Osler M, Weber J, Eldon K, Lange A. Ritodrine in the treatment of preterm labour. A clinical trial to compare a standard treatment with three regimens involving the use of ritodrine. Br J Obstet Gynaecol. 1980. 87:949–957.

10. Larsen JF, Eldon K, Lange AP, Leegaard M, Osler M, Olsen JS, Permin M. Ritodrine in the treatment of preterm labor: second Danish Multicenter Study. Obstet Gynecol. 1986. 67:607–613.
11. Canadian Preterm Labor Investigators Group. Treatment of preterm labor with the beta-adrenergic agonist ritodrine. N Engl J Med. 1992. 327:308–312.
12. Gamissans O, Canas E, Cararach V, Ribas J, Puerto B, Edo A. A study of indomethacin combined with ritodrine in threatened preterm labor. Eur J Obstet Gynecol Reprod Biol. 1978. 8:123–128.

13. Spearing G. Alcohol, indomethacin, and salbutamol. A comparative trial of their use in preterm labor. Obstet Gynecol. 1979. 53:171–174.
14. Niebyl JR, Blake DA, White RD, Kumor KM, Dubin NH, Robinson JC, Egner PG. The inhibition of premature labor with indomethacin. Am J Obstet Gynecol. 1980. 136:1014–1019.

15. Zuckerman H, Shalev E, Gilad G, Katzuni E. Further study of the inhibition of premature labor by indomethacin. Part I. J Perinat Med. 1984. 12:19–23.

16. Stevenson DK, Wright LL, Lemons JA, Oh W, Korones SB, Papile LA, Bauer CR, Stoll BJ, Tyson JE, Shankaran S, Fanaroff AA, Donovan EF, Ehrenkranz RA, Verter J. Very low birth weight outcomes of the National Institute of Child Health and Human Development Neonatal Research Network, January 1993 through December 1994. Am J Obstet Gynecol. 1998. 179:1632–1639.

17. Bottoms SF, Paul RH, Mercer BM, MacPherson CA, Caritis SN, Moawad AH, Van Dorsten JP, Hauth JC, Thurnau GR, Miodovnik M, Meis PM, Roberts JM, McNellis D, Iams JD. Obstetric determinants of neonatal survival: antenatal predictors of neonatal survival and morbidity in extremely low birth weight infants. Am J Obstet Gynecol. 1999. 180:665–669.

18. Copper RL, Goldenberg RL, Creasy RK, DuBard MB, Davis RO, Entman SS, Iams JD, Cliver SP. A multicenter study of preterm birth weight and gestational age-specific neonatal mortality. Am J Obstet Gynecol. 1993. 168:78–84.

19. Robertson PA, Sniderman SH, Laros RK Jr, Cowan R, Heilbron D, Goldenberg RL, Iams JD, Creasy RK. Neonatal morbidity according to gestational age and birth weight from five tertiary care centers in the United States, 1983 through 1986. Am J Obstet Gynecol. 1992. 166:1629–1641.

20. Berkman ND, Thorp JM Jr, Lohr KN, Carey TS, Hartmann KE, Gavin NI, Hasselblad V, Idicula AE. Tocolytic treatment for the management of preterm labor: a review of the evidence. Am J Obstet Gynecol. 2003. 188:1648–1659.

21. How HY, Zafaranchi L, Stella CL, Recht K, Maxwell RA, Sibai BM, Spinnato JA. Tocolysis in women with preterm labor between 32 0/7 and 34 6/7 weeks of gestation: a randomized controlled pilot study. Am J Obstet Gynecol. 2006. 194:976–981.

22. Oh BJ, Lee BG. Epidemiology of the preterm birth. Korean J Obstet Gynecol. 1990. 33:188–201.
23. McCormick MC. The contribution of low birth weight to infant mortality and childhood morbidity. N Engl J Med. 1985. 312:82–90.

24. Jones JS, Istwan NB, Jacques D, Coleman SK, Stanziano G. Is 34 weeks an acceptable goal for a complicated singleton pregnancy? Manag Care. 2002. 11:42–47.
25. Grgić G, Fatusić Z, Bogdanović G. Stimulation of fetal lung maturation with dexamethasone in unexpected premature labor. Med Arh. 2003. 57:291–294.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download